Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Sasaki, Yuji; Kaneko, Masashi; Matsumiya, Masahiko*; Kumagai, Yuta
Journal of Molecular Liquids, 441, p.129013_1 - 129013_10, 2026/01
This study determined extraction and back-extraction conditions for the mutual separation of three light PGMs, Ru, Rh, and Pd. Results revealed that reagents containing soft N and S donor atoms efficiently extract and strip Pd through solvation. In comparison, Ru and Rh undergo ion-pair extraction, requiring both anionic metal species and cationic extractants. These essential chlorinated PGM anions and protonated extractants having amino N atoms are present in HCl media. D(Ru) and D(Rh) values of approximately 100 and 10, respectively, were obtained using nitrilo-triacet-amide (NTAamide), which exhibits tetradentate coordination. Refluxing in 3-6 M HCl at 250  C, a condition that promotes the formation of highly chlorinated PGM anionic species, increased D(Ru) and D(Rh). Based on these findings, a flow diagram for the mutual separation of three PGMs was developed.
C, a condition that promotes the formation of highly chlorinated PGM anionic species, increased D(Ru) and D(Rh). Based on these findings, a flow diagram for the mutual separation of three PGMs was developed.
Toigawa, Tomohiro; Tsubata, Yasuhiro; Kumagai, Yuta; Ban, Yasutoshi
Progress in Nuclear Science and Technology (Internet), 8, p.286 - 290, 2025/09
We propose a simple process simulation methodology that uses readily available information about radiation impact. A process simulation was conducted for a minor actinides (MA) separation process while considering the degradation of extraction ability by radiolysis. The simulation provided a processing limit of MA and enabled the evaluation of radiation stability.
Sasaki, Yuji; Kaneko, Masashi; Kumagai, Yuta; Ban, Yasutoshi
Progress in Nuclear Science and Technology (Internet), 8, p.202 - 204, 2025/09
Two extractants and a masking agent of TODGA (TetraOctyl-DiGlycolAmide), ADAAM (AlkylDiAmideAMine), and DTBA (DiethyleneTriamine-triacetic-BisAmide) were developed in JAEA. TODGA can extract both trivalent actinides (An) and lanthanides (Ln), DTBA may separate An from Ln, and ADAAM has high separation factor (SF: 6) for Am/Cm. The suitable conditions for the extraction, separation and isolations of An from Ln are investigated using these reagents. In this work, we show the basic information on extraction behavior of An and Ln using TODGA, DTBA and ADAAM and propose the suitable aqueous and the organic conditions for An+Ln extraction, An/Ln separation and Am/Cm separation.
Emori, Tatsuya; Kitatsuji, Yoshihiro; Ban, Yasutoshi
JAEA-Technology 2024-025, 20 Pages, 2025/03
Radioisotope Thermoelectric Generators (RTGs) using the decay heat of Pu-238 has been applied for outer planet missions far from Jupiter, where solar power is limited. However, no facilities are available to produce Pu-238 for space probes in Japan. Moreover, the use of nuclear materials for the space exploration is difficult in term of the regulation. Thus, we focused on Am-241 whose half-life is around 432 years as an alternative heat source for RTGs. This report describes the procedure of separating Am-241 decayed from Pu-241 in aged plutonium oxide. Two experiments were performed: one using solid-liquid extraction and the other combining liquid-liquid extraction and solid-liquid extraction. Packed columns were used in the experiments, with their number reduced by less than one-fifth in the latter experiment compared to the former. Furthermore, the time required for separation in the latter experiment was less than half that of the former. We performed the separation experiments six times, collecting a total of approximately 0.43 g of Am-241 as an oxalate salt.
Miyazaki, Yasunori; Sano, Yuichi; Ishigami, Ryoya*
EPJ Web of Conferences, 317, p.01006_1 - 01006_7, 2025/01
Times Cited Count:0 Percentile:0.00(Chemistry, Inorganic & Nuclear)The gamma-ray and He ion beam (which is simulated for alpha-ray from
ion beam (which is simulated for alpha-ray from  Am, for example) were irradiated on the TEHDGA adsorbent to evaluate the hydrogen gas production, leaching amount of organics in the immersed 3 M HNO
Am, for example) were irradiated on the TEHDGA adsorbent to evaluate the hydrogen gas production, leaching amount of organics in the immersed 3 M HNO solution, thermal characteristics and speciation of the degradation products. These were combined to assess the safety of the 1st run of the MA separation process from the raffinate at a dose rate of 1 kGy/h.
solution, thermal characteristics and speciation of the degradation products. These were combined to assess the safety of the 1st run of the MA separation process from the raffinate at a dose rate of 1 kGy/h.
Sasaki, Yuji; Kaneko, Masashi; Ban, Yasutoshi; Suzuki, Tatsuya*
Solvent Extraction Research and Development, Japan, 32(1), p.21 - 29, 2025/00
Times Cited Count:2 Percentile:69.11(Chemistry, Multidisciplinary)The magnitude of the masking effect of carboxylic, amic-acidic, and amidic compounds through Ln and Am extractions by tetraoctyl diglycolamide (TODGA) is compared. The compounds used are diglycol, ethylenediamine, diethylenetriamine-type, and two other amides (dioxaoctane diamide and nitrylotriacetamide). The results show that below pH 1.2, where carboxylic acids are less dissociated, amide O atoms have higher reactivity with lanthanides than O atoms in carboxyl groups. From observing the Ln patterns (D(Ln) vs. their atomic number), the compounds primarily show high reactivity, with middle and heavy Ln having a higher charge density than light Ln. Four amide compounds are employed in this work. Those with tertiary amine N atoms have pH dependence on D(Ln) due to protonation and dissociation from amine N atoms.
 -Ga
-Ga O
O (001)
(001)Tsai, Y. H.*; Kobata, Masaaki; Fukuda, Tatsuo; Tanida, Hajime; Kobayashi, Toru; Yamashita, Yoshiyuki*
Applied Physics Letters, 124(11), p.112105_1 - 112105_5, 2024/03
Times Cited Count:4 Percentile:53.37(Physics, Applied)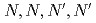 -tetraoctyldiglycolamide-impregnated polymer-coated silica particle using fluorescence microspectroscopy; Transfer mechanism and effect of polymer crosslinking degree
-tetraoctyldiglycolamide-impregnated polymer-coated silica particle using fluorescence microspectroscopy; Transfer mechanism and effect of polymer crosslinking degreeMiyagawa, Akihisa*; Takahashi, Takumi*; Kuzure, Yoshiaki*; Iwamoto, Hibiki*; Arai, Tsuyoshi*; Nagatomo, Shigenori*; Watanabe, So; Sano, Yuichi; Nakatani, Kiyoharu*
Analytical Sciences, 39(11), p.1929 - 1936, 2023/11
Times Cited Count:1 Percentile:6.06(Chemistry, Analytical)In this study, we revealed the Eu(III) distribution in a single diglycolamide-derivative extractant (TODGA)-impregnated polymer-coated silica particle. The reaction of Eu(III) with two TODGA molecules in the polymer layer was the rate-limiting process, as evidenced by the absence of any correlation between the rate constants (k and k
and k ) and concentrations of Eu(III) and HNO
) and concentrations of Eu(III) and HNO .
.
Morii, Shiori; Yomogida, Takumi; Asai, Shiho*; Ouchi, Kazuki; Oka, Toshitaka; Kitatsuji, Yoshihiro
Bunseki Kagaku, 72(10.11), p.441 - 448, 2023/10
Rapid analytical method for the determination of Zr-93 in radioactive wastes has been developed. Laser ablation (LA)-ICP-MS was applied to the analysis of Zr isotopes in simulated high-level radioactive waste (HLW). Sample preparation time was dramatically reduced by using a DGA resin as the adsorbent for Zr. Direct quantification of Zr isotopes in this resin sample was carried out by LA-ICP-MS. Laser settings were optimized to obtain a reliable isotope ratio of the sample by LA-ICP-MS. Quantification of Zr isotopes in the simulated HLW solution by isotope dilution mass spectrometry (IDMS) was examined. The amount of Zr-90 in the sample obtained by IDMS corresponded to a value calculated from the given concentration of Zr in the sample within uncertainty. Thus, this method can be applied for the quantification of Zr-93 in radioactive wastes.
Sasaki, Yuji
Bunri Gijutsu, 52(2), p.103 - 107, 2022/03
We develop all-inclusive partitioning method for actinides and fission products in high-level radioactive waste. This process is based on the sequential solvent extraction. In order to recover Cs and Sr for the management by interim storage, crown ether compounds are employed. For the removal of Pd and Mo due to production of a stable vitrified object, methylimino-dioctylacetamide (MIDOA) is taken as an extractant. DGA can extract both actinides and trivalent lanthanides. In order to separate each other, dietylenetriamine-triacetic-diamide (DTBA) for the stripping reagent of MA. For the mutual separation of Am/Cm, DGA and DOODA extraction system is taken into consideration.
Nomizu, Daiki; Sasaki, Yuji; Kaneko, Masashi; Matsumiya, Masahiko*; Katsuta, Shoichi*
Journal of Radioanalytical and Nuclear Chemistry, 331(3), p.1483 - 1493, 2022/03
Times Cited Count:7 Percentile:64.72(Chemistry, Analytical)We studied the successive formation of water soluble DGA (diglycolamide) and DOODA (dioxaoctanediamide) for the mutual separation of Ln in this extraction system. TODGA (tetraoctyl-diglycolamide) and DOODA(C8) (tetraoctyl-dioxaoctanediamide) have the opposite trend to extract light and heavy Ln through Ln-patterns. Metal-complexes of two folding Ln ions with water-soluble DOODA and three folding with DGA are found and their observed formation constants are calculated. The suitable separation condition (aqueous phase: 30 mM DOODA(C2) in 1 M nitric acid, organic phase: 0.1 M TODGA in n-dodecane) of multi-stage extraction (10  10) is conducted. From the present work, it is clear that La, Pr and Nd are mainly present in aqueous phase, instead Sm-Dy exist in the organic phase.
10) is conducted. From the present work, it is clear that La, Pr and Nd are mainly present in aqueous phase, instead Sm-Dy exist in the organic phase.
Sasaki, Yuji; Kaneko, Masashi; Ban, Yasutoshi; Matsumiya, Masahiko*; Nakase, Masahiko*; Takeshita, Kenji*
Separation Science and Technology, 57(16), p.2543 - 2553, 2022/00
Times Cited Count:5 Percentile:27.73(Chemistry, Multidisciplinary)The mutual separation of actinides (An) from lanthanides (Ln) using the masking agent of DTPA (diethylenetriamine-pentaacetic acid) or DTBA (diethylenetriamine-triacetic acid-bis(diethylacetamide)) in the aqueous phase through DGA extraction, referring TALSPEAK method, is focused. We investigate to obtain the same separation performance using commercially available DTPA on that using DTBA. In this work, we select lactic acid (LA) of pH buffer from 10 organic acids and ethylenediamine (ED) for the pH adjustment. Almost the same D and SF values are obtained among the conditions: TODGA-DTPA-LA-NaOH, TODGA-DTPA-LA-ED, and TODGA-DTBA-LA. The experimental results using batchwise multi-stage extractions show the average yields of Ln (La to Gd) and Am to be 3.73 and 98.1% in the aqueous phase using DGA-DTPA-LA-ED, to be 3.1 and 97.0% using DGA-DTPA-LA-NaOH, and to be 1.61 and 98.7% using DGA-DTBA-LA.
Sasaki, Yuji; Kaneko, Masashi; Matsumiya, Masahiko*; Nakase, Masahiko*; Takeshita, Kenji*
Solvent Extraction and Ion Exchange, 40(6), p.620 - 640, 2022/00
Times Cited Count:3 Percentile:15.58(Chemistry, Multidisciplinary)Owing to the chemical behavior of trivalent lanthanide and actinide ions with similar ionic radii, realizing this separation is still challenging. All lanthanides, Am, and Cm can be extracted using diglycolamide (DGA), and relatively high An/Ln separation efficiencies have been obtained using diethylenetriamine-triacetic-bisamide (DTBA). To improve the previous results as well as the separation conditions, we used organic acids for pH adjustment. The advantages of this modification included low HNO , DTBA concentrations and pH stability owing to the addition of lactic acid. Under these modified conditions, the recovery rates observed were as follows: 97.1% for Nd with the co-existence of 1.59% Am in organic phase, and 98.4% for Am with the co-existence of 2.95% Nd in aqueous phase.
, DTBA concentrations and pH stability owing to the addition of lactic acid. Under these modified conditions, the recovery rates observed were as follows: 97.1% for Nd with the co-existence of 1.59% Am in organic phase, and 98.4% for Am with the co-existence of 2.95% Nd in aqueous phase.
Matsutani, Takafumi; Sasaki, Yuji; Katsuta, Shoichi*
Analytical Sciences, 37(11), p.1603 - 1609, 2021/11
Times Cited Count:9 Percentile:41.17(Chemistry, Analytical)We investigated the chemical behavior of lanthanides (Ln) using diglycolamide extractant with multistage extraction. We obtained the breakthrough curves for light and middle Ln. Our study reveals that the metal extraction limit depends on their  values and metal concentrations used in the experiments. From the multistage extractions of 15 aqueous phases and 15 organic phases, three curves (extraction curves, back-extraction curves, and separation curves) were obtained by changing the nitric acid concentration. As an example, under a condition of the separation curve experiment (aqueous phase: 0.5 M HNO
values and metal concentrations used in the experiments. From the multistage extractions of 15 aqueous phases and 15 organic phases, three curves (extraction curves, back-extraction curves, and separation curves) were obtained by changing the nitric acid concentration. As an example, under a condition of the separation curve experiment (aqueous phase: 0.5 M HNO , organic phase: 0.1 M TDDGA (
, organic phase: 0.1 M TDDGA (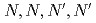 -tetradecyl-diglycolamide) in
-tetradecyl-diglycolamide) in  -dodecane), a recovery of more than 99% of Sm in the organic phase with less than 1% Nd can be obtained.
-dodecane), a recovery of more than 99% of Sm in the organic phase with less than 1% Nd can be obtained.
 -P for an advanced reprocessing
-P for an advanced reprocessingHoriuchi, Yusuke; Watanabe, So; Sano, Yuichi; Takeuchi, Masayuki; Kida, Fukuka*; Arai, Tsuyoshi*
Journal of Radioanalytical and Nuclear Chemistry, 330(1), p.237 - 244, 2021/10
Times Cited Count:11 Percentile:72.10(Chemistry, Analytical)Applicability of tetra2-ehylhexyl diglycolamide (TEHDGA) impregnated adsorbent for minor actinide (MA) recovery from high level liquid waste (HLLW) in extraction chromatography technology was investigated through batch-wise adsorption and column separation experiments. Distribution ratio of representative fission product elements were obtained by the batch-wise experiments, and TEHDGA adsorbent was shown to be preferable to TODGA adsorbent for decontamination of several species. All Ln(III) supplied into the TEHDGA adsorbent packed column was properly eluted from the column, and the applicability of the adsorbent was successfully showed by this study.
Sakamoto, Atsushi; Kibe, Satoshi*; Kawanobe, Kazunori*; Fujisaku, Kazuhiko*; Sano, Yuichi; Takeuchi, Masayuki; Suzuki, Hideya*; Tsubata, Yasuhiro; Ban, Yasutoshi; Matsumura, Tatsuro
JAEA-Research 2021-003, 30 Pages, 2021/06
Japan Atomic Energy Agency has been developing a solvent extraction process called SELECT to recover minor actinides (MA) from spent nuclear fuel. In the SELECT process, TDdDGA, HONTA, and ADAAM are used as the extractants for MA + Ln corecovery, MA/Ln separation and Am/Cm separation, respectively. These extractants do not contain phosphorus (P), and consist of carbon (C), hydrogen (H), oxygen (O), and nitrogen (N). In this study, in order to give beneficial information for designing flowsheet, the mass transfer coefficients of Ln between HNO solution and TDdDGA or HONTA / n-dodecane solvent were evaluated by the single drop technique. Prior to the evaluation of mass transfer coefficient, we had optimized the structure of the single drop apparatus to improve accuracy of the measurement. Based on the mass transfer coefficients obtained in HNO
solution and TDdDGA or HONTA / n-dodecane solvent were evaluated by the single drop technique. Prior to the evaluation of mass transfer coefficient, we had optimized the structure of the single drop apparatus to improve accuracy of the measurement. Based on the mass transfer coefficients obtained in HNO / TDdDGA-n-dodecane system, Ln behaviors in the counter-current extraction and back-extraction using mixer-settlers and centrifugal contactors were estimated by simple calculation, and they had a good agreement with our previous experimental results. We also confirmed the mass transfer coefficients of Ln in HNO
/ TDdDGA-n-dodecane system, Ln behaviors in the counter-current extraction and back-extraction using mixer-settlers and centrifugal contactors were estimated by simple calculation, and they had a good agreement with our previous experimental results. We also confirmed the mass transfer coefficients of Ln in HNO / HONTA - n-dodecane system are under 10
/ HONTA - n-dodecane system are under 10 m/s.
m/s.
Sasaki, Yuji; Morita, Keisuke; Matsumiya, Masahiko*; Ono, Ryoma*; Shiroishi, Hidenobu*
JOM, 73(4), p.1037 - 1043, 2021/04
Times Cited Count:4 Percentile:23.17(Materials Science, Multidisciplinary)The separation of Dy from Nd is studied from the viewpoint of recycling Dy from Nd magnets. Both metals are lanthanide elements, which means their mutual separation is difficult because of their similar chemical behaviors. All lanthanide elements can be extracted easily by using tetradodecyl-diglycolamide (TDdDGA) extractants, and it has a relatively high separation factor (SF) between Dy and Nd (SF over 10). In the present study, by performing eight extraction steps with the organic phase (0.1M TDdDGA in dodecane), ten steps with an aqueous phase (0.7 M HNO with metals), and six steps with another aqueous phase (0.7 M HNO
with metals), and six steps with another aqueous phase (0.7 M HNO without metals), approximately 99% Dy was recovered into the organic phase with 1% co-extraction of Nd.
without metals), approximately 99% Dy was recovered into the organic phase with 1% co-extraction of Nd.
Matsumiya, Masahiko*; Tsuchida, Yusuke*; Sasaki, Yuji; Ono, Ryoma*; Nakase, Masahiko*; Takeshita, Kenji*
Journal of Radioanalytical and Nuclear Chemistry, 327(1), p.597 - 607, 2021/01
Times Cited Count:5 Percentile:38.47(Chemistry, Analytical)To achieve trichotomic separation of light lanthanides (Ln), heavy Ln, and Am, batchwise multi-stage extractions using tetraoctyl-diglycolamide (TODGA) extractant from organic acids are studied. Malonic acid (MA) has high solubility in water and is used as the main component of the aqueous phase. It is clear that the separation factor (SF) for Nd/Am from MA and that for La/Am from MA + HNO are both around 30. The light Ln (e.g., La and Ce) flowed-out in 1 M MA+0.05 M HNO
are both around 30. The light Ln (e.g., La and Ce) flowed-out in 1 M MA+0.05 M HNO (1st soln.), Am is recovered into 3 M MA (2nd soln.), and middle and heavy Ln (Nd and other heavy Ln) are back-extracted into 0.1 M TEDGA/water (3rd soln.). This extraction method can give 95% recovery of Am with total Ln of less than 16% present in high-level radioactive waste.
(1st soln.), Am is recovered into 3 M MA (2nd soln.), and middle and heavy Ln (Nd and other heavy Ln) are back-extracted into 0.1 M TEDGA/water (3rd soln.). This extraction method can give 95% recovery of Am with total Ln of less than 16% present in high-level radioactive waste.
Toigawa, Tomohiro; Tsubata, Yasuhiro; Kai, Takeshi; Furuta, Takuya; Kumagai, Yuta; Matsumura, Tatsuro
Solvent Extraction and Ion Exchange, 39(1), p.74 - 89, 2021/00
Times Cited Count:2 Percentile:6.48(Chemistry, Multidisciplinary)Absorbed-dose estimation is essential for evaluation of the radiation feasibility of minor-actinide-separation processes. We propose a dose-evaluation method based on radiation permeability, with comparisons of heterogeneous structures seen in the solvent-extraction process, such as emulsions forming in the mixture of the organic and aqueous phases. A demonstration of radiation-energy-transfer simulation is performed with a focus on the minor-actinide-recovery process from high-level liquid waste with the aid of the Monte Carlo radiation-transport code PHITS. The simulation results indicate that the dose absorbed by the extraction solvent from alpha ray depends upon the emulsion structure, and that from beta and gamma ray depends upon the mixer-settler-apparatus size. Non-negligible contributions of well-permeable gamma rays were indicated in terms of the plant operation of the minor-actinide-separation process.
Toigawa, Tomohiro; Murayama, Rin*; Kumagai, Yuta; Yamashita, Shinichi*; Suzuki, Hideya; Ban, Yasutoshi; Matsumura, Tatsuro
UTNL-R-0501, p.24 - 25, 2020/12
This report summarizes the results obtained in FY2019 at Electron Linac Facility of University of Tokyo. The radiolysis process of a diglycolamide extractant, which is expected to be used in the separation process of minor actinides (MA), in dodecane and octanol solutions was investigated by pulse radiolysis. As a result, it was suggested that by adding alcohol, the decomposition process of the diglycolamide extractant was different from the decomposition processes in the single solvent of dodecane considered that the decomposition occurred via a radical cation species of the extractant.