Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 media
mediaKasamatsu, Yoshitaka; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Haba, Hiromitsu*; Ishii, Yasuo; Tome, Hayato; Nishinaka, Ichiro; Akiyama, Kazuhiko*; Kikunaga, Hidetoshi*; et al.
no journal, ,
Dubnium-262 was produced in the  Cm(
Cm( F, 5
F, 5 ) reaction at the JAEA tandem accelerator. Adsorption of Db on the anion-exchange resin was investigated in 0.89 M HF/0.3 M HNO
) reaction at the JAEA tandem accelerator. Adsorption of Db on the anion-exchange resin was investigated in 0.89 M HF/0.3 M HNO solution. The anion-exchange behavior of Nb, Ta, and Pa as homologues of Db was also examined in details in HF/HNO
solution. The anion-exchange behavior of Nb, Ta, and Pa as homologues of Db was also examined in details in HF/HNO solutions. From the comparison of those results, we found that the adsorption of Db on the anion-exchange resin is considerably weaker than that of Ta and is relatively similar to those of Nb and Pa in the studied conditions.
solutions. From the comparison of those results, we found that the adsorption of Db on the anion-exchange resin is considerably weaker than that of Ta and is relatively similar to those of Nb and Pa in the studied conditions.
 solutions
solutionsKasamatsu, Yoshitaka; Tome, Hayato; Toyoshima, Atsushi; Tsukada, Kazuaki; Asai, Masato; Ishii, Yasuo; Nishinaka, Ichiro; Sato, Tetsuya; Shinohara, Nobuo; Nagame, Yuichiro; et al.
no journal, ,
Anion-exchange behavior of  Db (half-life
Db (half-life 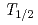 = 34 s) produced in the
= 34 s) produced in the  Cm(
Cm( F, 5
F, 5 ) reaction at the JAEA tandem accelerator was investigated in the mixed 0.89 M HF/0.3 M HNO
) reaction at the JAEA tandem accelerator was investigated in the mixed 0.89 M HF/0.3 M HNO solution ([F
solution ([F ] = 3
] = 3  10
10 M) with the automated ion-exchange separation apparatus coupled with the detection system for alpha-spectroscopy (AIDA). Anion-exchange behavior of its lighter homologues, Nb and Ta, was also studied under the same conditions using
M) with the automated ion-exchange separation apparatus coupled with the detection system for alpha-spectroscopy (AIDA). Anion-exchange behavior of its lighter homologues, Nb and Ta, was also studied under the same conditions using  Nb (
Nb (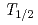 = 14.3 min) and
= 14.3 min) and 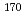 Ta (
Ta (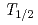 = 6.76 min) produced in the
= 6.76 min) produced in the 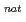 Ge(
Ge( F,
F, 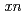 ) and
) and 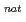 Gd(
Gd( F,
F, 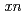 ) reactions, respectively. It was found that the adsorption probability on the anion-exchange resin is in the order of Ta
) reactions, respectively. It was found that the adsorption probability on the anion-exchange resin is in the order of Ta  Nb
Nb  Db under the present condition.
Db under the present condition.
 solutions
solutionsKasamatsu, Yoshitaka; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Haba, Hiromitsu*; Ishii, Yasuo; Tome, Hayato; Nishinaka, Ichiro; Akiyama, Kazuhiko*; Kikunaga, Hidetoshi*; et al.
no journal, ,
Dubnium-262 was produced in the nuclear reaction of  Cm(
Cm( F,5n)
F,5n) Db using the JAEA Tandem accelerator. The reaction products were rapidly transported to the chemistry laboratory and the anion-exchange behavior of Db in HF/HNO
Db using the JAEA Tandem accelerator. The reaction products were rapidly transported to the chemistry laboratory and the anion-exchange behavior of Db in HF/HNO media was investigated with an on-line rapid ion-exchange apparatus. Based on the comparison of the behavior of Db with that of its homologues (Nb, Ta, and Pa), the fluoride complex formation of Db was discussed.
media was investigated with an on-line rapid ion-exchange apparatus. Based on the comparison of the behavior of Db with that of its homologues (Nb, Ta, and Pa), the fluoride complex formation of Db was discussed.