Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
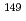 Sm M
Sm M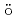 ssbauer and Sm L
ssbauer and Sm L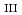 -edge X-ray absorption spectroscopies
-edge X-ray absorption spectroscopiesTsutsui, Satoshi; Higashinaka, Ryuji*; Nakamura, Raito*; Fujiwara, Kosuke*; Nakamura, Jin*; Kobayashi, Yoshio*; Ito, Takashi; Yoda, Yoshitaka*; Kato, Kazuo*; Nitta, Kiyofumi*; et al.
Hyperfine Interactions, 242(1), p.32_1 - 32_10, 2021/12
Times Cited Count:1 Percentile:84.42(Physics, Atomic, Molecular & Chemical)Yamagishi, Isao; Nagaishi, Ryuji; Kato, Chiaki; Morita, Keisuke; Terada, Atsuhiko; Kamiji, Yu; Hino, Ryutaro; Sato, Hiroyuki; Nishihara, Kenji; Tsubata, Yasuhiro; et al.
Journal of Nuclear Science and Technology, 51(7-8), p.1044 - 1053, 2014/07
Times Cited Count:19 Percentile:78.38(Nuclear Science & Technology)For safe storage of zeolite wastes generated by treatment of radioactive saline water at the Fukushima Daiichi Nuclear Power Station, properties of the Herschelite adsorbent were studied and its adsorption vessel was evaluated for hydrogen production and corrosion. Hydrogen production depends on its water level and dissolved species because hydrogen is oxidized by radicals in water. It is possible to evaluate hydrogen production rate in Herschelite submerged in seawater or pure water by taking into account of the depth effect of the water. The reference vessel of decay heat 504 W with or without residual pure water was evaluated for the hydrogen concentration by thermal hydraulic analysis using obtained fundamental properties. Maximum hydrogen concentration was below the lower explosive limit (4 %). The steady-state corrosion potential of a stainless steel 316L increased with absorbed dose rate but its increase was repressed by the presence of Herschelite. At 750 Gy/h and  60
60 C which were values evaluated at the bottom of the vessel of 504 W, the localized corrosion of SUS316L contacted with Herschelite would not immediately occur under 20,000 ppm of Cl
C which were values evaluated at the bottom of the vessel of 504 W, the localized corrosion of SUS316L contacted with Herschelite would not immediately occur under 20,000 ppm of Cl concentration.
concentration.
Yamagishi, Isao; Nagaishi, Ryuji; Terada, Atsuhiko; Kamiji, Yu; Kato, Chiaki; Morita, Keisuke; Nishihara, Kenji; Tsubata, Yasuhiro; Ji, W.*; Fukushima, Hisashi*; et al.
IAEA-CN-211 (Internet), 5 Pages, 2013/01
Since the accident at Fukushima Daiichi Nuclear Power Station, a large amount of radioactive contaminated water has been generated to cool damaged reactor cores. Adsorption of cesium with zeolite-like media was employed for treatment of this contaminated saline water. As spent zeolite media are highly radioactive, their safe storage is a pressing issue. Japan Atomic Energy Agency has extensively conducted R&D on the management of secondary wastes produced by the operation of the treatment system. Subjects on the safe storage of spent zeolites include the analysis of their characteristics and the evaluation of effectiveness of the present safety measures in consideration of decay heat emission and hydrogen generation by water radiolysis as well as durability of vessels exposed to saline. Preliminary results obtained are described in the present paper.
Kondo, Takafumi*; Asano, Akira*; Yang, J.*; Norizawa, Kimihiro*; Takahashi, Kenji*; Taguchi, Mitsumasa; Nagaishi, Ryuji; Kato, Ryuji*; Yoshida, Yoichi*
Radiation Physics and Chemistry, 78(12), p.1157 - 1160, 2009/12
Times Cited Count:28 Percentile:85.84(Chemistry, Physical)Spectrum and kinetic behavior of solvated electrons (e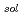
 ) in alkyl ammonium ionic liquids (ILs),
) in alkyl ammonium ionic liquids (ILs), 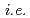 DEMMA-TFSI, DEMMA-BF4, TMPA-TFSI, PP13-TFSI, P13-TFSI and P14-TFSI were investigated by pulse radiolysis method. The e
DEMMA-TFSI, DEMMA-BF4, TMPA-TFSI, PP13-TFSI, P13-TFSI and P14-TFSI were investigated by pulse radiolysis method. The e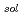
 in the ILs have same absorption peak at 1100 nm with a molar absorption coefficient of 1.5-2.3
in the ILs have same absorption peak at 1100 nm with a molar absorption coefficient of 1.5-2.3 10
10 dm
dm mol
mol cm
cm . The reaction rate constant of e
. The reaction rate constant of e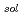
 with Pyrene (Py) was 1.5-3.5
with Pyrene (Py) was 1.5-3.5 10
10 dm
dm mol
mol s
s . These values were about one order of magnitude higher than that of diffusion controlled limit, which was calculated from measured viscosity. The dry electron (e
. These values were about one order of magnitude higher than that of diffusion controlled limit, which was calculated from measured viscosity. The dry electron (e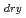
 ) reacts with Py, and its rate constant in DEMMA-TFSI was 7.9
) reacts with Py, and its rate constant in DEMMA-TFSI was 7.9 10
10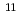 dm
dm mol
mol s
s , three orders of magnitude higher than that of the e
, three orders of magnitude higher than that of the e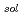
 reactions.
reactions.
Yang, J.*; Kondo, Takafumi*; Norizawa, Kimihiro*; Nagaishi, Ryuji; Taguchi, Mitsumasa; Takahashi, Kenji*; Kato, Ryuji*; Anishchik, S. V.*; Yoshida, Yoichi*; Tagawa, Seiichi*
Radiation Physics and Chemistry, 77(10-12), p.1233 - 1238, 2008/10
Times Cited Count:25 Percentile:82.53(Chemistry, Physical)First observation of picosecond dynamics in ionic liquid of DEMMA-TFSI in radiation chemistry was reported. It is found that the electron produced by ionization is solvated to full solvation in ionic liquid with a rate constant of 3.9 10
10 s
s . The yield of solvated electrons in picosecond time region is 1.2
. The yield of solvated electrons in picosecond time region is 1.2 10
10 mol J
mol J . The dry electron in ionic liquid reacts rapidly with biphenyl and pyrene with a rate constant of 3.8 to 7.9
. The dry electron in ionic liquid reacts rapidly with biphenyl and pyrene with a rate constant of 3.8 to 7.9 10
10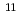 L mol
L mol s
s . The geminate ion recombination in n-dodecane and n-hexane was also observed by monitoring the transient absorptions at 523 nm.
. The geminate ion recombination in n-dodecane and n-hexane was also observed by monitoring the transient absorptions at 523 nm.
Kato, Ryuji*; Yoshida, Yoichi*; Katsumura, Yosuke; Takahashi, Kenji*
Journal of Physical Chemistry B, 111(18), p.4770 - 4774, 2007/05
Times Cited Count:54 Percentile:76.97(Chemistry, Physical)Kimura, Takaumi; Nagaishi, Ryuji; Kato, Yoshiharu; Yoshida, Zenko
Journal of Alloys and Compounds, 323-324(1-4), p.164 - 168, 2001/07
Times Cited Count:50 Percentile:87.64(Chemistry, Physical)no abstracts in English
Kimura, Takaumi; Nagaishi, Ryuji; Kato, Yoshiharu; Yoshida, Zenko
Radiochimica Acta, 89(3), p.125 - 130, 2001/05
Times Cited Count:66 Percentile:96.81(Chemistry, Inorganic & Nuclear)no abstracts in English
Takahashi, Kenji*; Hiejima, Yusuke*; Kato, Ryuji*; Nagaishi, Ryuji; Taguchi, Mitsumasa; Kojima, Takuji; Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*
no journal, ,
Solvated electrons were studied using the pulse radiolysis and photo ionization methods. The Vis-NIR spectra and reactivity of the solvated electrons in ionic liquids are discussed.
Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*; Nagaishi, Ryuji; Taguchi, Mitsumasa; Takahashi, Kenji*; Kato, Ryuji*
no journal, ,
The effect of the coexistence water on the solvated electrons in ammonium ionic liquids was studied by the electron pulse radiolysis method. The optical absorption band of the solvation electron decreased by coexistence of water, and new absorption band like a hydrated electron were generated in the medium wavelength region between solvated electron and hydrated electron. Furthermore, if quantity of water increase in the hydrophilic ionic liquid, it was found that the spectrum changed into the hydrated electron.
Kondo, Takafumi*; Yang, J.*; Yoshida, Yoichi*; Nagaishi, Ryuji; Taguchi, Mitsumasa; Takahashi, Kenji*; Kato, Ryuji*
no journal, ,
no abstracts in English
Yamagishi, Isao; Kato, Chiaki; Nagaishi, Ryuji; Arisaka, Makoto; Uruga, Kazuyoshi*; Tsukada, Takeshi*
no journal, ,
no abstracts in English