Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Liu, J.; Miyahara, Naoya; Miwa, Shuhei; Takano, Masahide; Hidaka, Akihide; Osaka, Masahiko
Journal of Nuclear Materials, 527, p.151819_1 - 151819_7, 2019/12
Times Cited Count:2 Percentile:21.58(Materials Science, Multidisciplinary)To evaluate the effect of each constituent element on the evaporation rate of ruthenium (Ru) from fission-produced alloy precipitates, the oxidation and evaporation behaviors of metallic Ru, molybdenum (Mo), palladium (Pd), rhodium (Rh) and Mo-Ru-Pd-Rh alloy powders were investigated by thermogravimetric analysis under oxidizing atmospheres from 1473 to 1723 K. The findings led to the following conclusions: (1) The quick oxidation of Mo into condensed Mo oxides can effectively suppress the oxidation and evaporation of Ru in alloy powders; (2) After the complete evaporation of Mo, the evaporation loss rate of Ru would be directly influenced by the Ru activity in the Ru-Pd-Rh alloys, which is determined by the composition of alloys.
Kihara, Shinji; Amazawa, Hiroya; Sakai, Akihiro; Nakata, Hisakazu; Kugo, Teruhiko; Matsuda, Norihiro; Oizumi, Akito; Sasamoto, Hiroshi; Mitsui, Seiichiro; Miyahara, Kaname
JAEA-Research 2013-033, 320 Pages, 2014/07
JAEA performed decontamination experiments at two test sites that combined a range of buildings and different types of land use, located in Date and Minami Soma municipalities as field pilot projects in order to accumulate knowledge and data for full-scale decontamination activities performed by local governments. In the pilot projects, we established its plan using practical decontamination methods that can be easily implemented, according to decontamination targets (e.g., forests, agricultural land, residential house and roads) at each site. As a result of the decontamination, the average air dose rates were reduced to approximately one half of the values before decontamination.
Umeda, Koji; Oi, Takao; Osawa, Hideaki; Oyama, Takuya; Oda, Chie; Kamei, Gento; Kuji, Masayoshi*; Kurosawa, Hideki; Kobayashi, Yasushi; Sasaki, Yasuo; et al.
JAEA-Review 2007-050, 82 Pages, 2007/12
This report shows the annual report which shows the summarized results and topic outline of each project on geological disposal technology in the fiscal year of 2006.
Saito, Yuji; Kimura, Hiroaki*; Suzuki, Yoshio*; Nakatani, Takeshi*; Matsushita, Tomohiro*; Muro, Takayuki*; Miyahara, Tsuneaki*; Fujisawa, M.*; Soda, Kazuo*; Ueda, Shigenori*; et al.
Nuclear Instruments and Methods in Physics Research A, 467-468(Part.1), p.553 - 556, 2001/07
Times Cited Count:23 Percentile:82.42(Instruments & Instrumentation)no abstracts in English
Saito, Yuji; Kimura, Hiroaki*; Suzuki, Yoshio*; Nakatani, Takeshi*; Matsushita, Tomohiro*; Muro, Takayuki*; Miyahara, Tsuneaki*; Fujisawa, M.*; Soda, Kazuo*; Ueda, Shigenori*; et al.
Review of Scientific Instruments, 71(9), p.3254 - 3259, 2000/09
Times Cited Count:187 Percentile:98.99(Instruments & Instrumentation)no abstracts in English
Sugimoto, Masayoshi; Jameson, R. A.*; V.Teplyakov*; D.Berwald*; B.Blind*; D.Bruhwiler*; H.Deitinghoff*; Ferdinand, R.*; Kinsho, Michikazu; Klein, H.*; et al.
Journal of Nuclear Materials, 258-263, p.367 - 371, 1998/00
Times Cited Count:0 Percentile:0.01(Materials Science, Multidisciplinary)no abstracts in English
Katsuta, Hiroji; Noda, Kenji; ; Sugimoto, Masayoshi; Maekawa, Hiroshi; Konishi, Satoshi; Nakamura, Hideo; *; Oyama, Yukio; Jitsukawa, Shiro; et al.
Nihon Genshiryoku Gakkai-Shi, 40(3), p.162 - 191, 1998/00
no abstracts in English
Niwa, Hajime; Kawata, Norio; Ieda, Yoshiaki; Sato, Ikken; Ohno, Shuji; Uto, Nariaki; Miyahara, Shinya; Kondo, Satoru; Kamide, Hideki; Yamaguchi, Akira; et al.
PNC TN9410 94-154, 317 Pages, 1995/03
None
Tanabe, Tetsuro*; Fujine, Michihiko*; Noguchi, Hiroshi*; Yagi, Yasufumi*; Hirano, Yoichi*; Shimizu, Hajime*; Akiba, Masato; Araki, Masanori; Kubota, Yusuke*; Miyahara, Akira*
Journal of Nuclear Materials, 200(1), p.120 - 127, 1993/03
Times Cited Count:9 Percentile:66.93(Materials Science, Multidisciplinary)no abstracts in English
Miyahara, Shinya; ; Himeno, Yoshiaki
Nuclear Technology, 97(2), p.212 - 226, 1992/02
Times Cited Count:8 Percentile:61.53(Nuclear Science & Technology)None
Miyahara, Shinya; ; Watanabe, Tomoo; Haga, Kazuo; Himeno, Yoshiaki
Nuclear Technology, 97(2), p.177 - 185, 1992/02
Times Cited Count:7 Percentile:57.46(Nuclear Science & Technology)None
*; Shimada, Taihei; Kabasawa, M.*; Harami, Taikan; Yonehara, Hiroto; *; *;
JAERI-M 91-045, 110 Pages, 1991/03
no abstracts in English
Haga, Kazuo; ; Watanabe, Tomoo; ; Himeno, Yoshiaki
PNC TN9410 91-091, 13 Pages, 1991/01
A series of tests has been conducted to obtain gas-liquid equilibrium partition coefficient Kd of volatile fission products such as cesium,iodine,and tellurium in sodium. In the test a sodium pool mixed with an FP simulant was heated by an electric furnace and the solvent of trapped vapors by filters was quantitatively analyzed. The results are,(1)Cs shows the highest Kd (20-100), (2)Kd of iodine scatters as wide as 0.02-0.5at 450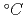 and 0.3-0.8 at 650
and 0.3-0.8 at 650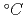 ,(3)the Kd values of Cs and I agree well with the theoretical ones reported by Castleman et al., and (4)if a sodium-telluride which is hard to vaporize than pure Te is assumed, measured Kd of Te agrees with that theoretical.
,(3)the Kd values of Cs and I agree well with the theoretical ones reported by Castleman et al., and (4)if a sodium-telluride which is hard to vaporize than pure Te is assumed, measured Kd of Te agrees with that theoretical.
Miyahara, Shinya; Haga, Kazuo; Himeno, Yoshiaki
PNC TN9410 91-027, 16 Pages, 1991/01
A series of tests was conducted to studythe mechanical release of non-volatile fission products during sodium-concrete reaction,in which hydrodynamic break-up by the hydrogen bubble bursting is predominant at the sodium pool surface. In thetests,non-radioactive materials,namely strontium oxide,europium oxide,and ruthenium particles whose sizes ranged from some microns to several tens of microns,were used as simulated non-volatile fission products. The following results were obtained from the present study: (1) The sodium aerosol release rate during the sodium-concrete reaction was larger than that of natural evaporation. The difference,however,became smaller with the increase in the sodium temperature; nearly 10 times at 400 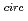 C and 3 times at 700
C and 3 times at 700 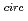 C. (2) Retention factors of these non-volatile materials in sodium pool increased inthe range of 0.5 to the fourth power of10 with the increase in the sodium temperature from 400
C. (2) Retention factors of these non-volatile materials in sodium pool increased inthe range of 0.5 to the fourth power of10 with the increase in the sodium temperature from 400 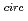 C to 700
C to 700 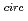 C
C
Sasaki, Noriaki; Yusa, Yasuhisa; Yamada, Kazuo; Nodaka, Masayuki*; *; Kawamura, Kazuhiro; Miyahara, Kaname; Arai, Takashi; Kamei, Gento; Hirose, Ikuro; et al.
PNC TN8440 88-018, 170 Pages, 1988/12
None
Himeno, Yoshiaki; ; Morii, Tadashi*; *
PNC TN9410 88-145, 11 Pages, 1988/10
Using sodium from 180kg to 3 metric tons, a series of tests has been conducted to develop the fire mitigation system and to study a design basis sodium leak accident and its consequences in the auxiliary building of the fast breeder reactor. In the test, flow pattern of a realistic leak from the sodium piping was investigated at first. Combustions of sodium in an open pool and a pool with a reduced opening were also studied together with combustion and flow of a sodium on an inclined steel liner and in a drain piping. Transient thermal conduction of a steel lined floor concrete during a sodium leak was tested and evaluated. In next, based on the results obtained, a fire mitigation system was developed and was mounted in a two storied concrete test rig. Then, a large scale test starting from a leak and ending by a self-extinguishment of a fire in the smothering tank was carried out. In final, consequences of sodium aerosols deposition on the reactor components and the electrical instruments have been experimentally studied.
; Matsuki, Takuo*; Hiroi, Hiroshi*; Himeno, Yoshiaki
PNC TN9410 88-092, 82 Pages, 1988/08
Post-test calculations of the large-scale sodium leak demonstration test, Run-D2, were performed using the ASSCOPS code in order to validate the applicability of the code to the evaluation of the fire suppression function of the PNC-type smothering tank. In the analysis, radiation coefficients between aerosols in the gas phase and structures and a pool surface in the smothering tank defined in the code were varied as parameter. The following three cases were calculated. (a)No aerosols suspending was assumed in the gas phase and thus, 1.0 was used as the coefficient between the structures and the pool surface. (b)The coefficient between the aerosols and the pool surface was 0.65, and that between structures and the aerosols was 0.73. (c)The coefficient between the aerosols and the pool surface was the same as case (b) and 0.5 was used as that between structures and the aerosols. The comparison between analysis and experiment with regard to the temperature of the various parts showed that the agreement was within +30% and -20%. From these results, it was concluded that the code was available for the evaluation of the fire suppression function of the smothering tank.
Himeno, Yoshiaki; ; Morii, Tadashi*
PNC TN9410 88-094, 18 Pages, 1988/05
Flow pattern of a realistic sodium leak from the sodium piping equipped with jackets and thermal insulator was experimentally investigated. Then, based on this result, the fire mitigation system consisting of an inclined liner, a drain piping, and a smothering tank has been developed. The performance of the system was, in final, validated in the large-scale sodium leak and fire test in the SAPFIRE facility.
Kawada, Koji*; ; Hiroi, Hiroshi*; Himeno, Yoshiaki
PNC TN9410 88-004, 44 Pages, 1988/01
Since sodium leak and fire researches have been performed at high-temperatures simulating an accident during the rated reactor operation knowledge of sodium leak and fire at low-temperatures which may happen at Ex-vessel Storage Tank or during the partial power operation of the reactor was very li,ited. Therefore, the present test was carried out to clarify the ignition temperatures and the temperature at which aerosol starts to release during sodium spray, column, and pool fires. Sodium spray and column tests were conducted at Sodiu Fire Test Rig (SOFT-1), while sodium pool test was carried out at Sodium Leak Fire and Aerosol Test Rig (SOLFA-1). The following test results were obtained from these tests. (1)Sodium Spray Test. The ignition temperature was 160 C depending upon the droplet diameter of sprayed sodium. (2)Sodium Column Test. (a)Sodium main flow did not ignite, while the scattered sodium droplets ignited. (b)The ignition temperature of the main flow rebounded on a pan was 180
C depending upon the droplet diameter of sprayed sodium. (2)Sodium Column Test. (a)Sodium main flow did not ignite, while the scattered sodium droplets ignited. (b)The ignition temperature of the main flow rebounded on a pan was 180 C. (c)The ignition temperature of deposits on a pan was 160
C. (c)The ignition temperature of deposits on a pan was 160  C. (3)Sodium Pool Test. (a)The ignition temperature of the static pool ranged from 280 to 315
C. (3)Sodium Pool Test. (a)The ignition temperature of the static pool ranged from 280 to 315 C. (b)Temperature at which aerosol starts to release was determined to be 140 to 160
C. (b)Temperature at which aerosol starts to release was determined to be 140 to 160 C by visual in spection. (c)After extinguished artificially by closing a lid, sodium reignited at temperatures higher than 80
C by visual in spection. (c)After extinguished artificially by closing a lid, sodium reignited at temperatures higher than 80  C when the lid was reopend.
C when the lid was reopend.
F.Beonio-Brocchieri*; H.Bunz*; W.Scholck*; I.H.Dunbar*; J.Gauvain*; *; *; Soda, Kunihisa;
Nuclear Technology, 81, p.193 - 204, 1988/00
Times Cited Count:10 Percentile:70.71(Nuclear Science & Technology)no abstracts in English