Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Inagawa, Jun; Kitatsuji, Yoshihiro; Otobe, Haruyoshi; Nakada, Masami; Takano, Masahide; Akie, Hiroshi; Shimizu, Osamu; Komuro, Michiyasu; Oura, Hirofumi*; Nagai, Isao*; et al.
JAEA-Technology 2021-001, 144 Pages, 2021/08
Plutonium Research Building No.1 (Pu1) was qualified as a facility to decommission, and preparatory operations for decommission were worked by the research groups users and the facility managers of Pu1. The operation of transportation of whole nuclear materials in Pu1 to Back-end Cycle Key Element Research Facility (BECKY) completed at Dec. 2020. In the operation included evaluation of criticality safety for changing permission of the license for use nuclear fuel materials in BECKY, cask of the transportation, the registration request of the cask at the institute, the test transportation, formulation of plan for whole nuclear materials transportation, and the main transportation. This report circumstantially shows all of those process to help prospective decommission.
Uosaki, Kohei*; Morita, Jun*; Katsuzaki, Tomoko*; Takakusagi, Satoru*; Tamura, Kazuhisa; Takahashi, Masamitsu; Mizuki, Junichiro; Kondo, Toshihiro*
Journal of Physical Chemistry C, 115(25), p.12471 - 12482, 2011/06
Times Cited Count:13 Percentile:40.03(Chemistry, Physical)Ag/AgCl reaction at the Ag bilayer, which was underpotentially prepared on a Au(111) surface, was investigated using electrochemical quartz crystal microbalance (EQCM), scanning tunneling microscopy (STM), surface X-ray scattering (SXS), and electrochemical techniques. When the potential was scanned positively from -200 mV, the Cl ion was adsorbed on the Au(111) electrode surface around 0 mV, and then the phase transition of the adsorbed Cl
ion was adsorbed on the Au(111) electrode surface around 0 mV, and then the phase transition of the adsorbed Cl ion layer from random orientation to (
ion layer from random orientation to (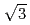

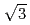 ) structure took place at around +130 mV. The Ag bilayer and Cl
) structure took place at around +130 mV. The Ag bilayer and Cl ions were oxidatively reacted to form the AgCl monolayer with (
ions were oxidatively reacted to form the AgCl monolayer with (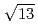

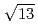 )
) 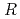 13.9
13.9 structure around +200 mV, accompanied with the formation of AgCl monocrystalline clusters on the AgCl monolayer surface. The structure of the AgCl monolayer on the Au(111) surface was changed from (
structure around +200 mV, accompanied with the formation of AgCl monocrystalline clusters on the AgCl monolayer surface. The structure of the AgCl monolayer on the Au(111) surface was changed from (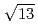

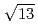 )R13.9
)R13.9 structure to (4
structure to (4 4) structure around +500 mV.
4) structure around +500 mV.
Kondo, Toshihiro*; Morita, Jun*; Hanaoka, Kazuya*; Takakusagi, Satoru*; Tamura, Kazuhisa; Takahashi, Masamitsu; Mizuki, Junichiro; Uosaki, Kohei*
Journal of Physical Chemistry C, 111(35), p.13197 - 13204, 2007/09
Times Cited Count:84 Percentile:89.3(Chemistry, Physical)Potential-dependent surface structures of Au(111) and Au(100) single-crystal electrodes in a 50 mM H SO
SO solution were investigated at an atomic level using in situ surface X-ray scattering (SXS) techniques. It was confirmed that both the Au(111) and Au(100) surfaces were reconstructed with an attached submonolayer of an oxygen species, most probably water, at 0 V (vs Ag/AgCl). Results at +0.95 V supported a previously suggested model for both the Au(111) and the Au(100) electrodes that, based on infrared and scanning tunneling microscopy measurements, the surfaces were a (1
solution were investigated at an atomic level using in situ surface X-ray scattering (SXS) techniques. It was confirmed that both the Au(111) and Au(100) surfaces were reconstructed with an attached submonolayer of an oxygen species, most probably water, at 0 V (vs Ag/AgCl). Results at +0.95 V supported a previously suggested model for both the Au(111) and the Au(100) electrodes that, based on infrared and scanning tunneling microscopy measurements, the surfaces were a (1 1) structure with the coadsorbed sulfate anion and hydronium cation (H
1) structure with the coadsorbed sulfate anion and hydronium cation (H O
O ). At +1.05 V, where a small amount of an anodic current flowed, adsorption of a monolayer of oxygen species was observed on both surfaces.
). At +1.05 V, where a small amount of an anodic current flowed, adsorption of a monolayer of oxygen species was observed on both surfaces.
Morita, Yosuke; Yagi, Toshiaki; *
Denki Gakkai Yuden, Zetsuen Zairyo Kenkyukai Shiryo; DEI-99-12, p.23 - 26, 1999/02
no abstracts in English
Morita, Yosuke; Yagi, Toshiaki; *
Denki Gakkai Yuden, Zetsuen Zairyo Kenkyukai Shiryo; DEI-99-13, p.27 - 30, 1999/02
no abstracts in English
Yagi, Toshiaki; Morita, Yosuke; Seguchi, Tadao; *
Denki Gakkai Yuden, Zetsuen Zairyo Kenkyukai Shiryo; DEI-94-90, 0, p.21 - 28, 1994/12
no abstracts in English
; ; ;
JAERI-M 86-001, 23 Pages, 1986/02
no abstracts in English
; ; ; ; ; *
JAERI-M 84-044, 51 Pages, 1984/03
no abstracts in English
Kugo, Teruhiko; Hamada, Jumpei; Mineo, Hideaki; Okubo, Nariaki; Takano, Masahide; Matsumura, Tatsuro; Watanabe, Masayuki; Iwamoto, Osamu; Morita, Yasuji; Maekawa, Fujio
no journal, ,
The status of development of human resources in JAEA for research and development to support nuclear energy utilization is reported.