Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 neutron imaging and diffraction analysis revealing spatial lithiation phase evolution in an ultra-thick graphite electrode
neutron imaging and diffraction analysis revealing spatial lithiation phase evolution in an ultra-thick graphite electrodeStrobl, M.*; Baur, M. E.*; Samothrakitis, S.*; Molamud, F.*; Zhang, X.*; Tung, P. K. M.*; Schmidt, S.*; Woracek, R.*; Lee, J.*; Kiyanagi, Ryoji; et al.
Advanced Energy Materials, p.2405238_1 - 2405238_9, 2025/01
Times Cited Count:0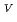 O
O
 /U
/U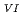 O
O
 redox equilibrium in [emim]Tf
redox equilibrium in [emim]Tf N-based liquid electrolytes towards uranium redox-flow battery
N-based liquid electrolytes towards uranium redox-flow batteryTakao, Koichiro*; Ouchi, Kazuki; Komatsu, Atsushi; Kitatsuji, Yoshihiro; Watanabe, Masayuki
European Journal of Inorganic Chemistry, 27(14), p.e202300787_1 - e202300787_7, 2024/05
Times Cited Count:1 Percentile:0.00(Chemistry, Inorganic & Nuclear)Electrochemical behavior of U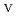 O
O
 /U
/U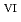 O
O
 in 1-ethyl-3-methylimidazolium bis(trifluoromethyl)sulfonylamide ([emim]Tf
in 1-ethyl-3-methylimidazolium bis(trifluoromethyl)sulfonylamide ([emim]Tf N) ionic liquid was studied to clarify what are required to attain its redox reversibility for utilizing depleted U as an electrode active material in a redox flow battery. As a result, reversibility of the U
N) ionic liquid was studied to clarify what are required to attain its redox reversibility for utilizing depleted U as an electrode active material in a redox flow battery. As a result, reversibility of the U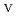 O
O
 /U
/U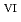 O
O
 redox reaction was successfully achieved in use of a glassy carbon working electrode under presence of Cl
redox reaction was successfully achieved in use of a glassy carbon working electrode under presence of Cl in [emim]Tf
in [emim]Tf N. To improve diffusivity of solutes, [emim]Tf
N. To improve diffusivity of solutes, [emim]Tf N diluted with an auxiliary molecular solvent, N,N-dimethylformamide (DMF). We have succeeded in demonstrating a reversible redox reaction of [U
N diluted with an auxiliary molecular solvent, N,N-dimethylformamide (DMF). We have succeeded in demonstrating a reversible redox reaction of [U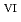 O
O Cl
Cl ]
] + e
+ e = [U
= [U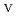 O
O Cl
Cl ]
] in the 50:50 v/v [emim]Tf
in the 50:50 v/v [emim]Tf N-DMF liquid electrolyte containing Cl
N-DMF liquid electrolyte containing Cl .
.
Ieda, Junichi
Kozan, 77(1), p.15 - 23, 2024/01
no abstracts in English
Hirano, Tatsumi*; Maeda, Takehiro*; Murata, Tetsuyuki*; Yamamoto, Takahiro*; Matsubara, Eiichiro*; Shobu, Takahisa; Shiro, Ayumi*; Yasuda, Ryo*; Takamatsu, Daiko*
SPring-8/SACLA Riyo Kenkyu Seikashu (Internet), 11(5), p.345 - 353, 2023/10
no abstracts in English
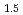 M
M O
O cathode materials for aqueous photo-rechargeable batteries
cathode materials for aqueous photo-rechargeable batteriesShimokawa, Kohei*; Matsubara, Shogo*; Kawaguchi, Tomoya*; Okamoto, Akihiro*; Ichitsubo, Tetsu
Chemical Communications, 59(51), p.7947 - 7950, 2023/06
Times Cited Count:2 Percentile:21.35(Chemistry, Multidisciplinary) frameworks
frameworksShimokawa, Kohei*; Hatakeyama, Takuya*; Li, H.*; Ichitsubo, Tetsu
Current Opinion in Electrochemistry, 38, p.101209_1 - 101209_8, 2023/04
Times Cited Count:6 Percentile:22.85(Chemistry, Physical)Ye, X.*; Shimokawa, Kohei*; Kezuka, Yuto*; Hatakeyama, Takuya*; Li, H.*; Ichitsubo, Tetsu
Journal of Physical Chemistry C, 127(11), p.5210 - 5218, 2023/03
Times Cited Count:2 Percentile:20.33(Chemistry, Physical) Sr
Sr CoO
CoO at initial electrochemical process in an alkaline solution
at initial electrochemical process in an alkaline solutionMatsuzaki, Akira*; Hirayama, Masaaki*; Oguchi, Shoya*; Komo, Mamoru*; Ikezawa, Atsunori*; Suzuki, Kota*; Tamura, Kazuhisa; Arai, Hajime*; Kanno, Ryoji*
Electrochemistry (Internet), 90(10), p.107001_1 - 107001_8, 2022/10
Times Cited Count:0 Percentile:0.00(Electrochemistry)Oxygen reduction and evolution reactions (ORR and OER) of perovskite-type La Sr
Sr CoO
CoO were characterized using two-dimensional model electrodes with different reaction planes. Synthesized by pulsed laser deposition, these thin and flat electrodes can reveal the reaction plane dependence of the ORR activity. From steady-state polarization measurements in KOH (aq.), the ORR activity was the highest on the (001) film during the first ORR/OER cycle, and it decreased significantly during the second cycle. In-situ synchrotron X-ray diffraction clarified crystal structure changes in the bulk and surface regions of La
were characterized using two-dimensional model electrodes with different reaction planes. Synthesized by pulsed laser deposition, these thin and flat electrodes can reveal the reaction plane dependence of the ORR activity. From steady-state polarization measurements in KOH (aq.), the ORR activity was the highest on the (001) film during the first ORR/OER cycle, and it decreased significantly during the second cycle. In-situ synchrotron X-ray diffraction clarified crystal structure changes in the bulk and surface regions of La Sr
Sr CoO
CoO , and these changes are associated with forming oxygen defects during the initial electrochemical process. Furthermore, the La
, and these changes are associated with forming oxygen defects during the initial electrochemical process. Furthermore, the La Sr
Sr CoO
CoO surface partially decomposed upon reacting. Therefore, the interfacial structures formed in the electrochemical reaction field is important for enhancing ORR and OER activities.
surface partially decomposed upon reacting. Therefore, the interfacial structures formed in the electrochemical reaction field is important for enhancing ORR and OER activities.
 /Br
/Br
 redox mediator for Li-Air batteries
redox mediator for Li-Air batteriesIto, Kimihiko*; Matsumura, Daiju; Song, C.*; Kubo, Yoshimi*
ACS Energy Letters (Internet), 7(6), p.2024 - 2028, 2022/06
Times Cited Count:6 Percentile:39.26(Chemistry, Physical)Ouchi, Kazuki; Komatsu, Atsushi; Takao, Koichiro*; Kitatsuji, Yoshihiro; Watanabe, Masayuki
Chemistry Letters, 50(6), p.1169 - 1172, 2021/06
Times Cited Count:2 Percentile:3.95(Chemistry, Multidisciplinary)The electrochemical behavior of uranium (IV) tetrachloride in ionic liquid-DMF mixture was studied for first time in order to build a redox flow battery (RFB) using U as an electrode active material. We found a quasi-reversible U /U
/U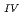 couple that could be applied to the anode reaction of the RFB.
couple that could be applied to the anode reaction of the RFB.
 GeP
GeP S
S
Yajima, Takeshi*; Hinuma, Yoyo*; Hori, Satoshi*; Iwasaki, Rui*; Kanno, Ryoji*; Ohara, Takashi; Nakao, Akiko*; Munakata, Koji*; Hiroi, Zenji*
Journal of Materials Chemistry A, 9(18), p.11278 - 11284, 2021/05
Times Cited Count:32 Percentile:82.59(Chemistry, Physical)Higuchi, Yuki*; Setoyama, Daigo*; Isegawa, Kazuhisa; Tsuchikawa, Yusuke; Matsumoto, Yoshihiro*; Parker, J. D.*; Shinohara, Takenao; Nagai, Yasutaka*
Physical Chemistry Chemical Physics, 23(2), p.1062 - 1071, 2021/01
Times Cited Count:13 Percentile:64.66(Chemistry, Physical)This study is the first report on liquid water and ice imaging conducted at a pulsed spallation neutron source facility. Neutron imaging can be utilised to visualise the water distribution inside polymer electrolyte fuel cells (PEFCs). Particularly, energy-resolved neutron imaging is a methodology capable of distinguishing between liquid water and ice, and is effective for investigating ice formation in PEFCs operating in a subfreezing environment. The distinction principle is based on the fact that the cross sections of liquid water and ice differ from each other at low neutron energies. In order to quantitatively observe transient freezing and thawing phenomena in a multiphase mixture (gas/liquid/solid) within real PEFCs with high spatial resolution, a pulsed neutron beam with both high intensity and wide energy range is most appropriate. In the validation study of the present work, we used water sealed in narrow capillary tubes to simulate the flow channels of a PEFC, and a pulsed neutron beam was applied to distinguish ice, liquid water and super-cooled water, and to clarify freezing and thawing phenomena of the water within the capillary tubes. Moreover, we have enabled the observation of liquid water/ice distributions in a large field of view (300 mm  300 mm) by manufacturing a sub-zero environment chamber that can be cooled down to -30
300 mm) by manufacturing a sub-zero environment chamber that can be cooled down to -30 C, as a step towards
C, as a step towards  visualisation of full-size fuel cells.
visualisation of full-size fuel cells.
Okuno, Yasuki*; Ishikawa, Norito; Akiyoshi, Masafumi*; Ando, Hirokazu*; Harumoto, Masaki*; Imaizumi, Mitsuru*
Japanese Journal of Applied Physics, 59(7), p.074001_1 - 074001_7, 2020/07
Times Cited Count:4 Percentile:14.27(Physics, Applied)Performance degradation prediction for space solar cells under irradiation with low-energy electrons is greatly affected by displacement threshold energy (Ed) when a displacement damage dose (DDD) model is used. According to recent studies, the Ed of P atoms is much lower than the conventional Ed value in InP-type solar cells irradiated with low-energy electrons. This indicates that the value of Ed typically used in DDD model leads to significant error in performance degradation prediction. In this study, degradation of AlInGaP solar cells is observed after irradiation with 60 keV electrons. The results suggest that the Ed of P atoms in AlInGaP solar cells is much smaller than the conventionally used Ed value. By using the DDD model with the Ed value obtained in this study, we demonstrated that the performance degradation predicted by the DDD model agrees well with the experimental results.
Shimizu, Takeshi*; Wang, H.*; Matsumura, Daiju; Mitsuhara, Kei*; Ota, Toshiaki*; Yoshikawa, Hirofumi*
ChemSusChem, 13(9), p.2256 - 2263, 2020/05
Times Cited Count:32 Percentile:77.79(Chemistry, Multidisciplinary)Zhang, Z,*; Wang, H.*; Yoshikawa, Hirofumi*; Matsumura, Daiju; Hatao, Shuya*; Ishikawa, Satoshi*; Ueda, Wataru*
ACS Applied Materials & Interfaces, 12(5), p.6056 - 6063, 2020/02
Times Cited Count:7 Percentile:27.06(Nanoscience & Nanotechnology)Kishi, Hirofumi*; Sakamoto, Tomokazu*; Asazawa, Koichiro*; Yamaguchi, Susumu*; Kato, Takeshi*; Zulevi, B.*; Serov, A.*; Artyushkova, K.*; Atanassov, P.*; Matsumura, Daiju; et al.
Nanomaterials (Internet), 8(12), p.965_1 - 965_13, 2018/12
Times Cited Count:13 Percentile:49.03(Chemistry, Multidisciplinary)Cui, Y.-T.*; Harada, Yoshihisa*; Niwa, Hideharu*; Oshima, Masaharu*; Hatanaka, Tatsuya*; Nakamura, Naoki*; Ando, Masaki*; Yoshida, Toshihiko*; Ishii, Kenji*; Matsumura, Daiju
NanotechJapan Bulletin (Internet), 11(4), 6 Pages, 2018/08
no abstracts in English
Wang, H.*; Idobe, Jin*; Matsumura, Daiju; Yoshikawa, Hirofumi*
Journal of Solid State Electrochemistry, 22(7), p.2067 - 2071, 2018/07
Times Cited Count:19 Percentile:42.67(Electrochemistry)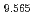 (Si
(Si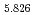 []
[]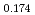 )O
)O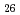 apatite without interstitial oxygens due to the overbonded channel oxygens
apatite without interstitial oxygens due to the overbonded channel oxygensFujii, Kotaro*; Yashima, Masatomo*; Hibino, Keisuke*; Shiraiwa, Masahiro*; Fukuda, Koichiro*; Nakayama, Susumu*; Ishizawa, Nobuo*; Hanashima, Takayasu*; Ohara, Takashi
Journal of Materials Chemistry A, 6(23), p.10835 - 10846, 2018/06
Times Cited Count:30 Percentile:67.85(Chemistry, Physical)Shimizu, Takeshi*; Wang, H.*; Tanifuji, Naoki*; Matsumura, Daiju; Yoshimura, Masashi*; Nakanishi, Koji*; Ota, Toshiaki*; Yoshikawa, Hirofumi*
Chemistry Letters, 47(5), p.678 - 681, 2018/05
Times Cited Count:12 Percentile:37.58(Chemistry, Multidisciplinary)