Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Hu, F.-F.*; Qin, T.-Y.*; Ao, N.*; Xu, P. G.; Su, Y. H.; Parker, J. D.*; Shinohara, Takenao; Shobu, Takahisa; Kang, G.-Z.*; Ren, M.-M.; et al.
Journal of Traffic and Transportation Engineering, 25(2), p.75 - 93, 2025/04
 Bh in the
Bh in the  Cm(
Cm(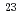 Na,5
Na,5 )
) Bh reaction and its decay properties
Bh reaction and its decay propertiesHaba, Hiromitsu*; Fan, F.*; Kaji, Daiya*; Kasamatsu, Yoshitaka*; Kikunaga, Hidetoshi*; Komori, Yukiko*; Kondo, Narumi*; Kudo, Hisaaki*; Morimoto, Koji*; Morita, Kosuke*; et al.
Physical Review C, 102(2), p.024625_1 - 024625_12, 2020/08
Times Cited Count:7 Percentile:55.65(Physics, Nuclear)Eichler, R.*; Asai, Masato; Brand, H.*; Chiera, N. M.*; Di Nitto, A.*; Dressler, R.*; D llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
EPJ Web of Conferences, 131, p.07005_1 - 07005_7, 2016/12
Times Cited Count:3 Percentile:71.33(Chemistry, Inorganic & Nuclear)In recent years gas-phase chemical studies assisted by physical pre-separation allowed for the productions and investigations of fragile single molecular species of superheavy elements. The latest highlight is the formation of very volatile hexacarbonyl compound of element 106, Sg(CO) . Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO)
. Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO) , W(CO)
, W(CO) , and Sg(CO)
, and Sg(CO) .
.
 and W(CO)
and W(CO)
Usoltsev, I.*; Eichler, R.*; Wang, Y.*; Even, J.*; Yakushev, A.*; Haba, Hiromitsu*; Asai, Masato; Brand, H.*; Di Nitto, A.*; D llmann, Ch. E.*; et al.
llmann, Ch. E.*; et al.
Radiochimica Acta, 104(3), p.141 - 151, 2016/03
Times Cited Count:34 Percentile:94.43(Chemistry, Inorganic & Nuclear)Conditions of the production and decomposition of hexacarbonyl complexes of short-lived Mo and W isotopes were investigated to study thermal stability of the heaviest group 6 hexacarbonyl complex Sg(CO) . A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO)
. A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO) and W(CO)
and W(CO) , indicating that the first bond dissociation energy of Sg(CO)
, indicating that the first bond dissociation energy of Sg(CO) could be determined with this technique.
could be determined with this technique.
Even, J.*; Ackermann, D.*; Asai, Masato; Block, M.*; Brand, H.*; Di Nitto, A.*; D llmann, Ch. E.*; Eichler, R.*; Fan, F.*; Haba, Hiromitsu*; et al.
llmann, Ch. E.*; Eichler, R.*; Fan, F.*; Haba, Hiromitsu*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(3), p.2457 - 2466, 2015/03
Times Cited Count:15 Percentile:74.22(Chemistry, Analytical)Rapid In situ synthesis of metal carbonyl complexes has been demonstrated using short-lived isotopes produced in nuclear fission and fusion reactions. The short-lived isotopes with high recoil energy directly react with carbon-monoxides and form carbonyl complexes. Only highly volatile complexes were fast transported in a gas stream to counting and chemistry devices. Short-lived Mo, Tc, Ru, Rh, W, Re, Os, and Ir were found to form volatile carbonyl complexes, while no volataile complex of Hf and Ta were detected. This technique has been applied to a chemical investigation of the superheavy element Sg (atomic number 106), and will be applicable to various fields of nuclear science with short-lived transition metal isotopes.
Even, J.*; Yakushev, A.*; D llmann, Ch. E.*; Haba, Hiromitsu*; Asai, Masato; Sato, Tetsuya; Brand, H.*; Di Nitto, A.*; Eichler, R.*; Fan, F. L.*; et al.
llmann, Ch. E.*; Haba, Hiromitsu*; Asai, Masato; Sato, Tetsuya; Brand, H.*; Di Nitto, A.*; Eichler, R.*; Fan, F. L.*; et al.
Science, 345(6203), p.1491 - 1493, 2014/09
Times Cited Count:67 Percentile:83.06(Multidisciplinary Sciences)A new superheavy element complex, a seaborgium carbonyl, has been successfully synthesized, and its adsorption property has been studied using a cryo-thermochromatography and  -detection apparatus COMPACT. Nuclear reaction products of short-lived
-detection apparatus COMPACT. Nuclear reaction products of short-lived  Sg preseparated with a gas-filled recoil ion separator GARIS at RIKEN were directly injected into a gas cell filled with He/CO mixture gas, and chemical reaction products of volatile carbonyl complexes were trasported to COMPACT. The Sg carbonyl complex detected with COMPACT was found to be very volatile with adsorption enthalpy of
Sg preseparated with a gas-filled recoil ion separator GARIS at RIKEN were directly injected into a gas cell filled with He/CO mixture gas, and chemical reaction products of volatile carbonyl complexes were trasported to COMPACT. The Sg carbonyl complex detected with COMPACT was found to be very volatile with adsorption enthalpy of 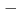 50 kJ/mol, from which we have concluded that this complex should be a Sg hexacarbonyl Sg(CO)
50 kJ/mol, from which we have concluded that this complex should be a Sg hexacarbonyl Sg(CO) . This is the first synthesis of organometallic compounds of transactinide elements for which only simple inorganic comounds have been synthesized so far.
. This is the first synthesis of organometallic compounds of transactinide elements for which only simple inorganic comounds have been synthesized so far.
Yakushev, A.*; Gates, J. M.*; T rler, A.*; Sch
rler, A.*; Sch del, M.; D
del, M.; D llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Block, M.*; Br
llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Block, M.*; Br chle, W.*; Dvorak, J.*; et al.
chle, W.*; Dvorak, J.*; et al.
Inorganic Chemistry, 53(3), p.1624 - 1629, 2014/02
Times Cited Count:113 Percentile:98.89(Chemistry, Inorganic & Nuclear)We report on a gas-solid chromatography study of the adsorption of element 114 (flerovium, Fl) on a Au surface. Fl was produced in the nuclear fusion reaction  Pu(
Pu( Ca, 3-4n)
Ca, 3-4n)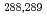 Fl and was isolated in-flight from the primary
Fl and was isolated in-flight from the primary  beam in a physical recoil separator. The adsorption behavior of Fl, its nuclear
beam in a physical recoil separator. The adsorption behavior of Fl, its nuclear  -decay product Cn, their lighter homologues in groups 14 and 12, i.e., Pb and Hg, and the noble gas Rn were studied simultaneously by isothermal gas chromatography and thermochromatography. Two Fl atoms were detected. They adsorbed on a Au surface at room temperature, but not as readily as Pb and Hg. The observed adsorption behavior of Fl points to a higher inertness compared to its nearest homologue in the group, Pb. However, the measured lower limit for the adsorption enthalpy of Fl on a Au surface points to the formation of a metal-metal bond of Fl with Au. Fl is the least reactive element in the group, but still a metal.
-decay product Cn, their lighter homologues in groups 14 and 12, i.e., Pb and Hg, and the noble gas Rn were studied simultaneously by isothermal gas chromatography and thermochromatography. Two Fl atoms were detected. They adsorbed on a Au surface at room temperature, but not as readily as Pb and Hg. The observed adsorption behavior of Fl points to a higher inertness compared to its nearest homologue in the group, Pb. However, the measured lower limit for the adsorption enthalpy of Fl on a Au surface points to the formation of a metal-metal bond of Fl with Au. Fl is the least reactive element in the group, but still a metal.
Kasamatsu, Yoshitaka*; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Li, Z.; Ishii, Yasuo; Tome, Hayato*; Sato, Tetsuya; Kikuchi, Takahiro; Nishinaka, Ichiro; et al.
Chemistry Letters, 38(11), p.1084 - 1085, 2009/10
Times Cited Count:17 Percentile:50.85(Chemistry, Multidisciplinary)We report on the characteristic anion-exchange behavior of the superheavy element dubnium (Db) with atomic number Z = 105 in HF/HNO solution at the fluoride ion concentration [F
solution at the fluoride ion concentration [F ] = 0.003 M. The result clearly demonstrates that the fluoro complex formation of Db is significantly different from that of the group-5 homologue Ta in the 6th period of the periodic table while the behavior of Db is similar to that of the lighter homologue Nb in the 5th period.
] = 0.003 M. The result clearly demonstrates that the fluoro complex formation of Db is significantly different from that of the group-5 homologue Ta in the 6th period of the periodic table while the behavior of Db is similar to that of the lighter homologue Nb in the 5th period.
 implanted silicon oxide thin films upon annealing treatments
implanted silicon oxide thin films upon annealing treatmentsYu, R. S.*; Maekawa, Masaki; Kawasuso, Atsuo; Sekiguchi, Takashi*; Wang, B. Y.*; Qin, X. B.*; Wang, Q. Z.*
Nuclear Instruments and Methods in Physics Research B, 267(18), p.3097 - 3099, 2009/09
Times Cited Count:3 Percentile:27.53(Instruments & Instrumentation)The structural evolution of silicon oxide films with Germanium implantation was traced with a positron beam equipped with positron annihilation Doppler broadening and lifetime spectrometers. Results indicate that the film structure change as a function of the annealing temperature could be divided into four stages: (I) T  300
300 C; (II) 300
C; (II) 300 C
C  T
T  500
500 C; (III) 600
C; (III) 600 C
C  T
T  800
800 C; (IV) T
C; (IV) T  900
900 C. In comparison with stage I, the increased positron annihilation Doppler broadening S values during stage II is ascribed to the annealing out of point defects and coalescence of intrinsic open volumes in silicon oxides. The obtained long positron lifetime and high S values without much fluctuation in stage III suggest a rather stable film structure. Further annealing above 900
C. In comparison with stage I, the increased positron annihilation Doppler broadening S values during stage II is ascribed to the annealing out of point defects and coalescence of intrinsic open volumes in silicon oxides. The obtained long positron lifetime and high S values without much fluctuation in stage III suggest a rather stable film structure. Further annealing above 900 C brings about dramatic change of the film structure with Ge precipitation. Positron annihilation spectroscopy is thereby a sensitive probe for the diagnosis of microstructure variation of silicon oxide thin films with nano-precipitation.
C brings about dramatic change of the film structure with Ge precipitation. Positron annihilation spectroscopy is thereby a sensitive probe for the diagnosis of microstructure variation of silicon oxide thin films with nano-precipitation.
 Cm(
Cm(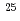 Mg, 4
Mg, 4 -5
-5 )
)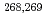 Hs reactions
Hs reactionsDvorak, J.*; Br chle, W.*; D
chle, W.*; D llmann, Ch. E.*; Dvorakova, Z.*; Eberhardt, K.*; Eichler, R.*; J
llmann, Ch. E.*; Dvorakova, Z.*; Eberhardt, K.*; Eichler, R.*; J ger, E.*; Nagame, Yuichiro; Qin, Z.*; Sch
ger, E.*; Nagame, Yuichiro; Qin, Z.*; Sch del, M.*; et al.
del, M.*; et al.
Physical Review C, 79(3), p.037602_1 - 037602_4, 2009/05
Times Cited Count:17 Percentile:68.93(Physics, Nuclear)no abstracts in English
 evaporation channel in the complete hot-fusion reaction
evaporation channel in the complete hot-fusion reaction 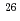 Mg +
Mg +  Cm leading to the new superheavy nuclide
Cm leading to the new superheavy nuclide 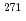 Hs
HsDvorak, J.*; Br chle, W.*; Chelnokov, M.*; D
chle, W.*; Chelnokov, M.*; D llmann, Ch. E.*; Dvorakova, Z.*; Eberhardt, K.*; J
llmann, Ch. E.*; Dvorakova, Z.*; Eberhardt, K.*; J ger, E.*; Kr
ger, E.*; Kr cken, R.*; Kuznetsov, A.*; Nagame, Yuichiro; et al.
cken, R.*; Kuznetsov, A.*; Nagame, Yuichiro; et al.
Physical Review Letters, 100(13), p.132503_1 - 132503_4, 2008/04
The analysis of a large body of heavy ion fusion reaction data with medium-heavy projectiles (6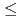 Z
Z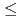 18) and actinide targets suggests a disappearance of the 3
18) and actinide targets suggests a disappearance of the 3 exit channel with increasing atomic number of the projectile. Here, we report a measurement of the excitation function of the reaction
exit channel with increasing atomic number of the projectile. Here, we report a measurement of the excitation function of the reaction  Cm(
Cm(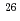 Mg,
Mg,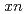 )
)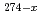 Hs and the observation of the new nuclide
Hs and the observation of the new nuclide 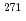 Hs produced in the 3
Hs produced in the 3 evaporation channel at a beam energy well below the Bass fusion barrier with a cross section comparable to the maxima of the 4
evaporation channel at a beam energy well below the Bass fusion barrier with a cross section comparable to the maxima of the 4 and 5
and 5 channels. This indicates the possible discovery of new neutron-rich transactinide nuclei using relatively light heavy ion beams of the most neutron-rich stable isotopes and actinide targets.
channels. This indicates the possible discovery of new neutron-rich transactinide nuclei using relatively light heavy ion beams of the most neutron-rich stable isotopes and actinide targets.
 evaporation channel in the complete hot-fusion reaction
evaporation channel in the complete hot-fusion reaction 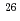 Mg+
Mg+ Cm leading to the new superheavy nuclide
Cm leading to the new superheavy nuclide 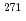 Hs
HsDvorak, J.*; Br chle, W.*; Chelnokov, M.*; D
chle, W.*; Chelnokov, M.*; D llmann, Ch. E.*; Dvorakova, Z.*; Eberhardt, K.*; J
llmann, Ch. E.*; Dvorakova, Z.*; Eberhardt, K.*; J ger, E.*; Kr
ger, E.*; Kr cken, R.*; Kuznetsov, A.*; Nagame, Yuichiro; et al.
cken, R.*; Kuznetsov, A.*; Nagame, Yuichiro; et al.
Physical Review Letters, 100(13), p.132503_1 - 132503_4, 2008/04
Times Cited Count:130 Percentile:94.90(Physics, Multidisciplinary)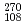 Hs
Hs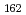
Dvorak, J.*; Br chle, W.*; Chelnokov, M.*; Dressler, R.*; D
chle, W.*; Chelnokov, M.*; Dressler, R.*; D llmann, Ch. E.*; Eberhardt, K.*; Gorshkov, V.*; J
llmann, Ch. E.*; Eberhardt, K.*; Gorshkov, V.*; J ger, E.*; Kr
ger, E.*; Kr cken, R.*; Kuznetsov, A.*; et al.
cken, R.*; Kuznetsov, A.*; et al.
Physical Review Letters, 97(24), p.242501_1 - 242501_4, 2006/12
Times Cited Count:162 Percentile:96.09(Physics, Multidisciplinary)no abstracts in English
 solutions
solutionsKasamatsu, Yoshitaka; Tome, Hayato; Toyoshima, Atsushi; Tsukada, Kazuaki; Asai, Masato; Ishii, Yasuo; Nishinaka, Ichiro; Sato, Tetsuya; Shinohara, Nobuo; Nagame, Yuichiro; et al.
no journal, ,
Anion-exchange behavior of  Db (half-life
Db (half-life 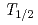 = 34 s) produced in the
= 34 s) produced in the  Cm(
Cm( F, 5
F, 5 ) reaction at the JAEA tandem accelerator was investigated in the mixed 0.89 M HF/0.3 M HNO
) reaction at the JAEA tandem accelerator was investigated in the mixed 0.89 M HF/0.3 M HNO solution ([F
solution ([F ] = 3
] = 3  10
10 M) with the automated ion-exchange separation apparatus coupled with the detection system for alpha-spectroscopy (AIDA). Anion-exchange behavior of its lighter homologues, Nb and Ta, was also studied under the same conditions using
M) with the automated ion-exchange separation apparatus coupled with the detection system for alpha-spectroscopy (AIDA). Anion-exchange behavior of its lighter homologues, Nb and Ta, was also studied under the same conditions using  Nb (
Nb (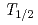 = 14.3 min) and
= 14.3 min) and  Ta (
Ta (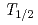 = 6.76 min) produced in the
= 6.76 min) produced in the 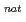 Ge(
Ge( F,
F, 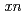 ) and
) and 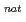 Gd(
Gd( F,
F, 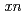 ) reactions, respectively. It was found that the adsorption probability on the anion-exchange resin is in the order of Ta
) reactions, respectively. It was found that the adsorption probability on the anion-exchange resin is in the order of Ta  Nb
Nb  Db under the present condition.
Db under the present condition.
 media
mediaKasamatsu, Yoshitaka; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Haba, Hiromitsu*; Ishii, Yasuo; Tome, Hayato; Nishinaka, Ichiro; Akiyama, Kazuhiko*; Kikunaga, Hidetoshi*; et al.
no journal, ,
Dubnium-262 was produced in the  Cm(
Cm( F, 5
F, 5 ) reaction at the JAEA tandem accelerator. Adsorption of Db on the anion-exchange resin was investigated in 0.89 M HF/0.3 M HNO
) reaction at the JAEA tandem accelerator. Adsorption of Db on the anion-exchange resin was investigated in 0.89 M HF/0.3 M HNO solution. The anion-exchange behavior of Nb, Ta, and Pa as homologues of Db was also examined in details in HF/HNO
solution. The anion-exchange behavior of Nb, Ta, and Pa as homologues of Db was also examined in details in HF/HNO solutions. From the comparison of those results, we found that the adsorption of Db on the anion-exchange resin is considerably weaker than that of Ta and is relatively similar to those of Nb and Pa in the studied conditions.
solutions. From the comparison of those results, we found that the adsorption of Db on the anion-exchange resin is considerably weaker than that of Ta and is relatively similar to those of Nb and Pa in the studied conditions.