Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Fe
Fe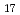 intermetallic compound
intermetallic compoundCao, Y.*; Zhou, H.*; Khmelevskyi, S.*; Lin, K.*; Avdeev, M.*; Wang, C.-W.*; Wang, B.*; Hu, F.*; Kato, Kenichi*; Hattori, Takanori; et al.
Chemistry of Materials, 35(8), p.3249 - 3255, 2023/04
Times Cited Count:2 Percentile:21.90(Chemistry, Physical)Hydrostatic and chemical pressure are efficient stimuli to alter the crystal structure and are commonly used for tuning electronic and magnetic properties in materials science. However, chemical pressure is difficult to quantify and a clear correspondence between these two types of pressure is still lacking. Here, we study intermetallic candidates for a permanent magnet with a negative thermal expansion (NTE). Based on in situ synchrotron X-ray diffraction, negative chemical pressure is revealed in Ho Fe
Fe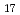 on Al doping and quantitatively evaluated by using temperature and pressure dependence of unit cell volume. A combination of magnetization and neutron diffraction measurements also allowed one to compare the effect of chemical pressure on magnetic ordering with that of hydrostatic pressure. Intriguingly, pressure can be used to control suppression and enhancement of NTE. Electronic structure calculations indicate that pressure affected the top of the majority band with respect to the Fermi level, which has implications for the magnetic stability, which in turn plays a critical role in modulating magnetism and NTE. This work presents a good example of understanding the effect of pressure and utilizing it to control properties of functional materials.
on Al doping and quantitatively evaluated by using temperature and pressure dependence of unit cell volume. A combination of magnetization and neutron diffraction measurements also allowed one to compare the effect of chemical pressure on magnetic ordering with that of hydrostatic pressure. Intriguingly, pressure can be used to control suppression and enhancement of NTE. Electronic structure calculations indicate that pressure affected the top of the majority band with respect to the Fermi level, which has implications for the magnetic stability, which in turn plays a critical role in modulating magnetism and NTE. This work presents a good example of understanding the effect of pressure and utilizing it to control properties of functional materials.

Haga, Yoshinori; Opletal, P.; Tokiwa, Yoshifumi; Yamamoto, Etsuji; Tokunaga, Yo; Kambe, Shinsaku; Sakai, Hironori
Journal of Physics; Condensed Matter, 34(17), p.175601_1 - 175601_7, 2022/04
Times Cited Count:28 Percentile:86.10(Physics, Condensed Matter)Nakajima, Kunihisa; Nakazono, Yoshihisa; Arai, Yasuo
Recent Advances in Actinide Science, p.448 - 450, 2006/06
A pyrochemical process for the metallurgical treatment of the spent nuclear fuels contains the Cd-distillation step, where Pu is recovered as a residue. For understanding this Cd-ditillation behavior the samples of PuCd +PuCd
+PuCd and PuCd
and PuCd +PuCd
+PuCd were prepared and Knudsen-cell mass-spectrometric measurements were carried out to determine the vapour pressures of Cd(g) over these samples. Further,the thermodynamic quantities of PuCd
were prepared and Knudsen-cell mass-spectrometric measurements were carried out to determine the vapour pressures of Cd(g) over these samples. Further,the thermodynamic quantities of PuCd and PuCd
and PuCd were evaluated from these vapour pressures.
were evaluated from these vapour pressures.
Nishi, Tsuyoshi; Takano, Masahide; Ito, Akinori; Akabori, Mitsuo; Minato, Kazuo; Kizaki, Minoru
JAERI-Tech 2005-051, 13 Pages, 2005/09
An apparatus has been developed to measure the thermal diffusivities of minor actinide (MA) compounds. By installing the laser flash apparatus in a glove box with highly purified inert gas atmosphere, the thermal diffusivities measurement of MA compounds of  -decay nuclides was enabled. A new sample holder has been also developed to measure the thermal diffusivities of very small samples. The performance of this new apparatus was confirmed by measuring the thermal diffusivities of small samples of tantalum, nickel and cerium oxides. The thermal diffusivity values obtained in this work agreed well with the literature values and the values measured by a conventional thermal diffusivities measurement apparatus. Accordingly, this apparatus was found to be useful for thermal diffusivities measurement of MA compounds.
-decay nuclides was enabled. A new sample holder has been also developed to measure the thermal diffusivities of very small samples. The performance of this new apparatus was confirmed by measuring the thermal diffusivities of small samples of tantalum, nickel and cerium oxides. The thermal diffusivity values obtained in this work agreed well with the literature values and the values measured by a conventional thermal diffusivities measurement apparatus. Accordingly, this apparatus was found to be useful for thermal diffusivities measurement of MA compounds.
Kurata, Yuji; Futakawa, Masatoshi; Saito, Shigeru
Journal of Nuclear Materials, 335(3), p.501 - 507, 2004/12
Times Cited Count:34 Percentile:87.63(Materials Science, Multidisciplinary)Corrosion tests were performed in oxygen-saturated liquid Pb-Bi at 450 C and 550
C and 550 C in a pot for 3000h for Al-surface-treated steels containing various levels of Cr contents. The Al surface treatments were achieved using a gas diffusion method and a melt dipping method. Al
C in a pot for 3000h for Al-surface-treated steels containing various levels of Cr contents. The Al surface treatments were achieved using a gas diffusion method and a melt dipping method. Al O
O , FeAl
, FeAl and AlCr
and AlCr produced by the gas diffusion method exhibited corrosion resistance to liquid Pb-Bi, while the surface layer produced by the melt dipping method suffered a severe corrosion attack. Fe
produced by the gas diffusion method exhibited corrosion resistance to liquid Pb-Bi, while the surface layer produced by the melt dipping method suffered a severe corrosion attack. Fe Al
Al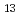 and Fe
and Fe Al
Al produced by the melt dipping method disappeared during the corrosion test at 550
produced by the melt dipping method disappeared during the corrosion test at 550 C and only FeAl remained.
C and only FeAl remained.
Kojima, Takuji
Shinku, 47(11), p.789 - 795, 2004/11
When flue gas/off gas is irradiated by EB, many free radicals such as OH and active oxygen atom are formed from major components of air: namely nitrogen, oxygen, water and carbon dioxide ecules. The similer reaction can be achieved using UV light and plasma-discharging, but ionizing radiations produce such free radicals at higher density. Such radiation-induced radicals react efficiently with air pollutants, SOx and NOx in coal/oil combustion flue gas at thermal power plants, dioxins in waste incineration flue gas and volatile organic compounds (VOC) even in very low concentration and decompose them into non-toxic substances or change them to removable substances. R & D on EB treatment of flue gas/off gas done in JAERI on the basis of this principle process, as an example, is described in the present paper.
Saeki, Masakatsu
JAERI-Review 2003-030, 50 Pages, 2003/11
In this review, synthetic methods of uranium and neptunium compounds mainly from solutions and their natures are summarized. For uranium, 3 compounds of trivalent, 4 compounds of tetravalent and 23 compounds of hexavalent are described in slight details. For neptunium, synthetic methods of stock solutions for pentavalent and hexavalent neptunium, 4 items (6 compounds) of trivalent, 8 items (19 compounds) of tetravalent, 28 items (29 compounds) of pentavalent, 10 items (14 compounds) of hexavalent and 5 items (9 compounds) of heptavalent are described in slight details. Furthermore, on compounds which can not be described in details, the names are listed with literatures. The materials used here have been collected during the research activities on Moessbauer spectroscopic studies of uranium and neptunium compounds, then, the reviews on Moessbauer spectroscopic studies of actinoid compounds are also listed.
 films by PLD for photocatalyst applications
films by PLD for photocatalyst applicationsYamaki, Tetsuya; Sumita, Taishi; Yamamoto, Shunya; Miyashita, Atsumi
Journal of Crystal Growth, 237-239(Part1), p.574 - 579, 2002/04
Pulsed laser deposition (PLD) with a KrF excimer laser was used to prepare epitaxial TiO films on a (0001) sapphire substrate. The crystalline structure and surface morphology of the films were then investigated as a function of the laser power density in the PLD. X-ray diffraction and Raman spectroscopy results showed that the films were composed of the (100) oriented rutile phase with a small amount of the (001) oriented anatase. The anatase-to-rutile ratio in such phase-mixed films was controlled by the laser power; the ratio increased as the power density decreased. From atomic force microscopy observations, the film deposited at the lowest power was found to have a rough surface consisting of coarse grains. This film exhibited the best photocatalytic performance during the decomposition of the organic dyes possibly due to the maximum relative content of anatase and the large surface area.
films on a (0001) sapphire substrate. The crystalline structure and surface morphology of the films were then investigated as a function of the laser power density in the PLD. X-ray diffraction and Raman spectroscopy results showed that the films were composed of the (100) oriented rutile phase with a small amount of the (001) oriented anatase. The anatase-to-rutile ratio in such phase-mixed films was controlled by the laser power; the ratio increased as the power density decreased. From atomic force microscopy observations, the film deposited at the lowest power was found to have a rough surface consisting of coarse grains. This film exhibited the best photocatalytic performance during the decomposition of the organic dyes possibly due to the maximum relative content of anatase and the large surface area.
Mori, Yuki*; Kato, Sayuri*; Mori, Hiroko*; Katayama, Yoshinori; Tsuji, Kazuhiko*
Review of High Pressure Science and Technology, 7, p.353 - 355, 1998/03
The temperature dependence of phase transitions in GaSb, AlSb, GaAs, GaP, InAs, ZnSe, and CdTe are studied by X-ray diffraction measurements under pressure upto 30 GPa at temperatures of 90-300K. The phase transitions depend on paths in a pressure-temperature phase diagram. The structure of the recovered phase after decompression depends on the ionicity in bonding: amorphous for small ionicity, the stable zincblende structure for large ionicity, and microcrystalline or moderate ionicity. These results are discussed by using a configuration-coordinate model.
; ; Kurasawa, T.; Katsuta, Hiroji;
Journal of Nuclear Materials, 133-134, p.181 - 185, 1985/00
Times Cited Count:61 Percentile:97.99(Materials Science, Multidisciplinary)no abstracts in English
 and PuCoC
and PuCoC
; ; ;
Journal of Nuclear Science and Technology, 19(3), p.257 - 260, 1982/00
Times Cited Count:3 Percentile:54.28(Nuclear Science & Technology)no abstracts in English

 F produced by the
F produced by the 
 O(t,n)
O(t,n)
 F reaction in neutron-irradiated lithium compounds containing oxygen
F reaction in neutron-irradiated lithium compounds containing oxygen; Tanaka, Kichizo
Journal of Chemical Physics, 72(5), p.3049 - 3052, 1980/00
Times Cited Count:5 Percentile:26.20(Chemistry, Physical)no abstracts in English
; ;
Bulletin of the Chemical Society of Japan, 36(8), p.928 - 937, 1963/00
Times Cited Count:7no abstracts in English
Nihon Genshiryoku Gakkai-Shi, 4(1), p.8 - 16, 1962/00
no abstracts in English
Nihon Genshiryoku Gakkai-Shi, 4(3), p.166 - 174, 1962/00
no abstracts in English
 Ge
Ge ; de Haas-van Alphen study
; de Haas-van Alphen studyHaga, Yoshinori; Yamamoto, Etsuji; Nakamura, Ai*; Honda, Fuminori*; Aoki, Dai*; Matsumoto, Yuji*; Yamamura, Tomoo*; Shirasaki, Kenji*; Yamagami, Hiroshi*
no journal, ,
 Ge
Ge
Haga, Yoshinori; Yamamoto, Etsuji; Nakamura, Ai*; Honda, Fuminori*; Aoki, Dai*; Matsumoto, Yuji*; Yamamura, Tomoo*; Shirasaki, Kenji*; Yamagami, Hiroshi*; Sumida, Kazuki
no journal, ,
no abstracts in English

Haga, Yoshinori; Opletal, P.; Tokiwa, Yoshifumi; Yamamoto, Etsuji; Tokunaga, Yo; Kambe, Shinsaku; Sakai, Hironori
no journal, ,