Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ikeda, Kazutaka*; Sashida, Sho*; Otomo, Toshiya*; Oshita, Hidetoshi*; Honda, Takashi*; Hawai, Takafumi*; Saito, Hiraku*; Ito, Shinichi*; Yokoo, Tetsuya*; Sakaki, Koji*; et al.
International Journal of Hydrogen Energy, 51(Part A), p.79 - 87, 2024/01
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)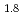 and TiH
and TiH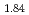 up to 21 GPa by incoherent inelastic neutron scattering
up to 21 GPa by incoherent inelastic neutron scatteringHattori, Takanori; Nakamura, Mitsutaka; Iida, Kazuki*; Machida, Akihiko*; Sano, Asami; Machida, Shinichi*; Arima, Hiroshi*; Oshita, Hidetoshi*; Honda, Takashi*; Ikeda, Kazutaka*; et al.
Physical Review B, 106(13), p.134309_1 - 134309_9, 2022/10
Times Cited Count:0 Percentile:0(Materials Science, Multidisciplinary)Hydrogen vibration excitations of fluorite-type ZrH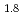 and TiH
and TiH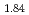 were investigated up to 21 GPa and 4 GPa, respectively, by incoherent inelastic neutron scattering experiments. The first excitation energies increased with pressure, as described by the equations
were investigated up to 21 GPa and 4 GPa, respectively, by incoherent inelastic neutron scattering experiments. The first excitation energies increased with pressure, as described by the equations 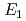 (meV) = 141.4(2) + 1.02(2)
(meV) = 141.4(2) + 1.02(2) (GPa) and
(GPa) and 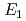 (meV) = 149.4(1) + 1.21(8)
(meV) = 149.4(1) + 1.21(8) (GPa) for ZrH
(GPa) for ZrH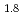 and TiH
and TiH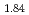 , respectively. Coupling with pressure dependence of lattice parameters, the relations between metal-hydrogen distance (
, respectively. Coupling with pressure dependence of lattice parameters, the relations between metal-hydrogen distance (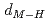 ) and
) and 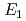 are found to be well described by the equations
are found to be well described by the equations 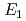 (meV) = 1.62(9)
(meV) = 1.62(9) 10
10
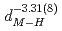 (
(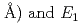 (meV) = 1.47(21)
(meV) = 1.47(21) 10
10
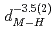 (AA), respectively. The slopes of these curves are much steep compared to the previously reported trend in various fluorite-type metal hydrides at ambient pressure. The hydrogen wave function spreading showed that the local potential field for a hydrogen atom shrinks more intensively than the tetrahedral site. These behavior is likely caused by the rigid metal ion core and the resulting confinement of the hydrogen atom in the narrower potential field at high pressures.
(AA), respectively. The slopes of these curves are much steep compared to the previously reported trend in various fluorite-type metal hydrides at ambient pressure. The hydrogen wave function spreading showed that the local potential field for a hydrogen atom shrinks more intensively than the tetrahedral site. These behavior is likely caused by the rigid metal ion core and the resulting confinement of the hydrogen atom in the narrower potential field at high pressures.
 He neutron spin filter at J-PARC
He neutron spin filter at J-PARCOkudaira, Takuya; Oku, Takayuki; Ino, Takashi*; Hayashida, Hirotoshi*; Kira, Hiroshi*; Sakai, Kenji; Hiroi, Kosuke; Takahashi, Shingo*; Aizawa, Kazuya; Endo, Hitoshi*; et al.
Nuclear Instruments and Methods in Physics Research A, 977, p.164301_1 - 164301_8, 2020/10
Times Cited Count:10 Percentile:79.13(Instruments & Instrumentation)Nakajima, Kenji; Kawakita, Yukinobu; Ito, Shinichi*; Abe, Jun*; Aizawa, Kazuya; Aoki, Hiroyuki; Endo, Hitoshi*; Fujita, Masaki*; Funakoshi, Kenichi*; Gong, W.*; et al.
Quantum Beam Science (Internet), 1(3), p.9_1 - 9_59, 2017/12
The neutron instruments suite, installed at the spallation neutron source of the Materials and Life Science Experimental Facility (MLF) at the Japan Proton Accelerator Research Complex (J-PARC), is reviewed. MLF has 23 neutron beam ports and 21 instruments are in operation for user programs or are under commissioning. A unique and challenging instrumental suite in MLF has been realized via combination of a high-performance neutron source, optimized for neutron scattering, and unique instruments using cutting-edge technologies. All instruments are/will serve in world-leading investigations in a broad range of fields, from fundamental physics to industrial applications. In this review, overviews, characteristic features, and typical applications of the individual instruments are mentioned.
Kodama, Katsuaki; Igawa, Naoki; Shamoto, Shinichi; Ikeda, Kazutaka*; Oshita, Hidetoshi*; Kaneko, Naokatsu*; Otomo, Toshiya*; Suzuya, Kentaro; Hoshikawa, Akinori*; Ishigaki, Toru*
JPS Conference Proceedings (Internet), 8, p.034002_1 - 034002_6, 2015/09
 O
O
Kodama, Katsuaki; Igawa, Naoki; Shamoto, Shinichi; Ikeda, Kazutaka*; Oshita, Hidetoshi*; Kaneko, Naokatsu*; Otomo, Toshiya*; Suzuya, Kentaro; Hoshikawa, Akinori*; Ishigaki, Toru*
JPS Conference Proceedings (Internet), 3, p.013012_1 - 013012_6, 2014/06
Shamoto, Shinichi; Kodama, Katsuaki; Imaki, Tadashi*; Nakatani, Takeshi; Oshita, Hidetoshi*; Kaneko, Naokatsu*; Masuko, Kenji*; Sakamoto, Kensaku; Yamaguchi, Kenji; Suzuya, Kentaro; et al.
JPS Conference Proceedings (Internet), 1, p.014011_1 - 014011_5, 2014/03
2D neutron diffraction imaging of an ammonite fossil was carried out at high-intensity total diffractometer NOVA in J-PARC. Observed diffraction profiles consist of calcite, siderite and amorphous structures.
 O
O
Kodama, Katsuaki; Igawa, Naoki; Shamoto, Shinichi; Ikeda, Kazutaka*; Oshita, Hidetoshi*; Kaneko, Naokatsu*; Otomo, Toshiya*; Suzuya, Kentaro
Journal of the Physical Society of Japan, 82(9), p.094601_1 - 094601_6, 2013/09
Times Cited Count:11 Percentile:58.33(Physics, Multidisciplinary)Machida, Akihiko; Honda, Mitsunori*; Hattori, Takanori; Sano, Asami; Watanuki, Tetsu; Katayama, Yoshinori; Aoki, Katsutoshi; Oshita, Hidetoshi*; Ikeda, Kazutaka*; Otomo, Toshiya*
Hamon, 23(2), p.131 - 136, 2013/05
Recently, we have revealed formation of NaCl-type LaD using synchrotron radiation X-ray diffraction and neutron diffraction measurements under high pressure. Previously, we have found that LaH decomposes into two phases, which have different hydrogen compositions, the H-poor and H-rich phases, at 11 GPa at room temperature by synchrotron radiation X-ray diffraction. Recent neutron diffraction measurements on LaD
decomposes into two phases, which have different hydrogen compositions, the H-poor and H-rich phases, at 11 GPa at room temperature by synchrotron radiation X-ray diffraction. Recent neutron diffraction measurements on LaD confirmed the formation of a NaCl-type LaD as the D-poor phase. This is the first observation of a formation of the rare-earth metal monodeuteride. Present result indicates that that rare-earth metal can form a series of stoichiometric hydrides, such as mono-, di-, and trihydride with the fcc metal lattice.
confirmed the formation of a NaCl-type LaD as the D-poor phase. This is the first observation of a formation of the rare-earth metal monodeuteride. Present result indicates that that rare-earth metal can form a series of stoichiometric hydrides, such as mono-, di-, and trihydride with the fcc metal lattice.
Kai, Tetsuya; Maekawa, Fujio; Oshita, Hidetoshi*; Sato, Hirotaka; Shinohara, Takenao; Oi, Motoki; Harada, Masahide; Uno, Shoji*; Otomo, Toshiya*; Kamiyama, Takashi*; et al.
Physics Procedia, 43, p.111 - 120, 2013/00
Times Cited Count:19 Percentile:98.08(Physics, Applied)A neutron imaging instrument is in construction at the pulsed neutron source of J-PARC. Some demonstration experiments in NOBORU/J-PARC have carried. To explore widespread applications of the newly built instruments it is indispensable to indicate the range of coverage of this technique. In the presentation, the authors describe the results of some of demonstration measurements, and then compare all elements with the values obtained by multiplying peak cross section and peak widths of neutron resonances in evaluated nuclear data libraries. This value is expected to be a good measure to exhibit visibility of each element in this technique.

Machida, Akihiko; Honda, Mitsunori*; Hattori, Takanori; Sano, Asami; Watanuki, Tetsu; Katayama, Yoshinori; Aoki, Katsutoshi; Komatsu, Kazuki*; Arima, Hiroshi*; Oshita, Hidetoshi*; et al.
Physical Review Letters, 108(20), p.205501_1 - 205501_5, 2012/05
Times Cited Count:17 Percentile:67.41(Physics, Multidisciplinary)Hydrogen atoms absorbed in a metal occupy the interstitial sites of the metal lattice. In an fcc metal lattice, each metal atom has two tetrahedral (T) and one octahedral (O) sites that can accommodate hydrogen. Rare-earth metal La forms T-site occupied LaH and fully occupied LaH
and fully occupied LaH . O-site occupied or NaCl-type monohydride has yet to be reported for rare-earth metals. Previous X-ray diffraction measurements revealed the pressure-induced decomposition of an fcc-LaH
. O-site occupied or NaCl-type monohydride has yet to be reported for rare-earth metals. Previous X-ray diffraction measurements revealed the pressure-induced decomposition of an fcc-LaH into H-rich and H-poor phases around 11 GPa. The present neutron diffraction measurements on LaD
into H-rich and H-poor phases around 11 GPa. The present neutron diffraction measurements on LaD confirm the formation of NaCl-type LaD as a counterpart of the D-rich LaD
confirm the formation of NaCl-type LaD as a counterpart of the D-rich LaD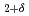 by disproportionation. First-principle calculations demonstrate that the NaCl-type LaH is stabilized at high pressures. Finding the NaCl-type LaH will pave the way for investigations on the site-dependent nature of hydrogen-metal interactions.
by disproportionation. First-principle calculations demonstrate that the NaCl-type LaH is stabilized at high pressures. Finding the NaCl-type LaH will pave the way for investigations on the site-dependent nature of hydrogen-metal interactions.
Suzuya, Kentaro; Takata, Shinichi; Shamoto, Shinichi; Otomo, Toshiya*; Misawa, Masakatsu*; Kaneko, Naokatsu*; Oshita, Hidetoshi*
no journal, ,
Design concept of high-intensity total diffractometer of J-PARC is wide-Q range measurements with the highest neutron intensity by the pulsed neutron source. The resolution of the diffractometer is reasonable for crystalline materials and the diffractometer will cover very wide research fields in term nano structure and/or non-crystalline materials. Hydrogen absorption mechanism will be intensively investigated with the diffractometer.
Honda, Mitsunori; Hattori, Takanori; Sano, Asami; Machida, Akihiko; Katayama, Yoshinori; Arima, Hiroshi; Komatsu, Kazuki*; Oshita, Hidetoshi*; Otomo, Toshiya*
no journal, ,
no abstracts in English
Honda, Mitsunori; Hattori, Takanori; Sano, Asami; Machida, Akihiko; Katayama, Yoshinori; Arima, Hiroshi; Komatsu, Kazuki*; Oshita, Hidetoshi*; Otomo, Toshiya*
no journal, ,
no abstracts in English
 study by high pressure neutron diffraction using NOVA at J-PARC
study by high pressure neutron diffraction using NOVA at J-PARCHonda, Mitsunori; Hattori, Takanori; Sano, Asami; Machida, Akihiko; Katayama, Yoshinori; Arima, Hiroshi; Komatsu, Kazuki*; Oshita, Hidetoshi*; Otomo, Toshiya*
no journal, ,
no abstracts in English
Honda, Mitsunori; Hattori, Takanori; Machida, Akihiko; Arima, Hiroshi; Sano, Asami; Katayama, Yoshinori; Aoki, Katsutoshi; Komatsu, Kazuki*; Oshita, Hidetoshi*; Otomo, Toshiya*
no journal, ,
no abstracts in English
Honda, Mitsunori; Hattori, Takanori; Machida, Akihiko; Arima, Hiroshi; Sano, Asami; Katayama, Yoshinori; Aoki, Katsutoshi; Komatsu, Kazuki*; Oshita, Hidetoshi*; Otomo, Toshiya*
no journal, ,
no abstracts in English

Aoki, Katsutoshi; Machida, Akihiko; Honda, Mitsunori; Hattori, Takanori; Sano, Asami; Watanuki, Tetsu; Katayama, Yoshinori; Komatsu, Kazuki*; Arima, Hiroshi; Otomo, Toshiya*; et al.
no journal, ,
Neutron diffraction measurements have revealed that LaD undergoes phase separation at high pressure with the relocation of deuterium atoms in the interstitial sites of La metal lattice. Deuterium atoms, which occupy the tetrahedral sites of the fcc metal lattice in LaD
undergoes phase separation at high pressure with the relocation of deuterium atoms in the interstitial sites of La metal lattice. Deuterium atoms, which occupy the tetrahedral sites of the fcc metal lattice in LaD , move into the empty octahedral sites at 11 GPa to form LaD and LaD
, move into the empty octahedral sites at 11 GPa to form LaD and LaD both having fcc metal lattices. Mono-hydride with an NaCl structure, which is common for mono-hydrides of transition metals, is formed in rare-earth metals for the first time. The first-principle calculations showed that LaH
both having fcc metal lattices. Mono-hydride with an NaCl structure, which is common for mono-hydrides of transition metals, is formed in rare-earth metals for the first time. The first-principle calculations showed that LaH is stable at low pressure and it undergoes a phase separation into LaH and LaH
is stable at low pressure and it undergoes a phase separation into LaH and LaH at 10 GPa, which is excellent agreement with the experimental results. Enthalpy comparison shows that unusual volume contraction in LaH
at 10 GPa, which is excellent agreement with the experimental results. Enthalpy comparison shows that unusual volume contraction in LaH than LaH
than LaH explains the phase separation phenomena. Lattice dynamics calculations on these lanthanum hydrides shed light on the detailed mechanism.
explains the phase separation phenomena. Lattice dynamics calculations on these lanthanum hydrides shed light on the detailed mechanism.

Aoki, Katsutoshi; Machida, Akihiko; Honda, Mitsunori; Hattori, Takanori; Sano, Asami; Watanuki, Tetsu; Katayama, Yoshinori; Komatsu, Kazuki*; Arima, Hiroshi; Otomo, Toshiya*; et al.
no journal, ,
Synchrotron X-ray and neutron diffraction measurements have revealed that LaD undergoes phase separation at high pressure with the relocation of deuterium atoms in the interstitial sites of La metal lattice. Synchrotron X-ray and neutron experiments were made at BL22XU, SPring-8 and a total scattering device, NOVA, J-PARC. Deuterium atoms, which occupy the tetrahedral sites of the fcc metal lattice in LaD
undergoes phase separation at high pressure with the relocation of deuterium atoms in the interstitial sites of La metal lattice. Synchrotron X-ray and neutron experiments were made at BL22XU, SPring-8 and a total scattering device, NOVA, J-PARC. Deuterium atoms, which occupy the tetrahedral sites of the fcc metal lattice in LaD , move into the empty octahedral sites at 11 GPa to form LaD and LaD
, move into the empty octahedral sites at 11 GPa to form LaD and LaD both having fcc metal lattices. Mono-hydride with an NaCl structure, which is common for mono-hydrides of transition metals, is formed in rare-earth metals for the first time. The first-principle calculations showed that LaH
both having fcc metal lattices. Mono-hydride with an NaCl structure, which is common for mono-hydrides of transition metals, is formed in rare-earth metals for the first time. The first-principle calculations showed that LaH is stable at low pressure and it undergoes a phase separation into LaH and LaH
is stable at low pressure and it undergoes a phase separation into LaH and LaH at 10 GPa, which is excellent agreement with the experimental results.
at 10 GPa, which is excellent agreement with the experimental results.
Kawakita, Yukinobu; Otomo, Toshiya*; Ueno, Hiroki; Tsubota, Masami*; Kumara, L. S. R.*; Oshita, Hidetoshi*; Suzuya, Kentaro
no journal, ,
Ag (GeSe
(GeSe )
) ternary alloy exhibits an extensive compositional range of bulk glass forming and a rapid enhancement in ionic conductivity from 10
ternary alloy exhibits an extensive compositional range of bulk glass forming and a rapid enhancement in ionic conductivity from 10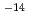 to 10
to 10 S/cm of these glasses at Ag concentration of x=0.3 with increasing Ag content. Structural models of this material have already been proposed by reverse Monte Carlo simulation based on experimental structural data of neutron and X-ray diffractions and X-ray absorption fine structure. However, there observed micro-phase separation tendency in these glasses. This tendency might relate to formation of ionic conduction path in supercooled liquid on quenching process. The proposed models will be examined by structure factors over a wide Q range including relatively low momentum-transfer region which have been newly obtained by the total neutron scattering instrument installed at BL21 in MLF, J-PARC. This work was supported by NEDO under "Advanced Fundamental Research Project on Hydrogen Storage Materials".
S/cm of these glasses at Ag concentration of x=0.3 with increasing Ag content. Structural models of this material have already been proposed by reverse Monte Carlo simulation based on experimental structural data of neutron and X-ray diffractions and X-ray absorption fine structure. However, there observed micro-phase separation tendency in these glasses. This tendency might relate to formation of ionic conduction path in supercooled liquid on quenching process. The proposed models will be examined by structure factors over a wide Q range including relatively low momentum-transfer region which have been newly obtained by the total neutron scattering instrument installed at BL21 in MLF, J-PARC. This work was supported by NEDO under "Advanced Fundamental Research Project on Hydrogen Storage Materials".