Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kinuhata, Hiroshi*; Yamamoto, Masahiko; Taguchi, Shigeo; Surugaya, Naoki; Sato, Soichi; Kodama, Takashi*; Tamauchi, Yoshikazu*; Shibata, Yuki*; Anzai, Kiyoshi*; Matsuoka, Shingo*
Nuclear Technology, 192(2), p.155 - 159, 2015/11
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology)Experiments using a small-scale apparatus with 30 ml actual high-level liquid waste from the Tokai Reprocessing Plant were carried out to show that the hydrogen concentration in the gas phase reaches a steady-state value of much less than 4% (lower explosive limit) in the absence of sweeping-air. The H concentration reached a steady-state value as was expected and it was compared with a value predicted from an equation with parameters which had been obtained using the simulated solution. Satisfactory agreement showed that the Pd-ion catalytic H
concentration reached a steady-state value as was expected and it was compared with a value predicted from an equation with parameters which had been obtained using the simulated solution. Satisfactory agreement showed that the Pd-ion catalytic H consumption reaction previously found in the simulated solution proceeded equally well in the actual solution.
consumption reaction previously found in the simulated solution proceeded equally well in the actual solution.
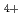 and N
and N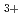 with electrons in atmospheric pressure nitrogen
with electrons in atmospheric pressure nitrogenIkezoe, Yasumasa; *; *
Chemical Physics Letters, 177(4-5), p.366 - 370, 1991/03
Times Cited Count:11 Percentile:43.53(Chemistry, Physical)no abstracts in English
 -CO-O
-CO-O mixture
mixtureIkezoe, Yasumasa; Shimizu, Saburo; Onuki, Kaoru; *; *; Tagawa, S.*; Tabata, Yoneho*
Journal of Physical Chemistry, 93(4), p.1193 - 1197, 1989/00
Times Cited Count:3 Percentile:19.38(Chemistry, Physical)no abstracts in English
 -O
-O -alkane(C
-alkane(C -C
-C ) mixtures studied by time-resolved atmospheric pressure ionization mass spectrometry
) mixtures studied by time-resolved atmospheric pressure ionization mass spectrometry*; Ikezoe, Yasumasa
J.Phys.Chem., 92(5), p.1126 - 1133, 1988/05
no abstracts in English
Ikezoe, Yasumasa; *; *; A.Viggiano*
Gas Phase Ion-Molecule Reaction Rate Constants Through 1986, 224 Pages, 1987/00
no abstracts in English
*;
Nihon Genshiryoku Gakkai-Shi, 28(8), p.701 - 709, 1986/00
Times Cited Count:1 Percentile:20.71(Nuclear Science & Technology)no abstracts in English
*; ;
UTNL-R-0190, p.8 - 16, 1986/00
no abstracts in English
 addition
additionIkezoe, Yasumasa; ; Shimizu, Saburo; Nakajima, Hayato; ; *
Radiation Physics and Chemistry, 26(4), p.445 - 449, 1985/00
no abstracts in English

 O
O (CO)
(CO) )
) (CO
(CO )
) in a CO
in a CO -CO-
-CO-
 O
O mixture
mixtureIkezoe, Yasumasa; ; Shimizu, Saburo; ; *; *
J.Phys.Chem., 88(24), p.5945 - 5948, 1984/00
no abstracts in English
Ikezoe, Yasumasa; *;
Shitsuryo Bunseki, 32(5), p.449R - 453R, 1984/00
no abstracts in English
Ikezoe, Yasumasa; ; Shimizu, Saburo; *
Radiation Physics and Chemistry, 17, p.69 - 70, 1981/00
no abstracts in English
*; *; *;
Canadian Journal of Chemistry, 52(14), p.2579 - 2589, 1974/00
Times Cited Count:4no abstracts in English
Kodama, Takashi*; Nakano, Masamichi*; Matsuoka, Shingo*; Matsuura, Chihiro*; Ito, Yasuo*; Kurosu, Katsuya*; Shiraishi, Hirotsugu; Katsumura, Yosuke*
no journal, ,
It has been known that although a considerable amount of hydrogen is produced radiolytically in the high level liquid waste, only small part is emitted into the gas phase when the liquid depth is large. We report here the results of an experimental study which shows that the liquid-depth effect is caused not by the reaction between hydrogen and radicals, as has been previously suggested, but by Pd-catalyzed reaction between hydrogen and nitric acid. The method for evaluating the magnitute of the effect is also proposed.
Kodama, Takashi*; Nakano, Masanao*; Hayashi, Yoshiaki*; Matsuoka, Shingo*; Ito, Yasuo*; Matsuura, Chihiro*; Shiraishi, Hirotsugu; Katsumura, Yosuke*
no journal, ,
We have reported in the previous meeting that the accumulation of radiolytically produced hydrogen in high-level liquid waste is suppressed owing to some oxidation reaction catalyzed by Pd in the solution. In this report, we present the results of a hydrogen bubbling experiment with mock high-level liquid waste, made to clarify the species involved in the reaction. Analysis was made on nitrous acid and nitric acid in the solution, and also on nitrogen oxide in the outlet gas. It was found that although the concentration of nitrous acid present in the solution is small, nitrogen oxide, consisting mostly of NO, is detected in the gas at concentration corresponding to the decrease of nitric acid in the solution. The amount of the produced nitrogen oxide matched roughly with an estimated amount of reacted hydrogen, confirming that nitric acid is the oxidant.
Kodama, Takashi*; Nakano, Masanao*; Hayashi, Yoshiaki*; Matsuoka, Shingo*; Ito, Yasuo*; Matsuura, Chihiro*; Shiraishi, Hirotsugu; Katsumura, Yosuke*
no journal, ,
We report here an evaluation for hydrogen concentration that may be attained in the upper space of the high-level liquid waste tank when the gas sweeping function happens to be lost. The parameters are the rate of radiolytic production of hydrogen, the rate of Pd-catalyzed hydrogen consuming reaction, and the solubility of hydrogen. For the first, a literature value on nitric acid solution was used, while for the second and third, experimental estimate was made in the present study with mock high-level liquid waste. All data have been taken at ambient temperature. For simplicity, it was assumed that both the gas and the liquid are homogeneous, and that the two phases are in equilibrium regarding hydrogen concentration. These led to an estimate of 0.6% for the hydrogen concentration in the gas phase, a value smaller than the lower limit of explosion.
Tashiro, Shinsuke; Matsumoto, Tetsuya; Kataoka, Osamu; Amano, Yuki; Abe, Hitoshi; Yamane, Yuichi; Yoshida, Kazuo; Ishikawa, Jun; Uchiyama, Gunzo; Ueda, Yoshinori*; et al.
no journal, ,
Measurements on the release ratios of aerial radioactive materials from mocked fuel reprocessing liquid waste under its boiling to dryness process were performed using labo-scaled experiments. Test sample, dissolved 27 elements into nitric acid and arranged to 2M acidity, was heated up to 300  C under the constant air ventilation. Steam, gaseous and airborne materials were collected or absorbed at the condenser, the air filter and the washing bottles. The accumulated release ratios of mocked FP elements from samples were determined using ICP-MS. From the accumulated release ratios determined from the condensed samples, the major release of Cs and Ru could be involved the release of mist and gaseous RuO
C under the constant air ventilation. Steam, gaseous and airborne materials were collected or absorbed at the condenser, the air filter and the washing bottles. The accumulated release ratios of mocked FP elements from samples were determined using ICP-MS. From the accumulated release ratios determined from the condensed samples, the major release of Cs and Ru could be involved the release of mist and gaseous RuO , respectively. Besides, accumulated release ratios of Ru was about 10 to 1000 times higher than Cs, differed from the literature using fuel reprocessing liquid waste. Its differences could be influenced the nitrous acid, which was reduced the generation of RuO
, respectively. Besides, accumulated release ratios of Ru was about 10 to 1000 times higher than Cs, differed from the literature using fuel reprocessing liquid waste. Its differences could be influenced the nitrous acid, which was reduced the generation of RuO .
.
Tomiyama, Masahiro; Yasuda, Takeshi; Tsutagi, Koichi; Yoshino, Yasuyuki; Shirato, Yoji; Nakamura, Yoshinobu; Kinuhata, Hiroshi*; Kodama, Takashi*; Nakano, Masanao*; Tamauchi, Yoshikazu*; et al.
no journal, ,
no abstracts in English
Kinuhata, Hiroshi*; Kodama, Takashi*; Nakano, Masanao*; Tamauchi, Yoshikazu*; Matsuoka, Shingo*; Tomiyama, Masahiro; Yasuda, Takeshi; Tsutagi, Koichi; Yoshino, Yasuyuki; Shirato, Yoji; et al.
no journal, ,
no abstracts in English
Tashiro, Shinsuke; Matsumoto, Tetsuya; Kataoka, Osamu; Amano, Yuki; Abe, Hitoshi; Yamane, Yuichi; Yoshida, Kazuo; Ishikawa, Jun; Uchiyama, Gunzo; Ueda, Yoshinori*; et al.
no journal, ,
The release behavior of radioactive materials from high-level radioactive liquid wastes (HLW) from reprocessing plants under an accidents of boiling to dryness of HLW condition has been studied. The influences of FP concentration in the simulated HLW on the release ratio of FP from the waste were measured in the laboratory-scaled experiments using non-radioactive simulated HLW which was prepared by dissolving 27 FP elements into nitric acid and adjusted to 2 M acidity. The simulated HLW was heated up to 300 C under the constant air ventilation condition. The accumulated release ratios of FP elements from samples were determined using ICP-MS analysis. It was found that the accumulated release ratio of Ru was decreased with the increase of the initial Ru concentration in the simulated HLW. However, those of Cs and Nd were not influenced by the initial concentrations of them.
C under the constant air ventilation condition. The accumulated release ratios of FP elements from samples were determined using ICP-MS analysis. It was found that the accumulated release ratio of Ru was decreased with the increase of the initial Ru concentration in the simulated HLW. However, those of Cs and Nd were not influenced by the initial concentrations of them.
Yamane, Yuichi; Amano, Yuki; Yanagida, Yoshinori; Kawasaki, Yasushi; Sato, Makoto; Hayasaka, Hiromi; Tashiro, Shinsuke; Abe, Hitoshi; Uchiyama, Gunzo; Ueda, Yoshinori*; et al.
no journal, ,
The release and transport characteristics of radioactive materials at a boiling accident of the high active liquid waste (HALW) in a reprocessing plant have been studied for improving experimental data of source terms of the boiling accident. This paper describes an experiment using a small test device having an electric furnace, in which 100 mL of the HALW was heated from room temperature to 300 C and the amount of materials released during heating was measured. The amount of materials as a function of its initial concentration in the HALW is reported.
C and the amount of materials released during heating was measured. The amount of materials as a function of its initial concentration in the HALW is reported.