Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ono, Hideo; Kohara, Shinji*; Umesaki, Norimasa*; Suzuya, Kentaro
Journal of Non-Crystalline Solids, 293-295, p.125 - 135, 2001/11
no abstracts in English
Ono, Hideo; Kohara, Shinji*; Umesaki, Norimasa*; Suzuya, Kentaro
Journal of Non-Crystalline Solids, 293-295, p.125 - 135, 2001/11
Times Cited Count:23 Percentile:70.53(Materials Science, Ceramics)no abstracts in English
 O
O by MD simulation
by MD simulationSuzuki, Yoshihiro*; Takase, Keiichi*; Akiyama, Isao*; Suzuya, Kentaro; Umesaki, Norimasa*; Otori, Norikazu*
Materials Transactions, 42(11), p.2242 - 2246, 2001/11
Times Cited Count:7 Percentile:47.33(Materials Science, Multidisciplinary)We have performed the molecular dynamics (MD) simulations for vitreous P O
O using isotropic pair potentials composed only of coulombic and repulsive interaction. The obtained P-O pair distribution function reproduced the two peaks expected from the results of neutron diffraction experiments, in the nearest-neighbor P-O correlation. The neutron-weighted real-space correlation function were also in semi-quantitative agreement with that from the experimental results. The distribution of coordination number for O around P and P around O showed that most P atoms form tetrahedral PO
using isotropic pair potentials composed only of coulombic and repulsive interaction. The obtained P-O pair distribution function reproduced the two peaks expected from the results of neutron diffraction experiments, in the nearest-neighbor P-O correlation. The neutron-weighted real-space correlation function were also in semi-quantitative agreement with that from the experimental results. The distribution of coordination number for O around P and P around O showed that most P atoms form tetrahedral PO units in the glass and three-fifths of O atoms are bridging oxygen, O
units in the glass and three-fifths of O atoms are bridging oxygen, O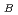 , and the others are terminal one, O
, and the others are terminal one, O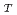 . The pair distribution functions for P-O
. The pair distribution functions for P-O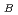 and P-O
and P-O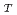 clarified the PO
clarified the PO units have three long P-O
units have three long P-O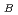 bonds and one short P-O
bonds and one short P-O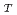 bond. We have concluded that the short-range structure for vitreous P
bond. We have concluded that the short-range structure for vitreous P O
O agrees well with the picture derived from many experiments.
agrees well with the picture derived from many experiments.
Kohara, Shinji*; Suzuya, Kentaro; Kashihara, Yasuharu*; Matsumoto, Norimasa; Umesaki, Norimasa*; Sakai, Ichiro*
Nuclear Instruments and Methods in Physics Research A, 467-468(Part.2), p.1030 - 1033, 2001/07
Times Cited Count:138 Percentile:99.12(Instruments & Instrumentation)no abstracts in English
 glass
glassSuzuya, Kentaro; Shibata, Kaoru*; Umesaki, Norimasa*; Kitamura, Naoyuki*; Kohara, Shinji*
Journal of the Physical Society of Japan, Vol.70, Supplement A, p.256 - 258, 2001/05
no abstracts in English
 O-B
O-B O
O melts and glasses
melts and glassesUmesaki, Norimasa*; Kita, Y.*; Iida, T.*; Handa, K.*; Kohara, Shinji*; Suzuya, Kentaro; Fukunaga, Toshiharu*; Misawa, Masakatsu*
Physics and Chemistry of Glasses, 41(5), p.304 - 308, 2000/10
no abstracts in English
 O
O
Yoneda, Yasuhiro*; Kohara, Shinji*; Suzuya, Kentaro; Umesaki, Norimasa*
Physics and Chemistry of Glasses, 41(5), p.282 - 285, 2000/10
no abstracts in English
Otori, Norikazu; *
JNC TJ9400 2001-002, 56 Pages, 1999/03
High temperature Raman spectroscopy and X-ray diffraction technique were applied to a series of sodium oxides, iron oxides and sodium and iron double oxide from ambient to high temperature for the purpose of examining the usefulness of these methods for the in situ chemical analysis in corrosive environments at elevated temperature on the basis of the necessity for direct observations of chemical reactions between burning sodium and iron-base materials. As a result of Raman scattering measurements at backscattering geometly using a furnace containing a crucible cell, an intense peak assignable to a lattice vibration was observed up to 673 K for Na O, that to stretching mode of O
O, that to stretching mode of O
 ion up to 873K, and that to stretching mode of O
ion up to 873K, and that to stretching mode of O
 ion up to 773K for Na
ion up to 773K for Na O
O . The temperature range in which the method is applicable to the detection of these materials was discussed. As for Na
. The temperature range in which the method is applicable to the detection of these materials was discussed. As for Na O, the structural change may limit the upper temperature, whereas, as to Na
O, the structural change may limit the upper temperature, whereas, as to Na O
O , the color change or the decomposition may limit it. The color change suggests the change of electronic structure such as generation of color center. The spectra for
, the color change or the decomposition may limit it. The color change suggests the change of electronic structure such as generation of color center. The spectra for  -NaFeO
-NaFeO were clearly observed up to 873K, while the background caused by radiation at high temperature considerably increased. For higher temperature measurements, the present method will be hopeful, using an ultraviolet light source in order to reduce the background. As a result of X-ray diffraction measurements, all the obtained patterns agreed well with JCPDS cards data, which shows that the method is useful for the chemical analysis of these materials at high temperature up to 1073K. The obtained spectra and difiraction patterns are collected in this report in order to be available on the chemical analysis of more complex mixtures.
were clearly observed up to 873K, while the background caused by radiation at high temperature considerably increased. For higher temperature measurements, the present method will be hopeful, using an ultraviolet light source in order to reduce the background. As a result of X-ray diffraction measurements, all the obtained patterns agreed well with JCPDS cards data, which shows that the method is useful for the chemical analysis of these materials at high temperature up to 1073K. The obtained spectra and difiraction patterns are collected in this report in order to be available on the chemical analysis of more complex mixtures.
 O-B
O-B O
O glasses and melts
glasses and meltsUmesaki, Norimasa*; Kita, Y.*; Handa, K.*; Kohara, Shinji*; Suzuya, Kentaro; Fukunaga, T.*; Misawa, M.*; Iida, T.*
Denki Kagaku Oyobi Kogyo Butsuri Kagaku, 67(6), p.541 - 546, 1999/00
no abstracts in English
 O-B
O-B O
O (M: Na and K) glasses and melts by neutron diffraction
(M: Na and K) glasses and melts by neutron diffractionHanda, K.*; Kita, Y.*; Kohara, Shinji*; Suzuya, Kentaro; Fukunaga, Toshiharu*; Misawa, Masakatsu*; Iida, T.*; *; Umesaki, Norimasa*
Journal of Physics and Chemistry of Solids, 60(8-9), p.1465 - 1471, 1999/00
Times Cited Count:16 Percentile:64.77(Chemistry, Multidisciplinary)no abstracts in English
Otori, Norikazu*; *
PNC TJ9642 98-001, 24 Pages, 1998/03
Raman spectroscopy was applied to sodium oxides and sodium and iron double oxide from ambient to high temperature for the purpose of establishing it as an in-situ method of chemical analysis at elevated temperature, based on the necessity for direct observations of chemical reactions between burning sodium and iron-base materials. X-ray diffraction method was also applied to the iron double oxide from ambient to high temperature in order to determine its phase directly. The several findings were obtained as follows. As a result of Raman scattering measurements of Na O, an intense peak, which could be assigned to a lattice vibration in the antifluorite structure, was observed around 200 cm
O, an intense peak, which could be assigned to a lattice vibration in the antifluorite structure, was observed around 200 cm up to 823 K. The color of commercially available Na
up to 823 K. The color of commercially available Na O
O was light yellow but changed to black above 573 K. The Raman scattering was, however, observed for the O
was light yellow but changed to black above 573 K. The Raman scattering was, however, observed for the O
 stretching mode from ambient to 773 K. Based on Raman scattering and X-ray diffraction measurements of a sintered mixture of 2Na
stretching mode from ambient to 773 K. Based on Raman scattering and X-ray diffraction measurements of a sintered mixture of 2Na CO
CO and Fe
and Fe O
O 3, the chemical changes might be proposed from ambient to high temperature for example as follows: [Na
3, the chemical changes might be proposed from ambient to high temperature for example as follows: [Na CO
CO
 Na
Na O+CO
O+CO ] and [NaFeO
] and [NaFeO +Na
+Na O
O  Na
Na FeO
FeO .]
.]
Urakawa, Satoru*; Igawa, Naoki; Umesaki, Norimasa*; Igarashi, Kazuo*; Shimomura, Osamu*; Ono, Hideo
High Pressure Research, 14(4-6), p.375 - 382, 1996/12
Times Cited Count:6 Percentile:53.11(Physics, Multidisciplinary)no abstracts in English
Umesaki, Norimasa*; Ono, Hideo; Igarashi, Kazuo*; *
Journal of Non-Crystalline Solids, 150, p.302 - 306, 1992/00
Times Cited Count:0 Percentile:0.05(Materials Science, Ceramics)no abstracts in English
 -BeF
-BeF based fluoride glasses for IR transmission
based fluoride glasses for IR transmissionOno, Hideo; Igawa, Naoki; Ishii, Yoshinobu; Umesaki, Norimasa*; *
Journal of Nuclear Materials, 191-194, p.525 - 529, 1992/00
no abstracts in English
 BeF
BeF and NaBeF
and NaBeF by X-ray diffraction
by X-ray diffraction*; ; *; Furukawa, Kazuo
J.Chem.Soc.,Faraday Trans.,I, 78, p.2051 - 2058, 1982/00
no abstracts in English
*; *; ; *; Furukawa, Kazuo
J.Chem.Soc.,Faraday Trans.,I, 77, p.169 - 175, 1981/00
no abstracts in English
; *; *; Furukawa, Kazuo; *
J.Chem.Res., p.158 - 159, 1978/00
no abstracts in English
Myochin, Munetaka; Kofuji, Hirohide; Yamana, Hajimu*; Shirai, Osamu*; Yamamura, Tsutomu*; Umesaki, Norimasa*; Matsuura, Haruaki*; Kajinami, Akihiko*; Iwadate, Yasuhiko*; Otori, Norikazu*; et al.
no journal, ,
no abstracts in English
Hasegawa, Takayuki*; Uemura, Masaharu*; Takueuchi, Kazuki*; Kodaka, Takuya*; Fukada, Noboru*; Umesaki, Norimasa*; Fukushima, Sei*; Teraoka, Yuden; Haruyama, Yuichi*; Niibe, Masahito*; et al.
no journal, ,
no abstracts in English