Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Inagawa, Kohei*; Matsumura, Daiju; Taniguchi, Masashi*; Uegaki, Shinya*; Nakayama, Tomohito*; Urano, Junnosuke*; Aotani, Takuro*; Tanaka, Hirohisa*
Journal of Physical Chemistry C, 127(24), p.11542 - 11549, 2023/06
Times Cited Count:0 Percentile:0(Chemistry, Physical)Tanaka, Hirohisa*; Masaki, Sayaka*; Aotani, Takuro*; Inagawa, Kohei*; Iwata, Sogo*; Aida, Tatsuya*; Yamamoto, Tadasuke*; Kita, Tomoaki*; Ono, Hitomi*; Takenaka, Keisuke*; et al.
SAE Technical Paper 2022-01-0534 (Internet), 10 Pages, 2022/03
Reinecke, E.-A.*; Takenaka, Keisuke*; Ono, Hitomi*; Kita, Tomoaki*; Taniguchi, Masashi*; Nishihata, Yasuo; Hino, Ryutaro; Tanaka, Hirohisa*
International Journal of Hydrogen Energy, 46(23), p.12511 - 12521, 2021/03
Times Cited Count:4 Percentile:22.37(Chemistry, Physical)The safe decommissioning as well as decontamination of the radioactive waste resulting from the nuclear accident in Fukushima Daiichi represents a huge task for the next decade. At present, research and development on long-term safe storage containers has become an urgent task with international cooperation in Japan. One challenge is the generation of hydrogen and oxygen in significant amounts by means of radiolysis inside the containers, as the nuclear waste contains a large portion of sea water. The generation of radiolysis gases may lead to a significant pressure build-up inside the containers and to the formation of flammable gases with the risk of ignition and the loss of integrity. In the framework of the project "R&D on technology for reducing concentration of flammable gases generated in long-term waste storage containers" funded by the Japanese Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT), the potential application of catalytic recombiner devices inside the storage containers is investigated. In this context, a suitable catalyst based on the so-called intelligent automotive catalyst for use in a recombiner is under consideration. The catalyst is originally developed and mass-produced for automotive exhaust gas purification, and is characterized by having a self-healing function of precious metals (Pd, Pt and Rh) dissolved as a solid solution in the perovskite type oxides. The basic features of this catalyst have been tested in an experimental program. The test series in the REKO-4 facility has revealed the basic characteristics of the catalyst required for designing the recombiner system.
Ono, Hitomi*; Takenaka, Keisuke*; Kita, Tomoaki*; Taniguchi, Masashi*; Matsumura, Daiju; Nishihata, Yasuo; Hino, Ryutaro; Reinecke, E.-A.*; Takase, Kazuyuki*; Tanaka, Hirohisa*
E-Journal of Advanced Maintenance (Internet), 11(1), p.40 - 45, 2019/05
 X-ray absorption spectroscopy study on water formation reaction of palladium metal nanoparticle catalysts
X-ray absorption spectroscopy study on water formation reaction of palladium metal nanoparticle catalystsMatsumura, Daiju; Taniguchi, Masashi*; Tanaka, Hirohisa*; Nishihata, Yasuo
International Journal of Hydrogen Energy, 42(11), p.7749 - 7754, 2017/03
Times Cited Count:5 Percentile:14.05(Chemistry, Physical)Kamiji, Yu; Taniguchi, Masashi*; Nishihata, Yasuo; Nagaishi, Ryuji; Tanaka, Hirohisa*; Hirata, Shingo*; Hara, Mikiya; Hino, Ryutaro
E-Journal of Advanced Maintenance (Internet), 7(1), p.84 - 89, 2015/05
For hydrogen mitigation, a new type passive autocatalytic recombiner is under developing. This new recombiner has been developed from automotive monolithic catalyst in order to reduce weight and to improve hydrogen treating capacity, environmental resistance and product quality. In this study, activation energy of hydrogen-oxygen recombination reaction was examined to clarify the basic characteristics of the catalyst. In addition, the degradation of the catalyst by  -ray irradiation simulating the environmental condition in nuclear power plants was also examined. As a result, the activation energy was experimentally estimated at 5.75 kJ/mol. Besides, no significant differences were observed in compositional distribution from the EPMA results. On the other hand, specific surface area of the catalyst and surface area of the precious metals were increased. Moreover, catalyst performance test showed that
-ray irradiation simulating the environmental condition in nuclear power plants was also examined. As a result, the activation energy was experimentally estimated at 5.75 kJ/mol. Besides, no significant differences were observed in compositional distribution from the EPMA results. On the other hand, specific surface area of the catalyst and surface area of the precious metals were increased. Moreover, catalyst performance test showed that  -ray irradiation up to 1.0 MGy can increase activity of catalyst.
-ray irradiation up to 1.0 MGy can increase activity of catalyst.
Kamiji, Yu; Matsumura, Daiju; Taniguchi, Masashi*; Nishihata, Yasuo; Tanaka, Hirohisa*; Hirata, Shingo*; Hara, Mikiya; Hino, Ryutaro
Proceedings of 23rd International Conference on Nuclear Engineering (ICONE-23) (DVD-ROM), 4 Pages, 2015/05
In a severe accident at a nuclear power plant, a large amount of hydrogen can be released to primary containment vessel or reactor building. Passive autocatalytic recombiner (PAR) is one of the most effective systems for hydrogen mitigation and safety accident management. The new type PAR is under developing to improve conventional PARs, especially its size and weight. In this study, the influence of steam coexistence for the automotive catalyst activity was experimentally examined. These results show that the steam slightly affects the reaction start up and catalyst activity.
Jarrige, I.*; Ishii, Kenji; Matsumura, Daiju; Nishihata, Yasuo; Yoshida, Masahiro*; Kishi, Hirofumi*; Taniguchi, Masashi*; Uenishi, Mari*; Tanaka, Hirohisa*; Kasai, Hideaki*; et al.
ACS Catalysis, 5(2), p.1112 - 1118, 2015/02
Times Cited Count:18 Percentile:43.96(Chemistry, Physical)Kamiji, Yu; Taniguchi, Masashi*; Nishihata, Yasuo; Nagaishi, Ryuji; Tanaka, Hirohisa*; Hirata, Shingo*; Hara, Mikiya; Hino, Ryutaro
Proceedings of 2nd International Conference on Maintenance Science and Technology (ICMST-Kobe 2014), p.87 - 88, 2014/11
For hydrogen mitigation, a new type passive autocatalytic recombiner is under development. In this study, the activation energy of hydrogen-oxygen recombination reaction was examined to clarify the basic characteristics of the catalyst. In addition, the degradation of the catalyst by  -ray irradiation simulating the environmental condition in nuclear power plants was also examined. As a result, the activation energy was experimentally estimated at 5.75 kJ/mol. Besides, no significant differences were observed in the compositional distribution from the EPMA results between the non-irradiated and the irradiated catalyst. However, the irradiated catalyst showed much more activity because of larger specific surface area of the catalyst and surface area of the precious metals. It showed that
-ray irradiation simulating the environmental condition in nuclear power plants was also examined. As a result, the activation energy was experimentally estimated at 5.75 kJ/mol. Besides, no significant differences were observed in the compositional distribution from the EPMA results between the non-irradiated and the irradiated catalyst. However, the irradiated catalyst showed much more activity because of larger specific surface area of the catalyst and surface area of the precious metals. It showed that  -ray irradiation up to 1.0 MGy can increase activity of the catalyst.
-ray irradiation up to 1.0 MGy can increase activity of the catalyst.
 O(111) surfaces; The Case of NO dissociation
O(111) surfaces; The Case of NO dissociationKishi, Hirofumi*; Padama, A. A. B.*; Arevalo, R. L.*; Moreno, J. L. V.*; Kasai, Hideaki*; Taniguchi, Masashi*; Uenishi, Mari*; Tanaka, Hirohisa*; Nishihata, Yasuo
Journal of Physics; Condensed Matter, 24(26), p.262001_1 - 262001_5, 2012/07
Times Cited Count:9 Percentile:38.32(Physics, Condensed Matter)no abstracts in English
 O(111) surfaces; A Density functional theory based study
O(111) surfaces; A Density functional theory based studyPadama, A. A. B.*; Kishi, Hirofumi*; Arevalo, R. L.*; Moreno, J. L. V.*; Kasai, Hideaki*; Taniguchi, Masashi*; Uenishi, Mari*; Tanaka, Hirohisa*; Nishihata, Yasuo
Journal of Physics; Condensed Matter, 24(17), p.175005_1 - 175005_6, 2012/05
Times Cited Count:38 Percentile:78.54(Physics, Condensed Matter)no abstracts in English
Matsumura, Daiju; Nishihata, Yasuo; Mizuki, Junichiro; Taniguchi, Masashi*; Uenishi, Mari*; Tanaka, Hirohisa*
Journal of Applied Physics, 107(12), p.124319_1 - 124319_5, 2010/06
Times Cited Count:19 Percentile:60.14(Physics, Applied)The structural transformation of a Pd-perovskite automotive catalyst, LaFe Pd
Pd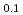 O
O , which has a high catalytic activity during aging, was studied by in situ time-resolved dispersive X-ray absorption fine structure spectroscopy at 473-773 K. An Al
, which has a high catalytic activity during aging, was studied by in situ time-resolved dispersive X-ray absorption fine structure spectroscopy at 473-773 K. An Al O
O -based conventional catalyst was also studied. In a reductive atmosphere, both catalysts showed similar temperature dependences of structural transformation from an oxide to a metal. However, different temperature dependence was observed in an oxidative atmosphere. A faster response in the structural change was observed in the Pd-perovskite catalyst than in the Pd/Al
-based conventional catalyst was also studied. In a reductive atmosphere, both catalysts showed similar temperature dependences of structural transformation from an oxide to a metal. However, different temperature dependence was observed in an oxidative atmosphere. A faster response in the structural change was observed in the Pd-perovskite catalyst than in the Pd/Al O
O catalyst. It was revealed that Pd-perovskite shows a considerably fast structural change to the oxidized state via the movement of Pd atoms into the perovskite crystal, in comparison with Pd/Al
catalyst. It was revealed that Pd-perovskite shows a considerably fast structural change to the oxidized state via the movement of Pd atoms into the perovskite crystal, in comparison with Pd/Al O
O showing two-step structural change for making PdO.
showing two-step structural change for making PdO.
 under redox atmosphere and CO/NO catalytic reaction studied by dispersive XAFS
under redox atmosphere and CO/NO catalytic reaction studied by dispersive XAFSMatsumura, Daiju; Okajima, Yuka; Nishihata, Yasuo; Mizuki, Junichiro; Taniguchi, Masashi*; Uenishi, Mari*; Tanaka, Hirohisa*
Journal of Physics; Conference Series, 190, p.012154_1 - 012154_6, 2009/11
Times Cited Count:11 Percentile:89.88(Physics, Condensed Matter)Pd/LaFeO is known to keep the metal particle size small even after the long time redox processes because Pd atoms make complex oxide with LaFeO
is known to keep the metal particle size small even after the long time redox processes because Pd atoms make complex oxide with LaFeO perovskite-type crystal under the oxidative atmosphere. We observed the local structure of Pd atoms by dispersive optics from the viewpoint of dynamical structure change of Pd during metal-oxide change and CO-NO catalytic reaction. It was recognized that, under the reductive atmosphere, Pd atoms show similar speed of movement from oxide to metal state both on LaFeO
perovskite-type crystal under the oxidative atmosphere. We observed the local structure of Pd atoms by dispersive optics from the viewpoint of dynamical structure change of Pd during metal-oxide change and CO-NO catalytic reaction. It was recognized that, under the reductive atmosphere, Pd atoms show similar speed of movement from oxide to metal state both on LaFeO and Al
and Al O
O . However, under the oxidative atmosphere, Pd atoms on LaFeO
. However, under the oxidative atmosphere, Pd atoms on LaFeO show faster movement from metal to oxide state with single reaction step than those on Al
show faster movement from metal to oxide state with single reaction step than those on Al O
O with two-step oxidation. In addition, many differences in structure and shape between Pd particles on LaFeO
with two-step oxidation. In addition, many differences in structure and shape between Pd particles on LaFeO and Al
and Al O
O are observed.
are observed.
 perovskite automotive catalyst by high-speed analysis with in situ DXAFS
perovskite automotive catalyst by high-speed analysis with in situ DXAFSUenishi, Mari*; Tanaka, Hirohisa*; Taniguchi, Masashi*; Tan, Isao*; Nishihata, Yasuo; Mizuki, Junichiro; Kobayashi, Tetsuhiko*
Catalysis Communications, 9(2), p.311 - 314, 2008/02
Times Cited Count:33 Percentile:62.75(Chemistry, Physical)In the LaFePdO perovskite catalyst, redox fluctuations of the exhaust gas suppress the growth of particles of precious metal by causing Pd to move in and out of the perovskite crystal. To observe the real movements of Pd directly, the time evolution of local structure around Pd by redox fluctuations was investigated by in situ energy-dispersive X-ray absorption fine-structure (DXAFS) analysis with a 10ms resolution. We proved that the change in structure of Pd is sufficiently fast to respond to the control frequency (1-4Hz) of an actual gasoline engine and that the Pd particles that segregate out are extremely fine.
perovskite catalyst, redox fluctuations of the exhaust gas suppress the growth of particles of precious metal by causing Pd to move in and out of the perovskite crystal. To observe the real movements of Pd directly, the time evolution of local structure around Pd by redox fluctuations was investigated by in situ energy-dispersive X-ray absorption fine-structure (DXAFS) analysis with a 10ms resolution. We proved that the change in structure of Pd is sufficiently fast to respond to the control frequency (1-4Hz) of an actual gasoline engine and that the Pd particles that segregate out are extremely fine.
Tanaka, Hirohisa*; Taniguchi, Masashi*; Uenishi, Mari*; Kajita, Nobuhiko*; Tan, Isao*; Nishihata, Yasuo; Mizuki, Junichiro; Narita, Keiichi*; Kimura, Mareo*; Kaneko, Kimiyoshi*
Angewandte Chemie; International Edition, 45(36), p.5998 - 6002, 2006/09
Times Cited Count:182 Percentile:94.91(Chemistry, Multidisciplinary)no abstracts in English
Tanaka, Hirohisa*; Uenishi, Mari*; Taniguchi, Masashi*; Tan, Isao*; Narita, Keiichi*; Kimura, Mareo*; Kaneko, Kimiyoshi*; Nishihata, Yasuo; Mizuki, Junichiro
Catalysis Today, 117(1-3), p.321 - 328, 2006/09
Times Cited Count:200 Percentile:98.01(Chemistry, Applied)no abstracts in English
Tanaka, Hirohisa*; Tan, Isao*; Uenishi, Mari*; Taniguchi, Masashi*; Nishihata, Yasuo; Mizuki, Junichiro
Key Engineering Materials, 317-318, p.827 - 832, 2006/08
no abstracts in English
Tan, Isao*; Taniguchi, Masashi*; Tanaka, Hirohisa*; Uenishi, Mari*; Kajita, Nobuhiko*; Nishihata, Yasuo; Mizuki, Junichiro; Niihara, Koichi*
Key Engineering Materials, 317-318, p.833 - 836, 2006/08
no abstracts in English
 perovskite automotive catalyst having a self-regenerative function
perovskite automotive catalyst having a self-regenerative functionTanaka, Hirohisa*; Tan, Isao*; Uenishi, Mari*; Taniguchi, Masashi*; Kimura, Mareo*; Nishihata, Yasuo; Mizuki, Junichiro
Journal of Alloys and Compounds, 408-412, p.1071 - 1077, 2006/02
Times Cited Count:51 Percentile:88.55(Chemistry, Physical)no abstracts in English
Naito, Kazuya*; Tanaka, Hirohisa*; Taniguchi, Masashi*; Uenishi, Mari*; Tan, Isao*; Kajita, Nobuhiko*; Takahashi, Ichiro*; Suzuki, Hiromasa*; Narita, Keiichi*; Hirai, Akimasa*; et al.
SAE 2006 World Congress & Exhibition Technical Papers, 8 Pages, 2006/00
no abstracts in English