Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Iwamoto, Chihiro*; Takamura, Masato*; Ueno, Kota*; Kataoka, Minami*; Kurihara, Ryo*; Xu, P. G.; Otake, Yoshie*
ISIJ International, 62(5), p.1013 - 1022, 2022/05
Times Cited Count:2 Percentile:30.25(Metallurgy & Metallurgical Engineering) -rays
-raysArisaka, Makoto; Watanabe, Masayuki; Ishizaki, Manabu*; Kurihara, Masato*; Chen, R.*; Tanaka, Hisashi*
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1543 - 1547, 2015/02
Times Cited Count:13 Percentile:72.94(Chemistry, Analytical)The influence of irradiation with  -rays to metal hexacyanoferrate (MHCF: M = Fe, Cu or Ni), which is known as an adsorbent for selective adsorption of cesium (Cs) ion in solution, on Cs adsorption ability and stability was investigated in HNO
-rays to metal hexacyanoferrate (MHCF: M = Fe, Cu or Ni), which is known as an adsorbent for selective adsorption of cesium (Cs) ion in solution, on Cs adsorption ability and stability was investigated in HNO solutions. Under the adsorbed dose conditions (50-300 kGy), it was found that the MHCF is fully stable although the radiolytic decomposition of MHCF was slightly observed with an increase of the total adsorbed dose, which was confirmed by an increment of Fe, Cu or Ni concentration in HNO
solutions. Under the adsorbed dose conditions (50-300 kGy), it was found that the MHCF is fully stable although the radiolytic decomposition of MHCF was slightly observed with an increase of the total adsorbed dose, which was confirmed by an increment of Fe, Cu or Ni concentration in HNO solution after the irradiation. The weight percent of the metal in the solution to initial weight of MHCF was less than unity. Moreover, no change in composition of carbon, hydrogen and nitrogen in MHCF was observed. On the other hand, the distribution coefficients of Cs to the irradiated MHCF were independent of the total adsorbed dose. This indicates that the Cs adsorption ability was maintained under
solution after the irradiation. The weight percent of the metal in the solution to initial weight of MHCF was less than unity. Moreover, no change in composition of carbon, hydrogen and nitrogen in MHCF was observed. On the other hand, the distribution coefficients of Cs to the irradiated MHCF were independent of the total adsorbed dose. This indicates that the Cs adsorption ability was maintained under  -ray irradiation.
-ray irradiation.
 adsorption
adsorption 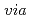 lattice defect sites of Prussian blue filled with coordination and crystallization water molecules
lattice defect sites of Prussian blue filled with coordination and crystallization water moleculesIshizaki, Manabu*; Akiba, Sae*; Otani, Asako*; Hoshi, Yuji*; Ono, Kenta*; Matsuba, Mayu*; Togashi, Takanari*; Kanaizuka, Katsuhiko*; Sakamoto, Masatomi*; Takahashi, Akira*; et al.
Dalton Transactions, 42(45), p.16049 - 16055, 2013/12
Times Cited Count:179 Percentile:99.58(Chemistry, Inorganic & Nuclear)We have revealed the fundamental mechanism of specific Cs adsorption into Prussian blue (PB) in order to develop high-performance PB-based Cs
adsorption into Prussian blue (PB) in order to develop high-performance PB-based Cs adsorbents in the wake of the Fukushima nuclear accident. We compared two types of PB nanoparticles with formulae of Fe
adsorbents in the wake of the Fukushima nuclear accident. We compared two types of PB nanoparticles with formulae of Fe
 [Fe
[Fe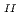 (CN)
(CN) ]3
]3 xH
xH O (x = 10-15) (PB-1) and (NH
O (x = 10-15) (PB-1) and (NH )0.70Fe
)0.70Fe 1.10[Fe
1.10[Fe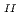 (CN)
(CN) ]
] 1.7H
1.7H O (PB-2) with respect to the Cs
O (PB-2) with respect to the Cs adsorption ability. The synthesised PB-1, by a common stoichiometric aqueous reaction between 4Fe
adsorption ability. The synthesised PB-1, by a common stoichiometric aqueous reaction between 4Fe and 3[Fe
and 3[Fe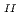 (CN)
(CN) ]
]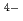 , showed much more efficient Cs
, showed much more efficient Cs adsorption ability than did the commercially available PB-2.
adsorption ability than did the commercially available PB-2.
Chen, R.*; Tanaka, Hisashi*; Kawamoto, Toru*; Asai, Miyuki*; Fukushima, Chikako*; Na, H.*; Kurihara, Masato*; Watanabe, Masayuki; Arisaka, Makoto; Nankawa, Takuya
Electrochimica Acta, 87, p.119 - 125, 2013/01
Times Cited Count:108 Percentile:94.84(Electrochemistry)A novel electrochemical adsorption system using a nanoparticle film of copper (II) hexacyanoferrate (III) was proposed for selectively removing cesium from wastewater. This system can be used for cesium separation without extra chemical reagents or any filtration treatment. Cesium uptake and elution can be simply controlled by switching the applied potentials between anodes and cathodes. Data from batch kinetic studies well fitted the intraparticle diffusion equation, reflecting a two-step process: a steepest ascent portion followed by a plateau extending to the equilibrium. The effective cesium removal with a high distribution coefficient (

 5
5 10
10 mL/g) can be adopted in a large pH range from 0.3 to 9.2, and in the presence of several diverse coexisting alkaline cations, suggesting it can be taken as a promising technology for actual nuclear wastewater treatment.
mL/g) can be adopted in a large pH range from 0.3 to 9.2, and in the presence of several diverse coexisting alkaline cations, suggesting it can be taken as a promising technology for actual nuclear wastewater treatment.
Chen, R.*; Tanaka, Hisashi*; Kawamoto, Toru*; Asai, Miyuki*; Fukushima, Chikako*; Kurihara, Masato*; Watanabe, Masayuki; Arisaka, Makoto; Nankawa, Takuya
Electrochemistry Communications, 25, p.23 - 25, 2012/11
Times Cited Count:50 Percentile:79.65(Electrochemistry)We first synthesized water-dispersed nanoparticle copper hexacyanoferrate (CuHCF) ink and then coated its nanoparticles on electrodes to electrochemically remove cesium from wastewater. Cesium uptake and elution can be controlled by switching the potentials between anodes and cathodes. Effective cesium removal can be adopted in a large pH range from 0.2 to 8.9, and in the presence of several diverse coexisting alkaline cations, suggesting that it can be taken as a promising technology for actual radioactive wastewater treatment. The prepared CuHCF nanoparticles can be simply and uniformly coated on electrodes by wet process like conventional printing methods, so any sizes or patterns are feasible at low cost, which indicated the potential as a promising sorption electrode of large size in the columns for sequential removal and recycle of Cs from wastewater.
Tanaka, Hisashi*; Kawamoto, Toru*; Asai, Miyuki*; Fukushima, Chikako*; Kurihara, Masato*; Arisaka, Makoto; Nankawa, Takuya; Watanabe, Masayuki
no journal, ,
The adsorption/desorption of cesium, which was included in the radioactive liquid waste was controlled by electrodes which were made by nanoparticle ink of hexacyano ferrous salts. As a result, we will report the development on the recovery method which was reusable.
Tanaka, Hisashi*; Asai, Miyuki*; Fukushima, Chikako*; Kawamoto, Toru*; Kurihara, Masato*; Arisaka, Makoto; Nankawa, Takuya; Watanabe, Masayuki
no journal, ,
Copper hexacyanoferrate complex could be repetitively adsorb cesium cation in terms of electrochemical reduction and oxidation. We demonstrated the adsorption performance of the thin membrane electrode which copper hexacyanoferrate complex was formed on a metal electrode and also attempted thickening of the copper hexacyanoferrate complex layer on the electrode. The copper hexacyanoferrate complex electrochemically adsorbed about 90% of Cs from the solution by 4 times repetition of the adsorption cycle. The electrochemical efficiency of the adsorption increased 50 times by thickening of the copper hexacyanoferrate complex layer on the electrode.
Tanaka, Hisashi*; Chen, R.*; Fukushima, Chikako*; Na, H.*; Asai, Miyuki*; Kawamoto, Toru*; Kurihara, Masato*; Arisaka, Makoto; Nankawa, Takuya; Watanabe, Masayuki
no journal, ,
Hexacyanoferrate complex could repeatedly adsorb and desorb cesium by electrochemical reduction and oxidation. We have recently developed to form a film on a metal electrode in terms of nano particle of the hexacyano ferrate complex "ink". The optimum condition of the electrochemical adsorption and desorption under the acidic condition would be reported in the present study.
 irradiation to metal hexacyanoferrate on adsorption of cesium ion
irradiation to metal hexacyanoferrate on adsorption of cesium ionArisaka, Makoto; Watanabe, Masayuki; Kurihara, Masato*; Chen, R.*; Tanaka, Hisashi*
no journal, ,
no abstracts in English
Arisaka, Makoto; Watanabe, Masayuki; Ishizaki, Manabu*; Kurihara, Masato*; Chen, R.*; Tanaka, Hisashi*
no journal, ,
The influence of irradiation with  -rays to MHCF on stability and Cs adsorption ability in HNO
-rays to MHCF on stability and Cs adsorption ability in HNO solutions was investigated. First, the radiolytic degradation of MHCF was evaluated by determination of the amount of M (= Fe, Cu) in HNO
solutions was investigated. First, the radiolytic degradation of MHCF was evaluated by determination of the amount of M (= Fe, Cu) in HNO solution, which is separated from the sample by filtration after
solution, which is separated from the sample by filtration after  -ray irradiation. The amount of Fe and Cu in the solutions to initial weight of MHCF increased slightly with an increase of the total absorbed dose. Under the condition in this study, the weight percent of Fe and Cu in the solutions to initial weight of MHCF was less than 0.05%. Moreover, the composition of carbon, nitrogen and hydrogen in MHCF is constant independent of the total absorbed dose. The adsorption experiment for Cs was performed using the irradiated MHCF as the adsorbent. The distribution coefficients of Cs onto MHCF were constant independent of the total absorbed dose. From the viewpoint of usage for a long period, it suggests that the MHCF is fully stable.
-ray irradiation. The amount of Fe and Cu in the solutions to initial weight of MHCF increased slightly with an increase of the total absorbed dose. Under the condition in this study, the weight percent of Fe and Cu in the solutions to initial weight of MHCF was less than 0.05%. Moreover, the composition of carbon, nitrogen and hydrogen in MHCF is constant independent of the total absorbed dose. The adsorption experiment for Cs was performed using the irradiated MHCF as the adsorbent. The distribution coefficients of Cs onto MHCF were constant independent of the total absorbed dose. From the viewpoint of usage for a long period, it suggests that the MHCF is fully stable.
Tanaka, Hisashi*; Chen, R.*; Asai, Miyuki*; Fukushima, Chikako*; Kawamoto, Toru*; Ishizaki, Manabu*; Kurihara, Masato*; Arisaka, Makoto; Nankawa, Takuya; Watanabe, Masayuki
no journal, ,
Hexacyanoferrate metal Complex (MHCF) is well-known as adsorption materials for Cesium ion. We have developed the effective processing method by making MHCF of nano particle ink. In this paper, we investigated the effect of the presence of other metal cations on electrochemical adsorption and desorption of cesium.
Chen, R.*; Tanaka, Hisashi*; Fukushima, Chikako*; Asai, Miyuki*; Kawamoto, Toru*; Kurihara, Masato*; Ishizaki, Manabu*; Arisaka, Makoto; Nankawa, Takuya; Watanabe, Masayuki
no journal, ,
Transition metal hexacyanoferrates (MHCFs) are preferred to be competitive ion exchangers over other materials due to their selectivity and high capacity. CuHCF has been much used as precipitants for alkali metal cations, especially cesium, to remove them from aqueous radioactive wastes. Instated of the conventional adsorption by using copper hexacyanoferrate powder, we developed and coated the CuHCF
has been much used as precipitants for alkali metal cations, especially cesium, to remove them from aqueous radioactive wastes. Instated of the conventional adsorption by using copper hexacyanoferrate powder, we developed and coated the CuHCF nanoparticles on the electrode substance, and then performed Cs removal in an electrochemical column System. The objective of this study was to develop a nanoparticle film of CuHCF
nanoparticles on the electrode substance, and then performed Cs removal in an electrochemical column System. The objective of this study was to develop a nanoparticle film of CuHCF for Cs removal and to suggest a promising column technology for sequential removal of actual radiocesium wastewater.
for Cs removal and to suggest a promising column technology for sequential removal of actual radiocesium wastewater.
Arisaka, Makoto; Watanabe, Masayuki; Ishizaki, Manabu*; Kurihara, Masato*; Chen, R.*; Tanaka, Hisashi*
no journal, ,
Recently, we reported an electrochemical application of MHCF (M = Fe, Cu or Ni) film for Cs recovery from nitric acid (HNO ) solutions. It was found that Cs loading and unloading can be easily controlled by modulating the film potential. In this study, the influence of irradiation with
) solutions. It was found that Cs loading and unloading can be easily controlled by modulating the film potential. In this study, the influence of irradiation with  -rays to MHCF films on stability in HNO
-rays to MHCF films on stability in HNO solutions was investigated.
solutions was investigated.
Kakuta, Ryunosuke*; Takamura, Masato*; Xu, P. G.; Iwamoto, Chihiro*; Takanashi, Takaoki*; Otake, Yoshie*; Kurihara, Ryo*; Takahashi, Susumu*; Suzuki, Hiroshi
no journal, ,
Takamura, Masato*; Iwamoto, Chihiro*; Xu, P. G.; Kakuta, Ryunosuke*; Kurihara, Ryo*; Hakoyama, Tomoyuki*; Ikeda, Yoshimasa*; Suzuki, Hiroshi; Otake, Yoshie*
no journal, ,
Takamura, Masato*; Iwamoto, Chihiro*; Xu, P. G.; Ueno, Kota*; Kurihara, Ryo*; Suzuki, Hiroshi; Otake, Yoshie*
no journal, ,
Iwamoto, Chihiro*; Takamura, Masato*; Ueno, Kota*; Kurihara, Ryo*; Xu, P. G.; Suzuki, Hiroshi; Otake, Yoshie*
no journal, ,
no abstracts in English
Xu, P. G.; Iwamoto, Chihiro*; Takamura, Masato*; Otake, Yoshie*; Kurihara, Ryo*; Ueno, Kota*; Kataoka, Minami*; Yamamoto, Kazuyoshi; Harjo, S.; Shobu, Takahisa
no journal, ,
Iwamoto, Chihiro*; Takamura, Masato*; Ueno, Kota*; Kataoka, Minami*; Kurihara, Ryo*; Xu, P. G.; Otake, Yoshie*
no journal, ,
Iwamoto, Chihiro*; Kurihara, Ryo*; Takamura, Masato*; Takahashi, Susumu*; Suzuki, Kosuke*; Xu, P. G.; Otake, Yoshie*
no journal, ,
no abstracts in English