Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ito, Hiroto*; Shiotsu, Hiroyuki; Tanaka, Yoichi*; Nishihara, Satomichi*; Sugiyama, Tomoyuki; Maruyama, Yu
JAEA-Data/Code 2018-012, 42 Pages, 2018/10
Chemical composition of fission products transported in nuclear facilities in severe accidents is controlled by slower chemical reaction rates, therefore, it could be different from that evaluated on the chemical equilibrium assumption. Hence, it is necessary to evaluate the chemical composition with reaction kinetics. On the other hand, databases applicable to the analysis of nuclear facilities have not been constructed because knowledge of reaction rates of complex chemical reactions in severe accidents is currently limited. Accordingly, we have developed the CHEMKEq code based on a partial mixed model with chemical equilibrium and reaction kinetics to decrease uncertainties of the chemical composition caused by the reaction rate. The CHEMKEq code, under mass conservation law, firstly evaluates chemical species obeying the chemical equilibrium model, and then, relatively slow reactions are solved by the reaction kinetics model. Moreover, the CHEMKEq code has a multiplicity of use in evaluations of chemical composition because general chemical equilibrium and reaction kinetics models are also available and databases required to calculation are external file formats. This report is the user's guide of the CHEMKEq code, showing models, solution methods, structure of the code and calculation examples. And information to run the CHEMKEq code is summarized in appendixes.
*; Tomimitsu, Hiroshi; *; *; *; Aizawa, Kazuya; Okayasu, Satoru; *; Kazumata, Yukio*; Y.C.Jiang*; et al.
Japanese Journal of Applied Physics, Part 1, 36(12A), p.7296 - 7301, 1997/12
Times Cited Count:6 Percentile:36.78(Physics, Applied)no abstracts in English
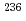 U using ICP-MS/MS
U using ICP-MS/MSTanaka, Yasuyuki; Do, V. K.; Motoki, Yoshiaki*; Ota, Yuki; Iwahashi, Hiroyuki; Kurosawa, Kiyoko*; Furuse, Takahiro
no journal, ,
We examined the measurement method of 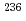 U using triple-quadrupole inductively coupled plasma-mass spectrometry (ICP-MS/MS, Agilent 8900). The measurable mass to charge ratio (m/z) is expanded up to 275, the device can measure
U using triple-quadrupole inductively coupled plasma-mass spectrometry (ICP-MS/MS, Agilent 8900). The measurable mass to charge ratio (m/z) is expanded up to 275, the device can measure 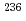 U
U O
O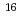 O (m/z = 268) generated by the reaction with
O (m/z = 268) generated by the reaction with 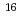 O, which was used as a reaction gas. This mass shift mode decreased the interference from hydride of
O, which was used as a reaction gas. This mass shift mode decreased the interference from hydride of  U compared to the previously reported analytical method, which improved limit of detection of
U compared to the previously reported analytical method, which improved limit of detection of 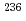 U using ICP-MS/MS. In this presentation, the optimized measurement condition of
U using ICP-MS/MS. In this presentation, the optimized measurement condition of 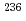 U measured at m/z = 268 by ICP-MS/MS is reported.
U measured at m/z = 268 by ICP-MS/MS is reported.