Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 in NaCl
in NaClAoyama, Takahito; Ogawa, Hiroaki; Kato, Chiaki; Ueno, Fumiyoshi
Metals, 11(3), p.511_1 - 511_13, 2021/03
Times Cited Count:3 Percentile:23.95(Materials Science, Multidisciplinary)The effect of Cu in bulk solution on pitting corrosion resistance of extra high purity type 316 stainless steel was investigated. Pitting occurred in 0.1 M NaCl-1 mM CuCl
in bulk solution on pitting corrosion resistance of extra high purity type 316 stainless steel was investigated. Pitting occurred in 0.1 M NaCl-1 mM CuCl whereas pitting was not initiated in 0.1 M NaCl. Although deposition of Cu
whereas pitting was not initiated in 0.1 M NaCl. Although deposition of Cu on the surface occurred regardless of potential region in 0.1 M NaCl-1 mM CuCl
on the surface occurred regardless of potential region in 0.1 M NaCl-1 mM CuCl , Cu
, Cu in bulk solution had no influence on the passive film formation. The decrease in pitting corrosion resistance in 0.1 M NaCl-1 mM CuCl
in bulk solution had no influence on the passive film formation. The decrease in pitting corrosion resistance in 0.1 M NaCl-1 mM CuCl resulted from the deposited Cu or Cu compound and continuous supply of Cu
resulted from the deposited Cu or Cu compound and continuous supply of Cu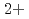 on the surface.
on the surface.
Yamamoto, Masahiro
Zairyo To Kankyo 2020 Koenshu (CD-ROM), p.9 - 16, 2020/05
The author has been continuing research and development for corrosion science for about forty years. One of the main targets of his research is applying computational science techniques on corrosion problems. The results are briefly introduced in this article. Also, the author organized some workshop for corrosion problems of 1F decommissioning procedure for several years. Such activities are evaluated for receiving the society award in JSCE.

 generation; The Role of NO
generation; The Role of NO
 in the crevice corrosion repassivation of type 316L stainless steel
in the crevice corrosion repassivation of type 316L stainless steelAoyama, Takahito; Sugawara, Yu*; Muto, Izumi*; Hara, Nobuyoshi*
Journal of the Electrochemical Society, 166(10), p.C250 - C260, 2019/01
Times Cited Count:5 Percentile:16.43(Electrochemistry)The role of NO
 in the repassivation of crevice corrosion of Type 316L stainless steel was investigated. In crevice corrosion tests, the solution was changed from 1 M NaCl to NaCl-NaNO
in the repassivation of crevice corrosion of Type 316L stainless steel was investigated. In crevice corrosion tests, the solution was changed from 1 M NaCl to NaCl-NaNO . NO
. NO
 led to complete repassivation. Repassivation of the crevice corrosion was found to take place in two steps. In the first step, the estimated current density inside the crevice gradually decreased from ca. 5 mA cm
led to complete repassivation. Repassivation of the crevice corrosion was found to take place in two steps. In the first step, the estimated current density inside the crevice gradually decreased from ca. 5 mA cm to ca. 5
to ca. 5  A cm
A cm . After that, the current density suddenly decreased to less than 0.1
. After that, the current density suddenly decreased to less than 0.1  A cm
A cm . From the potentiodynamic polarization in acidic solutions simulated inside the crevice (pH 0.2) and in situ observations of the crevice corrosion morphology, the first step was thought to be generated by the suppression of active dissolution by NO
. From the potentiodynamic polarization in acidic solutions simulated inside the crevice (pH 0.2) and in situ observations of the crevice corrosion morphology, the first step was thought to be generated by the suppression of active dissolution by NO
 . It would appear that the generation of NH
. It would appear that the generation of NH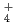 results in a pH increase and the further suppression of active dissolution, and then repassivation occurs.
results in a pH increase and the further suppression of active dissolution, and then repassivation occurs.
Shibata, Toshio*; *; *; Tsuru, Toru*; Inoue, Hiroyuki*
JNC TJ8400 2000-013, 38 Pages, 2000/02
None
 on crevice corrosion of type 316L stainless steel
on crevice corrosion of type 316L stainless steelAoyama, Takahito; Kato, Chiaki
no journal, ,
Alloyed Cu is known to inhibit the growth of pitting corrosion on stainless steels after pitting initiation. The Cu dissolved from the stainless steel matrix acts as an inhibitor in acidic chloride environments which is formed in pits. The Cu
dissolved from the stainless steel matrix acts as an inhibitor in acidic chloride environments which is formed in pits. The Cu suppresses active dissolution rate inside the pits
suppresses active dissolution rate inside the pits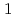 . This suppression effect is supposed to be also effective for active dissolution which promotes crevice corrosion. Therefore, if it is possible to introduce Cu
. This suppression effect is supposed to be also effective for active dissolution which promotes crevice corrosion. Therefore, if it is possible to introduce Cu to the inside of a crevice from the outside, introduced Cu
to the inside of a crevice from the outside, introduced Cu is supposed to inhibit crevice corrosion on stainless steel. However, diffusion of ions between inside and outside of the crevice is restricted by its geometry, and Cu
is supposed to inhibit crevice corrosion on stainless steel. However, diffusion of ions between inside and outside of the crevice is restricted by its geometry, and Cu does not migrate to inside of a crevice according to the electroneutrality principle. [Cu (EDTA (ethylenediaminetetraacetic acid))]
does not migrate to inside of a crevice according to the electroneutrality principle. [Cu (EDTA (ethylenediaminetetraacetic acid))] is a chelated Cu
is a chelated Cu which has negative charge, and is expected to migrate to inside of a crevice from the outside by electrochemical migration. In addition, it is reported that Cu
which has negative charge, and is expected to migrate to inside of a crevice from the outside by electrochemical migration. In addition, it is reported that Cu in [Cu(EDTA)]
in [Cu(EDTA)] could be easily substituted by Fe
could be easily substituted by Fe in low pH
in low pH . Therefore, Cu
. Therefore, Cu is considered to be introduced to the inside of the crevice as [Cu(EDTA)]
is considered to be introduced to the inside of the crevice as [Cu(EDTA)] , and to affect crevice corrosion of stainless steel. In this study, in situ observation of an inside of the crevice
, and to affect crevice corrosion of stainless steel. In this study, in situ observation of an inside of the crevice was performed to analyze the effect of [Cu(EDTA)]
was performed to analyze the effect of [Cu(EDTA)] on crevice corrosion.
on crevice corrosion.