Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kofuji, Hirohide; Amamoto, Ippei; Sasaki, Kazuya*; Yasumoto, Masaru*; Takasaki, Yasushi*; Myochin, Munetaka; Terai, Takayuki*
Denki Kagaku Oyobi Kogyo Butsuri Kagaku, 77(8), p.597 - 600, 2009/08
Times Cited Count:0 Percentile:0.01(Electrochemistry)The process flow of the phosphate conversion technique has been developed for the reduction of high-level radioactive waste (HLW) generated from the metal-electrorefining process. In this study, the results of thermodynamic calculations for the phosphate conversion reactions were examined by the basic experiments. The chlorides of rare earth elements (REE) turned out to be converted into phosphates easily. Furthermore, as the additive for the phosphate conversion reaction, high temperature behavior of lithium phosphate was evaluated to elucidate the thermodynamic property.
Kofuji, Hirohide; Amamoto, Ippei; Yasumoto, Masaru*; Sasaki, Kazuya*; Myochin, Munetaka; Terai, Takayuki*
Journal of Nuclear Materials, 389(1), p.173 - 178, 2009/05
Times Cited Count:4 Percentile:30.43(Materials Science, Multidisciplinary)Adaptation of the phosphate conversion technique was undertaken and evaluated for application to the recycle process of the spent electrolyte generated from metal electrorefining process which is a kind of pyrochemical reprocessing technologies. The conceptual flow sheet has been discussed based on the thermodynamic properties of constituent. However few data have been published relating to the phosphate, therefore, the thermodynamic data were attempted to be obtained by the calorimetry and vapor pressure measurements. The measurements have been started with pure substance such as lithium phosphate. Through the measurements of HTMS and TG-DTA, it was clarified that decomposition or phase transition of Li PO
PO occurred at specific temperatures below the melting point of Li
occurred at specific temperatures below the melting point of Li PO
PO . To avoid the influence of fragment substances which have high vapour pressures, improved method of the HTMS measurement was examined.
. To avoid the influence of fragment substances which have high vapour pressures, improved method of the HTMS measurement was examined.
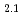 TiO
TiO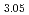 in hydrogen atmosphere
in hydrogen atmosphereHoshino, Tsuyoshi; Yasumoto, Masaru*; Tsuchiya, Kunihiko; Hayashi, Kimio; Nishimura, Hidetoshi*; Suzuki, Akihiro*; Terai, Takayuki*
Fusion Engineering and Design, 82(15-24), p.2269 - 2273, 2007/10
Times Cited Count:53 Percentile:94.94(Nuclear Science & Technology)no abstracts in English
 TiO
TiO heated at high temperature under various conditions
heated at high temperature under various conditionsHoshino, Tsuyoshi; Yasumoto, Masaru*; Tsuchiya, Kunihiko; Hayashi, Kimio; Nishimura, Hidetoshi*; Suzuki, Akihiro*; Terai, Takayuki*
Fusion Engineering and Design, 81(1-7), p.555 - 559, 2006/02
Times Cited Count:19 Percentile:76.11(Nuclear Science & Technology)no abstracts in English
*; J.Huang*; *; *; *; Yamaguchi, Kenji*; Sugimoto, Jun
JAERI-Tech 98-003, 32 Pages, 1998/02
no abstracts in English
 and BaUO
and BaUO in atmospheres simulating accident conditions
in atmospheres simulating accident conditionsHuang, J.*; Yamawaki, Michio*; Yamaguchi, Kenji*; Yasumoto, Masaru*; Sakurai, Hiroshi*; Suzuki, Yasufumi
Journal of Nuclear Materials, 248, p.257 - 261, 1997/09
no abstracts in English
 and BaUO
and BaUO in atmospheres simulating accident conditions
in atmospheres simulating accident conditionsHuang, J.*; *; Yamaguchi, Kenji*; *; *;
Journal of Nuclear Materials, 248, p.257 - 261, 1997/00
Times Cited Count:6 Percentile:47.9(Materials Science, Multidisciplinary)no abstracts in English
 by means of mass spectrometry
by means of mass spectrometry*; Huang, J.*; Yamaguchi, Kenji*; *; Sakurai, Hiroshi*;
Journal of Nuclear Materials, 231, p.199 - 203, 1996/00
Times Cited Count:11 Percentile:68.04(Materials Science, Multidisciplinary)no abstracts in English
 TiO
TiO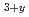
Hoshino, Tsuyoshi; Tsuchiya, Kunihiko; Hayashi, Kimio; Yasumoto, Masaru*; Nishimura, Hidetoshi*; Suzuki, Akihiro*; Terai, Takayuki*
no journal, ,
no abstracts in English
Kofuji, Hirohide; Amamoto, Ippei; Myochin, Munetaka; Terai, Takayuki*; Yasumoto, Masaru*; Wibowo, N.*
no journal, ,
no abstracts in English
Kofuji, Hirohide; Amamoto, Ippei; Yasumoto, Masaru*; Wibowo, N.*; Myochin, Munetaka; Terai, Takayuki*
no journal, ,
Adaptation of the phosphate conversion technique has been evaluated to recycle the spent electrolyte generated from metal electrorefining process which is a kind of pyrochemical reprocessing technologies. In the process, accumulated impurities such as fission products are precipitated as phosphate by adding lithium phosphate and separated from the molten salt. The conceptual flow sheet has been discussed based on the thermodynamic property of constituent. However few data have been published relating to the phosphate, they were attempted to be derived from the results of calorimetry and vapor pressure measurement. The measurements have been started with pure substance such as lithium phosphate, so that the current situation of the experiments will be introduced in this report.
Kofuji, Hirohide; Amamoto, Ippei; Yasumoto, Masaru*; Sasaki, Kazuya*; Wibowo, N.*; Myochin, Munetaka; Terai, Takayuki*
no journal, ,
Adaptation of the phosphate conversion technique has been evaluated to recycle the spent electrolyte generated from metal electrorefining process which is a kind of pyrochemical reprocessing technologies. In the process, accumulated impurities such as fission products are precipitated as phosphate by adding lithium phosphate and separated from the molten salt. The conceptual flow sheet has been discussed based on the thermodynamic property of constituent. However few data have been published relating to the phosphate, they were attempted to be derived from the results of calorimetry and vapor pressure measurement. The measurements have been started with pure substance such as lithium phosphate, so that the current situation of the experiments will be introduced in this report.
Kofuji, Hirohide; Amamoto, Ippei; Yasumoto, Masaru*; Sasaki, Kazuya*; Myochin, Munetaka; Terai, Takayuki*
no journal, ,
Adaptation of the phosphate conversion technique was undertaken and evaluated for application to the recycle process of the spent electrolyte generated from metal electrorefining process. To evaluate phosphate conversion reactions, fundamental experiments and estimation of thermodynamic behaviour of phosphate for the equilibrium calculation were carried out.
Kofuji, Hirohide; Amamoto, Ippei; Yasumoto, Masaru*; Takasaki, Yasushi*; Sasaki, Kazuya*; Myochin, Munetaka; Terai, Takayuki*
no journal, ,
Phosphate conversion process for recycling spent electrolyte originated from pyrochemical reprocessing has been studied. Feasibility of the process has been evaluated thermodynamically based on basic experiments. Current situation of phosphate conversion experiments and the estimations of thermodynamic properties will be introduced in this presentation.
Kofuji, Hirohide; Amamoto, Ippei; Sasaki, Kazuya*; Yasumoto, Masaru*; Takasaki, Yasushi*; Myochin, Munetaka; Terai, Takayuki*
no journal, ,
Phosphate conversion process for recycling spent electrolyte originated from pyrochemical reprocessing has been studied. Feasibility of the process has been evaluated thermodynamically based on basic experiments. Current situation of phosphate conversion experiments and the estimations of thermodynamic properties will be introduced in this presentation.
Kofuji, Hirohide; Sasaki, Kazuya*; Amamoto, Ippei; Sawada, Nobuyuki*; Yasumoto, Masaru*; Myochin, Munetaka; Terai, Takayuki*
no journal, ,
Adaptation of the phosphate conversion technique has been evaluated to recycle the spent electrolyte generated from metal electrorefining process which is a kind of pyrochemical reprocessing technologies. The accumulated impurities such as fission products are precipitated as phosphate by adding lithium phosphate and separated from the molten salt in the process. To discuss the conversion reaction, some thermodynamic properties are required however few data have been published relating to the phosphate, they were attempted to be derived from the results of thermal analysis. The pyrochemical behavior and estimation of heat capacity of lanthanum phosphate and lithium phosphate will be introduced in this report.