Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 glasses
glassesHosokawa, Shinya*; Kawakita, Yukinobu; Stellhorn, J. R.*; Pusztai, L.*; Blanc, N.*; Boudet, N.*; Ikeda, Kazutaka*; Otomo, Toshiya*
JPS Conference Proceedings (Internet), 33, p.011070_1 - 011070_7, 2021/03
Local- and intermediate-range atomic order in Ag ion conducting glasses Ag (GeSe
(GeSe )
) with x = 0.15, 0.28, 0.33, and 0.50 were investigated by using a combination of AXS, XRD, ND, and RMC modeling. By adding the ND pdf to AXS and XRD results, reasonable partial structure factors and partial pdf were obtained by the RMC procedure. In contrast to the previous AXS and RMC study, a large number of Ag-Ge and Ge-Ge correlations are observed in the first coordination shell region, which is consistent with an
with x = 0.15, 0.28, 0.33, and 0.50 were investigated by using a combination of AXS, XRD, ND, and RMC modeling. By adding the ND pdf to AXS and XRD results, reasonable partial structure factors and partial pdf were obtained by the RMC procedure. In contrast to the previous AXS and RMC study, a large number of Ag-Ge and Ge-Ge correlations are observed in the first coordination shell region, which is consistent with an  MD simulation. The coordination numbers around the Ge and Se mostly follow the 8-
MD simulation. The coordination numbers around the Ge and Se mostly follow the 8- rule over all Ag concentrations if Ag is not taken into account. With increasing the Ag concentration, the partial coordination numbers with Ge and Se atoms around Ag remarkably increases, while the Ag-Ag coordination number increases only slightly, indicating that the Ag conducting path is formed through the second neighboring Ag-Ag correlations.
rule over all Ag concentrations if Ag is not taken into account. With increasing the Ag concentration, the partial coordination numbers with Ge and Se atoms around Ag remarkably increases, while the Ag-Ag coordination number increases only slightly, indicating that the Ag conducting path is formed through the second neighboring Ag-Ag correlations.

Okamoto, Yoshihiro; Yaita, Tsuyoshi; Minato, Kazuo
Journal of Non-Crystalline Solids, 333(2), p.182 - 186, 2004/02
Times Cited Count:11 Percentile:68.45(Materials Science, Ceramics)The local structure of solid and molten SrCl was investigated by X-ray absorption fine structure(XAFS) technique. A superionic conduction state was observed below the melting point. The XAFS data at molten state was obtained at 1000
was investigated by X-ray absorption fine structure(XAFS) technique. A superionic conduction state was observed below the melting point. The XAFS data at molten state was obtained at 1000 C. According to the curve fitting analysis, the nearest Sr
C. According to the curve fitting analysis, the nearest Sr -Cl
-Cl distance and the coordination number are 2.99
distance and the coordination number are 2.99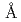 and 6.6 in molten state. XAFS functions calculated from molecular dynamics simulation results were copmpared with the experimental XAFS data.
and 6.6 in molten state. XAFS functions calculated from molecular dynamics simulation results were copmpared with the experimental XAFS data.
Sakuma, Takaaki*; Nakamura, Y.*; Hirota, M.*; Murakami, A.*; Ishii, Yoshinobu
Solid State Ionics, 127(3-4), p.295 - 300, 2000/01
Times Cited Count:8 Percentile:44.56(Chemistry, Physical)no abstracts in English
Sakuma, T.*; *; Murakami, A.*; *; Ishii, Yoshinobu
High Temp. Mater. Process., 18(1-2), p.41 - 48, 1999/00
no abstracts in English
*; *; *; Watanabe, Mamoru*; *; Morii, Yukio; Shimojo, Yutaka
Solid State Ionics, 113-115, p.471 - 475, 1998/00
Times Cited Count:3 Percentile:22.09(Chemistry, Physical)no abstracts in English
 Se
Se*; *; *; Shimojo, Yutaka; Morii, Yukio
Physica B; Condensed Matter, 213-214, p.399 - 401, 1995/00
Times Cited Count:12 Percentile:60.18(Physics, Condensed Matter)no abstracts in English

;
Solid State Ionics, 51, p.75 - 78, 1992/00
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)no abstracts in English
 (AgPO
(AgPO )
) glasses
glasses*; *; *; Funahashi, Satoru
Solid State Ionics, 53-56, p.1164 - 1167, 1992/00
Times Cited Count:5 Percentile:34.63(Chemistry, Physical)no abstracts in English
Kawakita, Yukinobu; Tahara, Shuta*
no journal, ,
Kawakita, Yukinobu; Tahara, Shuta*; Kikuchi, Tatsuya; Nakamura, Mitsutaka; Inamura, Yasuhiro; Maruyama, Kenji*; Yamauchi, Yasuhiro*; Nakajima, Kenji; Kawamura, Seiko; Kambara, Wataru*
no journal, ,
no abstracts in English