Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Al
Al Ga
Ga O
O scintillators by gamma-ray-induced positron annihilation lifetime spectroscopy
scintillators by gamma-ray-induced positron annihilation lifetime spectroscopyFujimori, Kosuke*; Kitaura, Mamoru*; Taira, Yoshitaka*; Fujimoto, Masaki*; Zen, H.*; Watanabe, Shinta*; Kamada, Kei*; Okano, Yasuaki*; Kato, Masahiro*; Hosaka, Masahito*; et al.
Applied Physics Express, 13(8), p.085505_1 - 085505_4, 2020/08
Times Cited Count:5 Percentile:33.01(Physics, Applied)To clarify the existence of cation vacancies in Ce-doped Gd Al
Al Ga
Ga O
O (Ce:GAGG) scintillators, we performed gamma-ray-induced positron annihilation lifetime spectroscopy (GiPALS). GiPAL spectra of GAGG and Ce:GAGG comprised two exponential decay components, which were assigned to positron annihilation at bulk and defect states. By an analogy with Ce:Y
(Ce:GAGG) scintillators, we performed gamma-ray-induced positron annihilation lifetime spectroscopy (GiPALS). GiPAL spectra of GAGG and Ce:GAGG comprised two exponential decay components, which were assigned to positron annihilation at bulk and defect states. By an analogy with Ce:Y Al
Al O
O , the defect-related component was attributed to Al/Ga-O divacancy complexes. This component was weaker for Ce, Mg:GAGG, which correlated with the suppression of shallow electron traps responsible for phosphorescence. Oxygen vacancies were charge compensators for Al/Ga vacancies. The lifetime of the defect-related component was significantly changed by Mg co-doping. This was understood by considering aggregates of Mg
, the defect-related component was attributed to Al/Ga-O divacancy complexes. This component was weaker for Ce, Mg:GAGG, which correlated with the suppression of shallow electron traps responsible for phosphorescence. Oxygen vacancies were charge compensators for Al/Ga vacancies. The lifetime of the defect-related component was significantly changed by Mg co-doping. This was understood by considering aggregates of Mg ions at Al/Ga sites with oxygen vacancies, which resulted in the formation of vacancy clusters.
ions at Al/Ga sites with oxygen vacancies, which resulted in the formation of vacancy clusters.
Mishima, Ria; Inaba, Yusuke*; Tachioka, Sotaro*; Harigai, Miki*; Watanabe, Shinta*; Onoe, Jun*; Nakase, Masahiko*; Matsumura, Tatsuro; Takeshita, Kenji*
Chemistry Letters, 49(1), p.83 - 86, 2020/01
Times Cited Count:4 Percentile:21.18(Chemistry, Multidisciplinary)Separation of platinum group metals (PGMs) from high-level liquid waste generated from the reprocessing of spent nuclear fuels is important to produce good quality vitrified glass for final disposal. A new sorbent, Aluminum hexacyanoferrate (AlHCF), was synthesized and the general sorption behavior of PGMs from concentrated nitric acid was examined. Nitric acid caused substantial elution of AlHCF but the sorption of Pd stabilized the structure. Consequently, Rh was sorbed in the presence of Pd, whereas single Rh sorption caused complete dissolution of AlHCF. Relation between sorbed mount of Pd vs eluted Al and Fe revealed that the elution ratio of Al and Fe was not the same as molar ratio of synthesized AlHCF, indicating the re-sorption of Fe resulted in formation of new structure. The sorption mechanism of PGMs by this new sorbent, AlHCF, not only the simple ion exchange, but also oxidation reduction reaction as well as kinetics play important rule. Understanding the general sorption and dissolution behavior will help improve the sorption performance of PGMs by AlHCF.
Mishima, Ria; Tachioka, Sotaro*; Inaba, Yusuke*; Harigai, Miki*; Matsumura, Tatsuro; Watanabe, Shinta*; Onoe, Jun*; Nakase, Masahiko*; Takeshita, Kenji*
no journal, ,
In Japan, the final disposal of high level liquid waste (HLLW) will be done after vitrification into borosilicate glass and then disposed into deep underground. In this vitrification process, there are some concerns. The first concern is precipitation of platinum group metals (PGMs) in the melter due to their low solubility into borosilicate glass. The second concern is the formation of yellow phase caused by Mo content. The final concern is the generation of huge number of vitrified glasses and the requirement of wide space needed for final disposal. Among many kinds of extractants and adsorbents for separation of such metal ions, metal hexacyanoferrate (HCFs) were reported to have an ability to adsorb PGMs. The objective of this study is to elucidate the adsorption behavior of aluminum hexacyanoferrate (AlHCF) for various metal ions and understand the relation between elution and adsorption. The effect of synthetic and workup conditions on PGM and Mo adsorption from simulated HLLW (sHLLW) was surveyed. Also, the relationship between adsorption of metal ions and elution of the AlHCF was studied. The synthesized AlHCF showed adsorption performance for PGMs and Mo in simulated HLLW. As a result of an adsorption test with a Pd single component solution to investigate the adsorption mechanism, the eluted element ratio was Fe:Al = 1:4 in the Pd adsorption test. However, the element ratio was Fe:Al = 3:4 in the original AlHCF. Therefore, it was suggested the existence of not only Pd adsorption, but also resorption and stabilization mechanisms.
Mishima, Ria; Tachioka, Sotaro*; Inaba, Yusuke*; Harigai, Miki*; Matsumura, Tatsuro; Watanabe, Shinta*; Onoe, Jun*; Nakase, Masahiko*; Takeshita, Kenji*
no journal, ,
In Japan, the final disposal of high level liquid waste (HLLW) will be done after vitrification into borosilicate glass and then disposed into deep underground. In this vitrification process, there are some concerns. The first concern is precipitation of platinum group metals (PGMs) in the melter due to their low solubility into borosilicate glass. The second concern is the formation of yellow phase caused by Mo content. The final concern is the generation of huge number of vitrified glasses and the requirement of wide space needed for final disposal. Among many kinds of extractants and adsorbents for separation of such metal ions, metal hexacyanoferrate (HCFs) were reported to have an ability to adsorb PGMs. The objective of this study is to elucidate the adsorption behavior of aluminum hexacyanoferrate (AlHCF) for various metal ions and understand the relation between elution and adsorption. The effect of synthetic and workup conditions on PGM and Mo adsorption from simulated HLLW (sHLLW) was surveyed. Also, the relationship between adsorption of metal ions and elution of the AlHCF was studied. The synthesized AlHCF showed adsorption performance for PGMs and Mo in simulated HLLW. As a result of an adsorption test with a Pd single component solution to investigate the adsorption mechanism, the eluted element ratio was Fe:Al = 1:4 in the Pd adsorption test. However, the element ratio was Fe:Al = 3:4 in the original AlHCF. Therefore, it was suggested the existence of not only Pd adsorption, but also resorption and stabilization mechanisms.
Kitaura, Mamoru*; Fujimori, Kosuke*; Taira, Yoshitaka*; Fujimoto, Masaki*; Zen, H.*; Hirade, Tetsuya; Kamada, Kei*; Watanabe, Shinta*; Onishi, Akimasa*
no journal, ,
Positron annihilation spectroscopy is the only way to investigate the properties of cation vacancies because they are negatively charged. We generated high-energy pulsed gamma rays by the vertical collision of an ultrashort pulse laser and electron beam. In this study, we investigated the vacancy-type defects present in the crystals of GAGG(Gd Al
Al Ga
Ga O
O ), GAGG: Ce and GAGG: Ce, Mg by positron annihilation lifetime spectroscopy using the high-energy gamma rays. The lifetime of the defect-related component was significantly changed by Mg co-doping. This was understood by considering aggregates of Mg
), GAGG: Ce and GAGG: Ce, Mg by positron annihilation lifetime spectroscopy using the high-energy gamma rays. The lifetime of the defect-related component was significantly changed by Mg co-doping. This was understood by considering aggregates of Mg ions at Al/Ga sites with oxygen vacancies, which resulted in the formation of vacancy clusters.
ions at Al/Ga sites with oxygen vacancies, which resulted in the formation of vacancy clusters.
 Al
Al Ga
Ga O
O crystals revealed by gamma-ray induced positron annihilation lifetime spectroscopy
crystals revealed by gamma-ray induced positron annihilation lifetime spectroscopyFujimori, Kosuke*; Kitaura, Mamoru*; Taira, Yoshitaka*; Fujimoto, Masaki*; Zen, H.*; Hirade, Tetsuya; Kamada, Kei*; Watanabe, Shinta*; Onishi, Akimasa*
no journal, ,
We generated high-energy pulsed gamma rays by the vertical collision of an ultrashort pulse laser and electron beam. In this study, we investigated the vacancy-type defects present in the crystals of GAGG(Gd Al
Al Ga
Ga O
O ), GAGG: Ce and GAGG: Ce, Mg by positron annihilation lifetime spectroscopy using the high-energy gamma rays. The lifetime of the defect-related component was significantly changed by Mg co-doping. This indicates that the Al/Ga vacancies disappear. This fact corresponds well with the suppression of the phosphorescence component and is an important result showing that the Mg co-doping is effective in suppressing the shallow electron capture center.
), GAGG: Ce and GAGG: Ce, Mg by positron annihilation lifetime spectroscopy using the high-energy gamma rays. The lifetime of the defect-related component was significantly changed by Mg co-doping. This indicates that the Al/Ga vacancies disappear. This fact corresponds well with the suppression of the phosphorescence component and is an important result showing that the Mg co-doping is effective in suppressing the shallow electron capture center.
Kobayashi, Toru; Nakase, Masahiko*; Watanabe, Shinta*; Maki, Ryosuke*; Maruyama, Satofumi*; Kikunaga, Hidetoshi*; Asano, Hidekazu*; Sakuragi, Tomofumi*; Hamada, Ryo*; Harigai, Miki*
no journal, ,
no abstracts in English
Minowa, Kazuki*; Watanabe, So; Ban, Yasutoshi; Nakase, Masahiko*; Watanabe, Shinta*; Matsuura, Haruaki*
no journal, ,
no abstracts in English
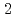
Doshi, Satoru*; Maeda, Kazuki*; Taira, Yoshitaka*; Watanabe, Shinta*; Hirade, Tetsuya
no journal, ,
It has been reported that oxygen vacancies in cerium oxide (CeO ) affect catalytic activity. There is a need for highly sensitive in-situ measurements of the state and concentration of oxygen vacancies during catalytic reactions, and we are considering using the positron annihilation method. This time, we measured samples with different particle sizes to clarify the sites where positrons are trapped. From the lifetime spectrum of a particle size of 2 nm, which has a high intensity of the long-lived component, we first determined the lifetime value of the longest-lived component to be 390.8
) affect catalytic activity. There is a need for highly sensitive in-situ measurements of the state and concentration of oxygen vacancies during catalytic reactions, and we are considering using the positron annihilation method. This time, we measured samples with different particle sizes to clarify the sites where positrons are trapped. From the lifetime spectrum of a particle size of 2 nm, which has a high intensity of the long-lived component, we first determined the lifetime value of the longest-lived component to be 390.8 1.6 ps. Next, in the lifetime spectrum of a particle size of 60 nm, where the longest lifetime component is the smallest, the longest lifetime component was fixed at 390.8 ps, and the lifetime value of the other positron trap site was determined to be 199.4
1.6 ps. Next, in the lifetime spectrum of a particle size of 60 nm, where the longest lifetime component is the smallest, the longest lifetime component was fixed at 390.8 ps, and the lifetime value of the other positron trap site was determined to be 199.4 7.4 ps. These values are close to the lifetimes of positrons trapped in surface and neutral oxygen defects in first-principles calculations.
7.4 ps. These values are close to the lifetimes of positrons trapped in surface and neutral oxygen defects in first-principles calculations.
 by using gamma-ray induced positron annihilation lifetime measurement
by using gamma-ray induced positron annihilation lifetime measurementDoshi, Satoru*; Maeda, Kazuki*; Taira, Yoshitaka*; Watanabe, Shinta*; Hirade, Tetsuya
no journal, ,
Gamma-ray-induced positron annihilation lifetime measurement (GiPALS), which utilizes the generation of positrons inside a sample by pair generation with gamma rays, has a significantly smaller background component than conventional measurement methods. It can perform measurements in harsh environments such as high temperature and high pressure because there is no positron source inside the sample. It has been reported that the positron annihilation lifetime spectrum of CeO can be fitted with two components. However, in reality, it is thought that there are at least three components: a component of annihilation in the bulk, a component of annihilation trapped in lattice defects, a component of annihilation trapped on the surface. Therefore, in this study, we tried to fit three components and assigned each by changing the particle size. In addition, we theoretically investigated the bulk, defect, and surface components in the positron annihilation lifetime of CeO
can be fitted with two components. However, in reality, it is thought that there are at least three components: a component of annihilation in the bulk, a component of annihilation trapped in lattice defects, a component of annihilation trapped on the surface. Therefore, in this study, we tried to fit three components and assigned each by changing the particle size. In addition, we theoretically investigated the bulk, defect, and surface components in the positron annihilation lifetime of CeO using first-principles calculations.
using first-principles calculations.