Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 BaCo(PO
BaCo(PO )
)
Sheng, J.*; Wang, L.*; Candini, A.*; Jiang, W.*; Huang, L.*; Xi, B.*; Zhao, J.*; Ge, H.*; Zhao, N.*; Fu, Y.*; et al.
Proceedings of the National Academy of Sciences of the United States of America, 119(51), p.e2211193119_1 - e2211193119_9, 2022/12
Times Cited Count:3 Percentile:28(Multidisciplinary Sciences)Zhang, P.*; Tang, X.*; Wang, Y.*; Wang, X.*; Gao, D.*; Li, Y.*; Zheng, H.*; Wang, Y.*; Wang, X.*; Fu, R.*; et al.
Journal of the American Chemical Society, 142(41), p.17662 - 17669, 2020/10
Times Cited Count:21 Percentile:74.59(Chemistry, Multidisciplinary)Solid-state topochemical polymerization (SSTP) is a promising method to construct functional crystalline polymeric materials, but in contrast to various reactions that happen in solution, only very limited types of SSTP reactions are reported. Diels-Alder (DA) and dehydro-DA (DDA) reactions are textbook reactions for preparing six-membered rings in solution but are scarcely seen in solid-state synthesis. Here, using multiple cutting-edge techniques, we demonstrate that the solid 1,4-diphenylbutadiyne (DPB) undergoes a DDA reaction under 10-20 GPa with the phenyl as the dienophile. The crystal structure at the critical pressure shows that this reaction is "distance-selected". The distance of 3.2 between the phenyl and the phenylethynyl facilitates the DDA reaction, while the distances for other DDA and 1,4-addition reactions are too large to allow the bonding. The obtained products are crystalline armchair graphitic nanoribbons, and hence our studies open a new route to construct the crystalline carbon materials with atomic-scale control.
between the phenyl and the phenylethynyl facilitates the DDA reaction, while the distances for other DDA and 1,4-addition reactions are too large to allow the bonding. The obtained products are crystalline armchair graphitic nanoribbons, and hence our studies open a new route to construct the crystalline carbon materials with atomic-scale control.
Nagai, Yuki; Shen, H.*; Qi, Y.*; Liu, J.*; Fu, L.*
Physical Review B, 96(16), p.161102_1 - 161102_6, 2017/10
Times Cited Count:54 Percentile:89.54(Materials Science, Multidisciplinary)no abstracts in English
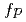 shell; Lifetime measurements in
shell; Lifetime measurements in 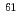 Zn
ZnQueiser, M.*; Vogt, A.*; Seidlitz, M.*; Reiter, P.*; Togashi, Tomoaki*; Shimizu, Noritaka*; Utsuno, Yutaka; Otsuka, Takaharu*; Homma, Michio*; Petkov, P.*; et al.
Physical Review C, 96(4), p.044313_1 - 044313_13, 2017/10
Times Cited Count:5 Percentile:41.41(Physics, Nuclear)no abstracts in English
Hata, Kuniki; Urushibara, Ayumi*; Yamashita, Shinichi*; Lin, M.*; Muroya, Yusa*; Shikazono, Naoya; Yokoya, Akinari; Fu, H.*; Katsumura, Yosuke*
Journal of Radiation Research, 56(1), p.59 - 66, 2015/01
Times Cited Count:6 Percentile:29.71(Biology)Wan, L. K.*; Peng, J.*; Lin, M.; Muroya, Yusa*; Katsumura, Yosuke*; Fu, H. Y.*
Radiation Physics and Chemistry, 81(5), p.524 - 530, 2012/05
Times Cited Count:16 Percentile:75.52(Chemistry, Physical)Rate constants of common crown ethers ((C H
H O)
O) , n=4, 5, 6) and their model compound, 1,4-dioxane(6C2) with some important oxidative radicals,
, n=4, 5, 6) and their model compound, 1,4-dioxane(6C2) with some important oxidative radicals,  OH, SO
OH, SO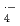 , and NO
, and NO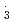 were determined in various aqueous solutions using pulse radiolysis and laser photolysis techniques. The rate constants for 6C2 and crown ethers with
were determined in various aqueous solutions using pulse radiolysis and laser photolysis techniques. The rate constants for 6C2 and crown ethers with  OH and SO
OH and SO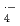 increase with the number of H-atom in the ethers, indicating that H-abstraction is dominant reaction between crown ethers and these two radicals. The presence of cations in solution has negligible effect on the rate constants for
increase with the number of H-atom in the ethers, indicating that H-abstraction is dominant reaction between crown ethers and these two radicals. The presence of cations in solution has negligible effect on the rate constants for  OH and SO
OH and SO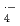 . However, for NO
. However, for NO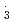 , the rate constants is not in proportional to H-atom number in crown ethers, and the 12-crown-4 is most reactive compared with the others. For the studied crown ethers, the rate constants of these oxidative radicals have the order:
, the rate constants is not in proportional to H-atom number in crown ethers, and the 12-crown-4 is most reactive compared with the others. For the studied crown ethers, the rate constants of these oxidative radicals have the order:  OH
OH  SO
SO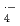
 NO
NO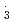 . This is the first report on the kinetic behavior of crown ethers with NO
. This is the first report on the kinetic behavior of crown ethers with NO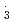 , and it would be helpful for the understanding of stability of crown ethers in the nuclear fuel reprocessing.
, and it would be helpful for the understanding of stability of crown ethers in the nuclear fuel reprocessing.
Fu, H. Y.*; Lin, M.; Muroya, Yusa*; Katsumura, Yosuke*
Research on Chemical Intermediates, 38(1), p.135 - 145, 2012/01
Times Cited Count:2 Percentile:2.92(Chemistry, Multidisciplinary)In the present work, pulse radiolysis was used to demonstrate the oxidizing nature of various intermediates of  -AcMetNH
-AcMetNH and
and  -AcMetOMe via the oxidation of tryptophan (Trp) in a free aqueous solution. The reaction was examined at different pH levels. Electron transfer processes involving Trp and the sulfur-containing three-electron-bonded intermediates were investigated. The results revealed that Trp-Met pairs can form very favorable redox couples in biological systems, which is consistent with earlier reports of more efficient and rapid intramolecular charge transfers in synthetic as well as natural peptides containing methionine (Met).
-AcMetOMe via the oxidation of tryptophan (Trp) in a free aqueous solution. The reaction was examined at different pH levels. Electron transfer processes involving Trp and the sulfur-containing three-electron-bonded intermediates were investigated. The results revealed that Trp-Met pairs can form very favorable redox couples in biological systems, which is consistent with earlier reports of more efficient and rapid intramolecular charge transfers in synthetic as well as natural peptides containing methionine (Met).
Fu, H. Y.*; Lin, M.; Katsumura, Yosuke*; Muroya, Yusa*
International Journal of Chemical Kinetics, 43(10), p.590 - 597, 2011/10
Times Cited Count:2 Percentile:3.88(Chemistry, Physical)Free radical scavenging and antioxidant activities of four natural flavonoids, namely silybin, naringenin, naringin and hesperetin, have been studied using nanosecond pulse radiolysis techniques. The kinetics and mechanisms of the reactions of silybin and analogues with various oxidizing radicals have been investigated. Further, the transient species has been assigned and radical scavenging rate constants have also been measured. Moreover, the structure-activity relationships (SAR) between chemical structures of the flavonoids and their radical scavenging activities are further analyzed by theoretical calculation. Combined our previous observation of the fast reparation of DNA damage and efficient DNA protection against radiation damage in vitro, it can be confirmed that test flavonoids are promising molecules to be used for their potential antioxidant properties.
Shi, W.-Q.*; Fu, H.-Y.*; Bounds, P. L.*; Muroya, Yusa*; Lin, M.; Katsumura, Yosuke*; Zhao, Y.-L.*; Chai, Z.-F.*
Radiation Research, 176(1), p.128 - 133, 2011/07
Times Cited Count:3 Percentile:17.22(Biology)3-Nitrotyrosine (3-NT) has been reported as an important biomarker of oxidative stress and potential source of reactive oxygen species (ROSs). In this work, the UV-visible absorption spectra of the transients formed by hydrated electron (e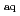
 ) reacting with 3-NT and its derivatives were investigated, the spectra showed many characteristics of aromatic nitro anion radical. The reaction rate constants of e
) reacting with 3-NT and its derivatives were investigated, the spectra showed many characteristics of aromatic nitro anion radical. The reaction rate constants of e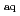
 reacting with 3-NT, N-acetyl-3-nitrotyrosine ethyl ester (NANTE) and nitrotyrosine-containing peptide Gly-nitroTyr-Gly at neutral pH were determined, respectively, which were almost two orders of magnitude higher than that of tyrosine and tyrosine-containing peptides. The pH-dependence of e
reacting with 3-NT, N-acetyl-3-nitrotyrosine ethyl ester (NANTE) and nitrotyrosine-containing peptide Gly-nitroTyr-Gly at neutral pH were determined, respectively, which were almost two orders of magnitude higher than that of tyrosine and tyrosine-containing peptides. The pH-dependence of e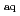
 decay rate constants in presence of 3-NT was also studied.
decay rate constants in presence of 3-NT was also studied.
 , Br
, Br
 , and Br
, and Br
 in aqueous solutions
in aqueous solutionsLin, M.; Archirel, P.*; Van-Oanh, N. T.*; Muroya, Yusa*; Fu, H.*; Yan, Y.*; Nagaishi, Ryuji; Kumagai, Yuta; Katsumura, Yosuke*; Mostafavi, M.*
Journal of Physical Chemistry A, 115(17), p.4241 - 4247, 2011/04
Times Cited Count:14 Percentile:44.71(Chemistry, Physical)The absorption spectra of Br
 and Br
and Br
 in aqueous solutions are investigated by pulse radiolysis techniques from room temperature to 380 and 350
in aqueous solutions are investigated by pulse radiolysis techniques from room temperature to 380 and 350 C, respectively. The weak temperature effect on the absorption spectra of Br
C, respectively. The weak temperature effect on the absorption spectra of Br
 and Br
and Br
 is because in these two systems, the transition occurs between two valence states. We performed classical dynamics of hydrated Br
is because in these two systems, the transition occurs between two valence states. We performed classical dynamics of hydrated Br system at 20 and 300
system at 20 and 300 C under pressure of 25 MPa. This shows that the first water shell is strongly bound to the anion whatever the temperature. The first two water shells form a cavity of a roughly spherical shape around the anion. By TDDFT method, we calculated the absorption spectra of hydrated Br
C under pressure of 25 MPa. This shows that the first water shell is strongly bound to the anion whatever the temperature. The first two water shells form a cavity of a roughly spherical shape around the anion. By TDDFT method, we calculated the absorption spectra of hydrated Br at two temperatures and we compared the results with the experimental data.
at two temperatures and we compared the results with the experimental data.
Hata, Kuniki; Lin, M.; Katsumura, Yosuke*; Muroya, Yusa*; Fu, H. Y.*; Yamashita, Shinichi; Nakagawa, Hidehiko*
Journal of Radiation Research, 52(1), p.15 - 23, 2011/01
Times Cited Count:20 Percentile:59.76(Biology)Yan, Y.*; Lin, M.; Katsumura, Yosuke*; Fu, H.*; Muroya, Yusa*
Radiation Physics and Chemistry, 79(12), p.1234 - 1239, 2010/12
Times Cited Count:2 Percentile:17.46(Chemistry, Physical)The absorption spectra of solvated electrons in various alcohols were measured at different temperatures using pulse radiolysis technique, and then compared with currently available experimental data on the temperature dependent energy of absorption maximum 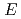
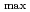 of solvated electrons in monohydric alcohols, diols and triol.
of solvated electrons in monohydric alcohols, diols and triol. 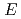
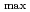 is larger in those alcohols having more OH groups at all the temperatures. The molecular structure effect, including OH numbers, OH position and carbon chain length, are investigated. For the primary alcohols with same OH group number and position, the temperature coefficient increases with increasing chain length. For the alcohols with same chain length and OH numbers, the temperature coefficient is larger for the symmetric alcohols than the asymmetric ones.
is larger in those alcohols having more OH groups at all the temperatures. The molecular structure effect, including OH numbers, OH position and carbon chain length, are investigated. For the primary alcohols with same OH group number and position, the temperature coefficient increases with increasing chain length. For the alcohols with same chain length and OH numbers, the temperature coefficient is larger for the symmetric alcohols than the asymmetric ones.
Fu, H. Y.*; Lin, M.; Katsumura, Yosuke*; Yokoya, Akinari; Hata, Kuniki; Muroya, Yusa*; Fujii, Kentaro; Shikazono, Naoya
Acta Biochimica et Biophysica Sinica, 42(7), p.489 - 495, 2010/07
Times Cited Count:12 Percentile:31.13(Biochemistry & Molecular Biology)Silybin (SLB) and similar analogues, namely, hesperetin (HESP), naringenin (NAN) and naringin (NAR), are believed to be active constituents of natural flavonoids that have been reported as chemopreventive agents for certain cancers. Moreover, SLB and analogues have been determined to fast repair DNA bases from oxidative damage by pulse radiolysis techniques. The present study was designed to evaluate the protective effects of SLB and analogues on soft X-ray-induced damage to plasmid DNA in vitro. Among the compounds tested, HESP and SLB were the most effective in preventing X-ray-induced formation of DNA single-strand breaks (SSB). A comparison of these results with other experiments showed that the ability of SLB and analogues to inhibit DNA damage in vitro correlated with the ability of the compounds to scavenge free radicals. Our work revealed that natural flavonoids, SLB and analogues may be used as potent protectors against radiation damage.
Fu, H.*; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Hata, Kuniki; Fujii, Kentaro; Yokoya, Akinari; Hatano, Yoshihiko
Radiation Physics and Chemistry, 78(12), p.1192 - 1197, 2009/12
Times Cited Count:30 Percentile:87.31(Chemistry, Physical)Lin, M.; Fu, H.*; Lampre, I.*; De Waele, V.*; Muroya, Yusa*; Yan, Y.*; Yamashita, Shinichi; Katsumura, Yosuke; Mostafavi, M.*
Journal of Physical Chemistry A, 113(44), p.12193 - 12198, 2009/10
Times Cited Count:7 Percentile:22.44(Chemistry, Physical)With a revisit of the absorption coefficient of the solvated electron in propane-1,2,3-triol, the temperature dependent behavior of the absorption spectrum of solvated electron was studied from room temperature to 573 K by pulse radiolysis techniques. The change in the absorption spectrum of solvated electron in propane-1,2,3-triol observed by cooling down from a high temperature to 333 K is compared with that occurring during the electron solvation process at 333 K. The effect of the specific molecular structure of propane-1,2,3-triol compared to other alcohols is discussed.
Fu, H. Y.*; Lin, M.; Muroya, Yusa*; Hata, Kuniki; Katsumura, Yosuke; Yokoya, Akinari; Shikazono, Naoya; Hatano, Yoshihiko
Free Radical Research, 43(9), p.887 - 897, 2009/09
Times Cited Count:24 Percentile:47.04(Biochemistry & Molecular Biology)Fu, H.*; Katsumura, Yosuke; Lin, M.; Hata, Kuniki; Muroya, Yusa*; Hatano, Yoshihiko
Journal of Radiation Research, 49(6), p.609 - 614, 2008/11
Times Cited Count:15 Percentile:45.11(Biology)The repair activities of silybin (SLB) and its analogues towards the oxidizing deoxyguanosine monophosphate (dGMP) hydroxyl radical adducts are investigated by pulse radiolytic techniques. On pulse irradiation of nitrous oxide saturated 2.0 mM dGMP aqueous solution containing 0.1 mM silybin at neutral pH, the transient absorption spectrum of the dGMP hydroxyl radical adducts decreases with the formation of the phenoxyl radical of silybin within tens of microseconds, indicating that there is a repair reaction between the dGMP hydroxyl radical adduct and silybin. The rate constant of the repair reaction is calculated to be 1.0 10
10 M
M s
s for silybin. The repair activity of hesperetin (HESP), naringin (NAN) and naringenin (NAR) towards hydroxyl radical adducts of dGMP are also studied.
for silybin. The repair activity of hesperetin (HESP), naringin (NAN) and naringenin (NAR) towards hydroxyl radical adducts of dGMP are also studied.
Fu, H.*; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*
Radiation Physics and Chemistry, 77(10-12), p.1300 - 1305, 2008/10
Times Cited Count:4 Percentile:29.49(Chemistry, Physical)In the present work, laser photolysis techniques have been used to study the transient species of silybin (abbreviated as SLB) in ethanol solutions. It has been demonstrated that the exposure of silybin to 248 nm excimer laser gives rise to monophotonic ionization with a high quantum yield. The transient species have been identified, and a series of related kinetics parameters have also been obtained. For the sake of comparison, we approached the problem using another way to reduce silybin, i.e. attack of solvated electrons, e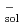 , produced in pulse radiolysis. The rate constant for reaction of e
, produced in pulse radiolysis. The rate constant for reaction of e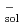 was determined to be 3.8
was determined to be 3.8 10
10 dm
dm mol
mol s
s at 720 nm, which is consistent with 4.6
at 720 nm, which is consistent with 4.6 10
10 dm
dm mol
mol s
s , obtained by laser photolysis.
, obtained by laser photolysis.
Fu, H.*; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Hata, Kuniki
no journal, ,
Hata, Kuniki; Katsumura, Yosuke; Lin, M.; Muroya, Yusa*; Fu, H.*; Kudo, Hisaaki*; Nakagawa, Keiichi*; Nakagawa, Hidehiko*
no journal, ,
The reactivity of edaravone with some oxidative radicals ( OH, N
OH, N
 , Br
, Br
 , SO
, SO
 , CClO
, CClO O
O
 ) has been measured by pulse radiolysis. The reaction product of edaravone with
) has been measured by pulse radiolysis. The reaction product of edaravone with  OH was different from those with other radicals. The rate constants of edaravone derivatives with
OH was different from those with other radicals. The rate constants of edaravone derivatives with  OH have also been determined by both direct measurement and a competition method using CO
OH have also been determined by both direct measurement and a competition method using CO
 and DMPO as competitors.
and DMPO as competitors.