Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yoshikawa, Hideki; Shibata, Masahiro; Sasamoto, Hiroshi; Iijima, Kazuki; Sato, Haruo; Kitamura, Akira; Ishidera, Takamitsu; Fujiwara, Kenso; Kurosawa, Susumu; Xia, X.; et al.
Hoshasei Haikibutsu Anzen Kenkyu Nenji Keikaku (Heisei-13-Nendo Heisei-17-Nendo) Kenkyu Seika Hokokushu, p.153 - 170, 2006/03
Heisei-17-Nendo) Kenkyu Seika Hokokushu, p.153 - 170, 2006/03
no abstracts in English
Xia, X.; Kamei, Gento; Iijima, Kazuki; Shibata, Masahiro
Materials Research Society Symposium Proceedings, Vol.932, p.933 - 941, 2005/09
To match the demand of establishment of reliable safety assessment methodology for HLW disposalin Japan, sorption behavior of radionuclides is required to be studied for sedimentary rock. Sorption of Se was conducted by batch sorption tests by using sedimentary rock samples and corresponding saline groundwater from Horonobe URL site under reducing condition, with consideration of site-specific conditions. Spectroscopic analyses were performed by X-ray absorption near-edge structure (XANES) after the sorption tests to identify the oxidation states of Se on the sedimentary rock. The sorption tests performed under various parameters allowed to identify that the dominant factors affecting the Se sorption are iron-bearing minerals or oxides and the oganic matter. The XANES analytical results revealed that most Se was Se(0) in the case of natural groundwater, while only some proportions of Se was Se(0) in the case of synthetic groundwater. These suggested that the organic matter probably affect the sorption through changing Se oxidation states, and in a natural sedimentary /saline groundwater system, reduction of selenium to be Se(0), could be expected to be an important sorption mechanism.
Xia, X.; Shibata, Masahiro; Kitamura, Akira; Kamei, Gento
P.779-781, p.779 - 781, 2004/08
A systematic sorption study on cesium, predicted to be a key radionuclide in the current safety assessment, was conducted on the sedimentary rock from the Horonobe URL, as the first step of linking sorption study in laboratory and modeling prediction of sorption at site.
Arima, Tatsumi*; Idemitsu, Kazuya*; Xia, X.; Ishidera, Takamitsu; Iijima, Kazuki
JNC TY8400 2004-005, 59 Pages, 2004/05
For the safety assessment of high level waste disposal, it is necessary to investigate the radionuclide migration in the presence of corrosion products of carbon steel under reducing condition. In this study, the reliable apparent diffusion coefficients of neptunium (Np) in bentonite were obtained by in-diffusion method and migration of the corrosion products was investigated. Furthermore, effects of the corrosion of carbon steel on Np diffusion were discussed. The corrosion of carbon steel under reducing condition provided Fe ions, which were considered to migrate in the interlayer spaces of montmorillonite sheets of bentonite exchanging with two Na
ions, which were considered to migrate in the interlayer spaces of montmorillonite sheets of bentonite exchanging with two Na ions. The corrosion rate of carbon steel was controlled by the diffusivity of Fe
ions. The corrosion rate of carbon steel was controlled by the diffusivity of Fe ions into bentonite when they were accumulated at the surface of carbon steel. The corrosion rate increased with increasing dry density of bentonite because of the increase of the diffusivity of Fe
ions into bentonite when they were accumulated at the surface of carbon steel. The corrosion rate increased with increasing dry density of bentonite because of the increase of the diffusivity of Fe ions. The corrosion rate was estimated to be ~0.1 micro m/y, which was remarkable lower than the setting value, 20 micro m/y, in the second progress report. The Np profiles in the bentonite consisted of two overlapping slopes, a fast and a slow fractions, for both the experiments with and without carbon steel. The apparent diffusion coefficients of the fast and the slow fraction of Np were 10
ions. The corrosion rate was estimated to be ~0.1 micro m/y, which was remarkable lower than the setting value, 20 micro m/y, in the second progress report. The Np profiles in the bentonite consisted of two overlapping slopes, a fast and a slow fractions, for both the experiments with and without carbon steel. The apparent diffusion coefficients of the fast and the slow fraction of Np were 10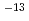 ~10
~10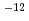 m
m /s and 10
/s and 10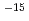 ~10
~10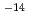 m
m /s, which were considered to be the diffusion of Np(V) and Np(IV), respectively. The corrosion of carbon steel provided strong reducing condition to keep most Np in the low oxidation state, Np(IV), which has lower solubility and mobility than Np(V). Therefore, it could be expected that the corrosion of carbon steel will restrain effectively migration of Np into the bentonite.
/s, which were considered to be the diffusion of Np(V) and Np(IV), respectively. The corrosion of carbon steel provided strong reducing condition to keep most Np in the low oxidation state, Np(IV), which has lower solubility and mobility than Np(V). Therefore, it could be expected that the corrosion of carbon steel will restrain effectively migration of Np into the bentonite.
Xia, X.; Ishidera, Takamitsu; Arima, Tatsumi*; Inagaki, Yaohiro*
P.290-291(2003), p.290 - 291, 2003/00
None
Ishidera, Takamitsu; Xia, X.; Idemitsu, Kazuya*; Kikuchi, Yoshio*
no journal, ,
no abstracts in English
Hama, Katsuhiro; Kunimaru, Takanori; Xia, X.; Nagao, Seiya*
no journal, ,
The status of study on organic matter in groundwater is presented.
Doi, Reisuke; Xia, X.; Shibata, Masahiro; Kitamura, Akira; Yoshikawa, Hideki
no journal, ,
Assessment for radionuclides sorption onto a rock in deep underground is important. The distribution coefficient, K is a available parameter to assess the sorption and the method to predict the sorption in deep environment has to be established. We applied a model that the sorption of Cs is dominated by the ion exchange reaction on the illite for experimental results of Cs sorption onto Horonobe sedimentary rock. The equilibrium concentration of Cs was calculated the geochemical calculation code, PHREEQC under the experiment condition with major cations and solid/liquid ratio to estimate the K
is a available parameter to assess the sorption and the method to predict the sorption in deep environment has to be established. We applied a model that the sorption of Cs is dominated by the ion exchange reaction on the illite for experimental results of Cs sorption onto Horonobe sedimentary rock. The equilibrium concentration of Cs was calculated the geochemical calculation code, PHREEQC under the experiment condition with major cations and solid/liquid ratio to estimate the K value. Comparing the experimental results with the calculated one, the model could explain well the dependence of K
value. Comparing the experimental results with the calculated one, the model could explain well the dependence of K values on Cs equilibrium concentration. By using the model, it is possible to assess for Cs sorption onto a sedimentary rock. However, there were some differences between the experimental K
values on Cs equilibrium concentration. By using the model, it is possible to assess for Cs sorption onto a sedimentary rock. However, there were some differences between the experimental K values and the calculated one. One of the causes for that was the uncertainties of analytical technique of illite content.
values and the calculated one. One of the causes for that was the uncertainties of analytical technique of illite content.