Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Strasser, P.*; Abe, Mitsushi*; Aoki, Masaharu*; Choi, S.*; Fukao, Yoshinori*; Higashi, Yoshitaka*; Higuchi, Takashi*; Iinuma, Hiromi*; Ikedo, Yutaka*; Ishida, Katsuhiko*; et al.
EPJ Web of Conferences, 198, p.00003_1 - 00003_8, 2019/01
Times Cited Count:13 Percentile:98.93(Quantum Science & Technology)Nagai, Takayuki; Kobayashi, Hidekazu; Shimamura, Keisuke; Oyama, Koichi; Sasage, Kenichi; Okamoto, Yoshihiro; Shiwaku, Hideaki; Yamanaka, Keisuke*; Ota, Toshiaki*
JAEA-Research 2018-005, 72 Pages, 2018/09
Addition of radioactive waste to a borosilicate glass frit affects the local structures of glass-forming elements and waste elements in a waste glass produced in a vitrification process. In this study, simulated waste glass samples were prepared from borosilicate glass frit including phosphorus pentaoxide, and we investigated local structures of sodium (Na), boron (B), and waste elements in these glass samples by using synchrotron XAFS measurements in soft and hard X ray region.
Muta, Hiroaki*; Nishikane, Ryoji*; Ando, Yusuke*; Matsunaga, Junji*; Sakamoto, Kan*; Harjo, S.; Kawasaki, Takuro; Oishi, Yuji*; Kurosaki, Ken*; Yamanaka, Shinsuke*
Journal of Nuclear Materials, 500, p.145 - 152, 2018/03
Times Cited Count:13 Percentile:76.73(Materials Science, Multidisciplinary)Okamoto, Yoshihiro; Nagai, Takayuki; Kaneko, Koji; Motokawa, Ryuhei; Haga, Yoshinori; Yamanaka, Keisuke*; Ota, Toshiaki*
2017-Nendo Seika Kokaigata Riyo Seika Hokokusho (Ritsumeikan Daigaku SR Senta), p.33 - 36, 2018/00
The high-level radioactive waste solution which generated from a spent nuclear fuel reprocessing process are mixed with a borosilicate glass frit in a vitrification process, and a chemical stable glass solidified body is produced. To understand local structure of light elements included in a glass solidified body, we carried out a B K-edge XANES measurement of raw glass frits and simulated radioactive waste glasses. As a result of this estimation of boron local structure in glass samples, it was found that the sp type boron (BO
type boron (BO ) percentage increases and the sp
) percentage increases and the sp type (BO
type (BO ) percentage by increasing the sodium content in glass frits.
) percentage by increasing the sodium content in glass frits.
Nagai, Takayuki; Kobayashi, Hidekazu; Sasage, Kenichi; Ayame, Yasuo; Okamoto, Yoshihiro; Shiwaku, Hideaki; Yamanaka, Keisuke*; Ota, Toshiaki*
JAEA-Research 2017-005, 54 Pages, 2017/06
Addition of radioactive waste to a borosilicate frit affects the local structures of boron (B) and waste elements in a waste glass. Synchrotron XAFS measurement was applied to investigate the local structural changes by using simulated waste borosilicate glass samples. Following results were obtained by the B K-edge XAFS analysis. It was confirmed that B K-edge XAFS analysis enables us to discriminate sp type boron (BO
type boron (BO ) from sp
) from sp type boron (BO
type boron (BO ). Addition of waste elements to a glass frit increases the percentage of BO
). Addition of waste elements to a glass frit increases the percentage of BO and decreases that of BO
and decreases that of BO . By decreasing the SiO
. By decreasing the SiO /Al
/Al O
O ratio or increasing the (SiO
ratio or increasing the (SiO +B
+B O
O )/Al
)/Al O
O ratio in the glass composition, the BO
ratio in the glass composition, the BO percentage increases and the BO
percentage increases and the BO percentage decreases. Addition of P
percentage decreases. Addition of P O
O decreases the BO
decreases the BO percentage and increases the BO
percentage and increases the BO percentage. Following results were obtained from XAFS measurement of the waste elements. Cerium (Ce) valence is more reduced with the increase of the B
percentage. Following results were obtained from XAFS measurement of the waste elements. Cerium (Ce) valence is more reduced with the increase of the B O
O content. Addition of P
content. Addition of P O
O has a tendency to reduce the Ce valence and to enhance deposition of Zr oxide. Deposition of ruthenium compounds separated from glass phase can not be improved by changing the B
has a tendency to reduce the Ce valence and to enhance deposition of Zr oxide. Deposition of ruthenium compounds separated from glass phase can not be improved by changing the B O
O content. This study was performed as a part of the project, "Improvement of vitrification process of high-level radioactive liquid wastes" on the foundation business of the Agency for Natural Resources and Energy.
content. This study was performed as a part of the project, "Improvement of vitrification process of high-level radioactive liquid wastes" on the foundation business of the Agency for Natural Resources and Energy.
Serizawa, Hiroyuki; Matsunaga, Junji*; Haga, Yoshinori; Nakajima, Kunihisa; Akabori, Mitsuo; Tsuru, Tomohito; Kaji, Yoshiyuki; Kashibe, Shinji*; Oishi, Yuji*; Yamanaka, Shinsuke*
Crystal Growth & Design, 13(7), p.2815 - 2823, 2013/07
Times Cited Count:5 Percentile:42.57(Chemistry, Multidisciplinary)Since the shape of the negative crystal closely relates to the morphology of the crystal habits, the formation and the growth mechanism is important subject in a field of the physical science. Whereas, the negative crystal formed in a large single crystal mass has been arousing interest as an expensive jewelry because of its mysterious appearance and rarity. However, it is difficult to control the shape of this polyhedral cavity embedded in a solid medium arbitrary. Here we report the recent discovery on the growth process of the negative crystal. We found that precipitated helium forms the negative crystal in UO ; the shape changes drastically with the condition of the helium precipitation. The transformation mechanism was discussed in this article. Our investigation implies that the shape of the negative crystal can be arbitrary controlled by controlling the precipitation condition.
; the shape changes drastically with the condition of the helium precipitation. The transformation mechanism was discussed in this article. Our investigation implies that the shape of the negative crystal can be arbitrary controlled by controlling the precipitation condition.
Fukuyama, Eriko*; Mine, Makoto*; Okumura, Masahiko; Sunaga, Tomoka*; Yamanaka, Yoshiya*
Physical Review A, 76(4), p.043608_1 - 043608_11, 2007/10
Times Cited Count:6 Percentile:33.51(Optics)We consider a trapped Bose-Einstein condensate (BEC) with a highly quantized vortex. For the BEC with a doubly, triply or quadruply quantized vortex, the numerical calculations have shown that the Bogoliubov-de Gennes equations, which describe the fluctuation of the condensate, have complex eigenvalues. In this paper, we obtain the analytic expression of the condition for the existence of complex modes, using the method developed by Rossignoli and Kowalski for the small coupling constant. To derive it, we make the two-mode approximation. With the derived analytic formula, we can identify the quantum numbers of the complex modes for each winding number of the vortex. Our result is consistent with those obtained by the numerical calculation in the case that the winding number is two, three or four. We prove that the complex modes always exist when the condensate has a highly quantized vortex.
Shimada, Yoshinori*; Nishimura, Hiroaki*; Nakai, Mitsuo*; Hashimoto, Kazuhisa*; Yamaura, Michiteru*; Tao, Y.*; Shigemori, Keisuke*; Okuno, Tomoharu*; Nishihara, Katsunobu*; Kawamura, Toru*; et al.
Applied Physics Letters, 86(5), p.051501_1 - 051501_3, 2005/01
Times Cited Count:113 Percentile:94.31(Physics, Applied)no abstracts in English
Kuroda, Masatoshi*; Yamanaka, Shinsuke*; Nagase, Fumihisa; Uetsuka, Hiroshi
Nuclear Engineering and Design, 203(2-3), p.185 - 194, 2001/01
Times Cited Count:14 Percentile:68.91(Nuclear Science & Technology)no abstracts in English
Kuroda, Masatoshi*; Yoshioka, Kunihiko*; Yamanaka, Shinsuke*; Anada, Hiroyuki*; Nagase, Fumihisa; Uetsuka, Hiroshi
Journal of Nuclear Science and Technology, 37(8), p.670 - 675, 2000/08
no abstracts in English
*; *; *; *; *; Kobayashi, Yasuhiko; Watanabe, Hiroshi; *; Akashi, Makoto*; *; et al.
J. Gen. Appl. Microbiol., 43, p.175 - 177, 1997/00
Times Cited Count:11 Percentile:37.63(Biotechnology & Applied Microbiology)no abstracts in English
Mano, Tadashi; Yoshikawa, Hideki; Fukunaga, Sakae*; Yui, Mikazu; Yamagata, Junji*; Asano, Hidekazu*; Yamanaka, Yumiko*
PNC TN8410 94-117, 60 Pages, 1993/03
None
*; Yoshida, Masaru; *; Asano, Masaharu; Kumakura, Minoru*; Mashimo, Toru*; Yamanaka, Hidetoshi*; *
Int. J. Pharm., 81, p.31 - 38, 1992/00
Times Cited Count:9 Percentile:41.18(Pharmacology & Pharmacy)no abstracts in English
 -caprolactone/
-caprolactone/ -valerolactone) and estracyt or estramustine
-valerolactone) and estracyt or estramustine*; Yoshida, Masaru; *; Asano, Masaharu; Kumakura, Minoru*; Mashimo, Toru*; Yamanaka, Hidetoshi*; *
Chemical and Pharmaceutical Bulletin, 39(8), p.2096 - 2099, 1991/00
no abstracts in English
 -caprolactone-co-
-caprolactone-co- -valerolactone) and estramustine
-valerolactone) and estramustine*; Yoshida, Masaru; *; Asano, Masaharu; Kumakura, Minoru; Mashimo, Toru*; Yamanaka, Hidetoshi*; *
Int. J. Pharm., 68, p.87 - 95, 1991/00
no abstracts in English
*; Yoshida, Masaru; Asano, Masaharu; Kumakura, Minoru; *; *; Mashimo, Toru*; Yuasa, Hisako*; Imai, K.*; Yamanaka, Hidetoshi*
European Polymer Journal, 26(12), p.1273 - 1277, 1990/00
Times Cited Count:34 Percentile:81.07(Polymer Science)no abstracts in English
Mine, Makoto*; Okumura, Masahiko; Sunaga, Tomoka*; Yamanaka, Yoshiya*
no journal, ,
no abstracts in English
Sunaga, Tomoka*; Fukuyama, Eriko*; Mine, Makoto*; Okumura, Masahiko; Yamanaka, Yoshiya*
no journal, ,
no abstracts in English
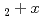
Matsunaga, Junji*; Kashibe, Shinji*; Serizawa, Hiroyuki; Nakajima, Kunihisa; Iwai, Takashi; Haga, Yoshinori; Oishi, Yuji*; Yamanaka, Shinsuke*
no journal, ,
The aim of this study is to clarify the behavior of helium precipitated in UO . The micro-structure of UO
. The micro-structure of UO matrix deformed by helium infusion was examined by FE-SEM and FE-TEM. It was deduced that the shape of the negative crystal is determined by the balance between the surface energy of the lattice plane and the inner pressure of helium.
matrix deformed by helium infusion was examined by FE-SEM and FE-TEM. It was deduced that the shape of the negative crystal is determined by the balance between the surface energy of the lattice plane and the inner pressure of helium.

Matsunaga, Junji*; Kashibe, Shinji*; Serizawa, Hiroyuki; Nakajima, Kunihisa; Iwai, Takashi; Haga, Yoshinori; Oishi, Yuji*; Yamanaka, Shinsuke*
no journal, ,
Helium generated in MOX fuel increases inner pressure of fuel rod and helium could also form additional bubbles in fuel pellet by the combination of radiation defects and high temperature. Therefore it is important to understand the behavior of helium in oxide fuel for reliable operation of MOX fuels. In the present study, the helium infusion treatments in high temperature and high pressure of helium were performed for both sintered polycrystalline UO fragments and hyperstoichiometric monograin UO
fragments and hyperstoichiometric monograin UO particles fabricated by the transportation method. It was suggested that the difference in the composition of UO
particles fabricated by the transportation method. It was suggested that the difference in the composition of UO is closely related with the condition of the formation of the negative crystal.
is closely related with the condition of the formation of the negative crystal.