Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
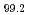 Zn
Zn Y
Y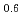 alloy using X-ray photoelectron spectroscopy
alloy using X-ray photoelectron spectroscopyMiyazaki, Hidetoshi*; Akatsuka, Tatsuyoshi*; Kimura, Koji*; Egusa, Daisuke*; Sato, Yohei*; Itakura, Mitsuhiro; Takagi, Yasumasa*; Yasui, Akira*; Ozawa, Kenichi*; Mase, Kazuhiko*; et al.
Materials Transactions, 64(6), p.1194 - 1198, 2023/06
Times Cited Count:1 Percentile:48.82(Materials Science, Multidisciplinary)We investigated the electronic structure of the Mg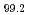 Zn
Zn Y
Y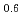 alloy using hard and soft X-ray photoemission spectroscopy and electronic band structure calculations to understand the mechanism of the phase stability of this material. Electronic structure of the Mg
alloy using hard and soft X-ray photoemission spectroscopy and electronic band structure calculations to understand the mechanism of the phase stability of this material. Electronic structure of the Mg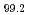 Zn
Zn Y
Y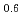 alloy showed a semi-metallic electronic structure with a pseudo-gap at the Fermi level. The observed electronic structure of the Mg
alloy showed a semi-metallic electronic structure with a pseudo-gap at the Fermi level. The observed electronic structure of the Mg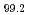 Zn
Zn Y
Y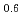 alloy suggests that the presence of a pseudogap structure is responsible for phase stability.
alloy suggests that the presence of a pseudogap structure is responsible for phase stability.
Sato, Yohei*; Egusa, Daisuke*; Miyazaki, Hidetoshi*; Kimura, Koji*; Itakura, Mitsuhiro; Terauchi, Masami*; Abe, Eiji*
Materials Transactions, 64(5), p.950 - 954, 2023/05
Times Cited Count:1 Percentile:48.82(Materials Science, Multidisciplinary)Dilute Mg-Zn-Y alloy with a mille-feuille structure (MFS) exhibits a mechanical strength comparable to Mg-Zn-Y alloy with long period stacking/ordered (LPSO) structure through kink deformation. In order to deepen understanding the thermal stability of the MFS-type Mg alloys, it is required to clarify the solute cluster structures composed of Zn and Y in solute enriched stacking faults (SESFs). In this study, electron energy-loss and energy dispersive X-ray spectroscopy based on scanning transmission electron microscopy (STEM-EELS/EDS) were conducted to investigate the electronic structure and composition of Zn and Y in the SESFs of the MFS-Mg alloy. Zn-L2,3 spectra indicated that the valence charges of Zn in the dilute Mg alloy were different from that of the LPSO-type Mg-Zn-Y alloy. In addition, the intensity ratio of L3/L2 in Y-L2,3 spectrum of the dilute MFS-Mg alloy was larger than that of the LPSO-Mg alloy, reflecting the electron occupancies of 4d3/2 and 4d5/2 orbitals of Y atoms were different from those of the LPSO-Mg alloys. STEM-EELS analysis of the SESF composition in the dilute MFS-Mg alloy indicated that the Zn/Y ratio should be lower than that of the LPSO-Mg alloy, which was confirmed also by STEM-EDS measurements. These results indicate that the cluster structure in the SESFs of the dilute MFS-Mg alloy should be different from the ideal Zn6Y8 cluster in the LPSO-type Mg-Zn-Y alloys.
Hosokawa, Shinya*; Kimura, Koji*; Yamasaki, Michiaki*; Kawamura, Yoshihito*; Yoshida, Koji*; Inui, Masanori*; Tsutsui, Satoshi*; Baron, A. Q. R.*; Kawakita, Yukinobu; Ito, Shinichi*
Journal of Alloys and Compounds, 695, p.426 - 432, 2017/02
Times Cited Count:3 Percentile:16.49(Chemistry, Physical) glasses
glassesStellhorn, J. R.*; Hosokawa, Shinya*; Pilgrim, W.-C.*; Kawakita, Yukinobu; Kamimura, Kenji*; Kimura, Koji*; Blanc, N.*; Boudet, N.*
Zeitschrift f r Physikalische Chemie, 230(3), p.369 - 386, 2016/03
r Physikalische Chemie, 230(3), p.369 - 386, 2016/03
Kitatsuji, Yoshihiro; Okugaki, Tomohiko*; Kasuno, Megumi*; Kubota, Hiroki*; Maeda, Koji*; Kimura, Takaumi; Yoshida, Zenko; Kihara, Sorin*
Journal of Chemical Thermodynamics, 43(6), p.844 - 851, 2011/06
Times Cited Count:8 Percentile:30.19(Thermodynamics)Standard Gibbs energies for transfer ( G
G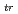
 ) of actinyl ions (AnO
) of actinyl ions (AnO
 ; z = 2 or 1; An: U, Np or Pu) between an aqueous solution and an organic solution were determined based on distribution method combined with voltammetry for ion transfer at the interface of two immiscible electrolyte solutions. The organic solutions examined were nitrobenzene, 1,2-dichloroethane, benzonitrile, acetophenone and 2-nitrophenyl octyl ether. Irrespective of the type of organic solutions,
; z = 2 or 1; An: U, Np or Pu) between an aqueous solution and an organic solution were determined based on distribution method combined with voltammetry for ion transfer at the interface of two immiscible electrolyte solutions. The organic solutions examined were nitrobenzene, 1,2-dichloroethane, benzonitrile, acetophenone and 2-nitrophenyl octyl ether. Irrespective of the type of organic solutions,  G
G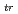
 of UO
of UO
 , NpO
, NpO
 and PuO
and PuO
 were nearly equal to each other and slightly larger than that of Mg
were nearly equal to each other and slightly larger than that of Mg . The
. The  G
G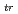
 of NpO
of NpO
 was extraordinary large compared with those of ordinary monovalent cations. The dependence of
was extraordinary large compared with those of ordinary monovalent cations. The dependence of  G
G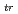
 of AnO
of AnO
 on the type of organic solutions was similar to that of H
on the type of organic solutions was similar to that of H or Mg
or Mg . The
. The  G
G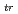
 of An
of An and An
and An were also discussed briefly.
were also discussed briefly.
 O
O
Fukunaga, Mamoru*; Sakamoto, Yuma*; Kimura, Hiroyuki*; Noda, Yukio*; Abe, Nobuyuki*; Taniguchi, Koji*; Arima, Takahisa*; Wakimoto, Shuichi; Takeda, Masayasu; Kakurai, Kazuhisa; et al.
Physical Review Letters, 103(7), p.077204_1 - 077204_4, 2009/08
Times Cited Count:50 Percentile:86.01(Physics, Multidisciplinary)Kitatsuji, Yoshihiro; Okugaki, Tomohiko*; Kasuno, Megumi*; Maeda, Koji*; Kimura, Takaumi; Yoshida, Zenko; Kihara, Sorin*
no journal, ,
no abstracts in English