Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
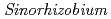 with
with 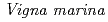 under high salt conditions using the ion beam mutation breeding
under high salt conditions using the ion beam mutation breedingMaruyama, Yudai*; Takeda, Kiyoko*; Tomooka, Norihiko*; Sato, Katsuya; Ono, Yutaka; Yokoyama, Tadashi*
JAEA-Review 2015-022, JAEA Takasaki Annual Report 2014, P. 99, 2016/02
Toda, Taro*; Maruyama, Takehiro*; Moritani, Kimikazu*; Moriyama, Hirotake*; Hayashi, Hirokazu
Denki Kagaku Oyobi Kogyo Butsuri Kagaku, 77(8), p.649 - 651, 2009/08
Times Cited Count:8 Percentile:19.4(Electrochemistry)The distribution coefficients of Am and Ce were measured in the eutectic LiCl-KCl/liquid Ga system at 773K. By using ZrCl as the oxide ion scavenger in order to avoid the formation of such oxychlorides as MO
as the oxide ion scavenger in order to avoid the formation of such oxychlorides as MO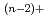 , the effect of oxide ion concentration was well controlled on the distribution coefficients of Am and Ce. The separation factor between Am and Ce was then obtained to be about 100. By comparing the present value with the other experimental and the predicted ones, it was confirmed that the Ga system was more selective than the Bi and Cd system.
, the effect of oxide ion concentration was well controlled on the distribution coefficients of Am and Ce. The separation factor between Am and Ce was then obtained to be about 100. By comparing the present value with the other experimental and the predicted ones, it was confirmed that the Ga system was more selective than the Bi and Cd system.
Toda, Taro*; Maruyama, Takehiro*; Moritani, Kimikazu*; Moriyama, Hirotake*; Hayashi, Hirokazu
Journal of Nuclear Science and Technology, 46(1), p.18 - 25, 2009/01
Times Cited Count:41 Percentile:92.56(Nuclear Science & Technology)The excess thermodynamic quantities of lanthanides and actinides in molten salts and liquid metals were studied for reductive extraction of minor actinides. The excess enthalpies and entropies of those elements in the molten chloride phase were found to be correlated with the ionic radii of metal ions possibly due to complex formation. In the liquid metal phase, on the other hand, the excess enthalpies were explained with Miedema's atomistic model and the excess entropies were explained with the vibrational entropy due to alloy formation. Using these correlations and models, some missing values of the excess thermodynamic quantities were evaluated and the separation factors of minor actinides from lanthanides were calculated in different reductive extraction systems. The higher separation factors were obtained in the system using aluminum or gallium than in the system using bismuth or cadmium as the liquid metal phase.
Toda, Taro*; Maruyama, Takehiro*; Moritani, Kimikazu*; Moriyama, Hirotake*; Hayashi, Hirokazu
Proceedings of 2008 Joint Symposium on Molten Salts (USB Flash Drive), p.933 - 938, 2008/10
The distribution coefficients of Am and Ce were measured in the LiCl-KCl/Ga system at 773 K. By using ZrCl as the oxide ion scavenger in order to avoid the formation of such oxychlorides as CeO
as the oxide ion scavenger in order to avoid the formation of such oxychlorides as CeO and AmO
and AmO , the effect of oxide ion concentration was well controlled on the distribution coefficients of Am and Ce. The separation factor between Am and Ce was then obtained to be about 100. By comparing the present value with the other experimental and the predicted ones, it was confirmed that solvent metals were ordered from the most selective to the less selective one as Al
, the effect of oxide ion concentration was well controlled on the distribution coefficients of Am and Ce. The separation factor between Am and Ce was then obtained to be about 100. By comparing the present value with the other experimental and the predicted ones, it was confirmed that solvent metals were ordered from the most selective to the less selective one as Al Ga
Ga Bi
Bi Cd.
Cd.
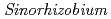 with
with 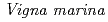 under high salt conditions using the ion beam mutation breeding
under high salt conditions using the ion beam mutation breedingYokoyama, Tadashi*; Maruyama, Yudai*; Takeda, Kiyoko*; Tomooka, Norihiko*; Sato, Katsuya; Ono, Yutaka
no journal, ,
no abstracts in English