Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 He spin filter at a pulsed neutron source
He spin filter at a pulsed neutron sourceOkudaira, Takuya; Ueda, Yuki; Hiroi, Kosuke; Motokawa, Ryuhei; Inamura, Yasuhiro; Takata, Shinichi; Oku, Takayuki; Suzuki, Junichi*; Takahashi, Shingo*; Endo, Hitoshi*; et al.
Journal of Applied Crystallography, 54(2), p.548 - 556, 2021/04
Times Cited Count:2 Percentile:33.1(Chemistry, Multidisciplinary)Neutron polarization analysis (NPA) for small-angle neutron scattering (SANS) experiments using a pulsed neutron source was successfully achieved by applying a  He spin filter as a spin analyzer for the scattered neutrons. The
He spin filter as a spin analyzer for the scattered neutrons. The  He spin filter covers a sufficient solid angle for performing SANS experiments, and the relaxation time of the
He spin filter covers a sufficient solid angle for performing SANS experiments, and the relaxation time of the  He polarization is sufficient for continuous use over a few days, thus reaching the typical duration required for a complete set of SANS experiments. Although accurate evaluation of the incoherent neutron scattering, which is predominantly attributable to hydrogen atoms in samples, is practically difficult using calculations based on the sample elemental composition, the developed NPA approach with consideration of the influence of multiple neutron scattering enabled reliable decomposition of the SANS intensity distribution into the coherent and incoherent scattering components. To date, NPA has not been well established as a standard technique for SANS experiments at pulsed neutron sources. This work is anticipated to greatly contribute to the accurate determination of the coherent neutron scattering component for scatterers in various types of organic sample systems in SANS experiments at J-PARC.
He polarization is sufficient for continuous use over a few days, thus reaching the typical duration required for a complete set of SANS experiments. Although accurate evaluation of the incoherent neutron scattering, which is predominantly attributable to hydrogen atoms in samples, is practically difficult using calculations based on the sample elemental composition, the developed NPA approach with consideration of the influence of multiple neutron scattering enabled reliable decomposition of the SANS intensity distribution into the coherent and incoherent scattering components. To date, NPA has not been well established as a standard technique for SANS experiments at pulsed neutron sources. This work is anticipated to greatly contribute to the accurate determination of the coherent neutron scattering component for scatterers in various types of organic sample systems in SANS experiments at J-PARC.
Miura, Daisuke*; Kumada, Takayuki; Sekine, Yurina; Motokawa, Ryuhei; Nakagawa, Hiroshi; Oba, Yojiro; Ohara, Takashi; Takata, Shinichi; Hiroi, Kosuke; Morikawa, Toshiaki*; et al.
Journal of Applied Crystallography, 54(2), p.454 - 460, 2021/04
Times Cited Count:1 Percentile:17.63(Chemistry, Multidisciplinary)We developed a spin-contrast-variation neutron powder diffractometry technique that extracts the structure factor of hydrogen atoms, namely, the contribution of hydrogen atoms to a crystal structure factor. Crystals of L-glutamic acid were dispersed in a dpolystyrene matrix containing 4-methacryloyloxy-2,2,6,6,-tetramethyl-1-piperidinyloxy (TEMPO methacrylate) to polarize their proton spins dynamically. The intensities of the diffraction peaks of the sample changed according to the proton polarization, and the structure factor of the hydrogen atoms was extracted from the proton-polarization dependent intensities. This technique is expected to enable analyses of the structures of hydrogen-containing materials that are difficult to determine with conventional powder diffractometry.
Kitazato, Kohei*; Milliken, R. E.*; Iwata, Takahiro*; Abe, Masanao*; Otake, Makiko*; Matsuura, Shuji*; Takagi, Yasuhiko*; Nakamura, Tomoki*; Hiroi, Takahiro*; Matsuoka, Moe*; et al.
Nature Astronomy (Internet), 5(3), p.246 - 250, 2021/03
Times Cited Count:43 Percentile:96.93(Astronomy & Astrophysics)Here we report observations of Ryugu's subsurface material by the Near-Infrared Spectrometer (NIRS3) on the Hayabusa2 spacecraft. Reflectance spectra of excavated material exhibit a hydroxyl (OH) absorption feature that is slightly stronger and peak-shifted compared with that observed for the surface, indicating that space weathering and/or radiative heating have caused subtle spectral changes in the uppermost surface. However, the strength and shape of the OH feature still suggests that the subsurface material experienced heating above 300  C, similar to the surface. In contrast, thermophysical modeling indicates that radiative heating does not increase the temperature above 200
C, similar to the surface. In contrast, thermophysical modeling indicates that radiative heating does not increase the temperature above 200  C at the estimated excavation depth of 1 m, even if the semimajor axis is reduced to 0.344 au. This supports the hypothesis that primary thermal alteration occurred due to radiogenic and/or impact heating on Ryugu's parent body.
C at the estimated excavation depth of 1 m, even if the semimajor axis is reduced to 0.344 au. This supports the hypothesis that primary thermal alteration occurred due to radiogenic and/or impact heating on Ryugu's parent body.
Narita, Hirokazu*; Kasuya, Ryo*; Suzuki, Tomoya*; Motokawa, Ryuhei; Tanaka, Mikiya*
Encyclopedia of Inorganic and Bioinorganic Chemistry (Internet), 28 Pages, 2020/12
Suzuki, Ryohei*; Tani, Yukinori*; Naito, Hirotaka*; Miyata, Naoyuki*; Tanaka, Kazuya
Catalysts, 10(1), p.44_1 - 44_15, 2020/01
Times Cited Count:10 Percentile:40.35(Chemistry, Physical)We prepared biogenic Mn oxides (BMOs) using 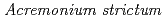 strain KR21-2, and subsequently conducted single or repeated treatment experiments in Cr(NO
strain KR21-2, and subsequently conducted single or repeated treatment experiments in Cr(NO )
) at pH6.0. Under aerobic conditions, newly formed BMOs exhibited a rapid production of Cr(VI) without a significant release of Mn(II), demonstrating that newly formed BMO mediates a catalytic oxidation of Cr(III) with a self-regeneration step of reduced Mn. In anaerobic solution, newly formed BMOs showed a cessation of Cr(III) oxidation in the early stage of the reaction, and subsequently had a much smaller Cr(VI) production with significant release of reduced Mn(II).
at pH6.0. Under aerobic conditions, newly formed BMOs exhibited a rapid production of Cr(VI) without a significant release of Mn(II), demonstrating that newly formed BMO mediates a catalytic oxidation of Cr(III) with a self-regeneration step of reduced Mn. In anaerobic solution, newly formed BMOs showed a cessation of Cr(III) oxidation in the early stage of the reaction, and subsequently had a much smaller Cr(VI) production with significant release of reduced Mn(II).
Narita, Hirokazu*; Maeda, Motoki*; Tokoro, Chiharu*; Suzuki, Tomoya*; Tanaka, Mikiya*; Motokawa, Ryuhei; Shiwaku, Hideaki; Yaita, Tsuyoshi
Analytical Sciences, 33(11), p.1305 - 1309, 2017/11
Times Cited Count:11 Percentile:40.7(Chemistry, Analytical)Hisamatsu, Shugo; Suzuki, Shinichi; Komoto, Shigeo*; Kishikawa, Keiki*; Yamamoto, Yusuke*; Motokawa, Ryuhei; Yaita, Tsuyoshi
Tetrahedron, 73(27-28), p.3993 - 3998, 2017/07
Times Cited Count:15 Percentile:57.79(Chemistry, Organic)Narita, Hirokazu*; Suzuki, Tomoya*; Motokawa, Ryuhei
Nihon Kinzoku Gakkai-Shi, 81(4), p.157 - 167, 2017/04
Times Cited Count:17 Percentile:62.95(Metallurgy & Metallurgical Engineering)Motokawa, Ryuhei; Kobayashi, Toru; Endo, Hitoshi*; Ikeda, Takashi; Yaita, Tsuyoshi; Suzuki, Shinichi; Narita, Hirokazu*; Akutsu, Kazuhiro*; Heller, W. T.*
Journal of Nuclear Science and Technology, 53(8), p.1205 - 1211, 2016/08
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology)Motokawa, Ryuhei; Endo, Hitoshi*; Yokoyama, Shingo*; Ogawa, Hiroki*; Kobayashi, Toru; Suzuki, Shinichi; Yaita, Tsuyoshi
Langmuir, 30(50), p.15127 - 15134, 2014/12
Times Cited Count:27 Percentile:62.29(Chemistry, Multidisciplinary) -isopropylacrylamide) hydrogels
-isopropylacrylamide) hydrogelsMiyamoto, Nobuyoshi*; Shimasaki, Kotaro*; Yamamoto, Kosuke*; Shintate, Morio*; Kamachi, Yuichiro*; Bastakoti, B. P.*; Suzuki, Norihiro*; Motokawa, Ryuhei; Yamauchi, Yusuke*
Chemistry; A European Journal, 20(46), p.14955 - 14958, 2014/11
Times Cited Count:18 Percentile:49.6(Chemistry, Multidisciplinary)Motokawa, Ryuhei; Endo, Hitoshi*; Yokoyama, Shingo*; Nishitsuji, Shotaro*; Kobayashi, Toru; Suzuki, Shinichi; Yaita, Tsuyoshi
Scientific Reports (Internet), 4, p.6585_1 - 6585_6, 2014/10
Times Cited Count:36 Percentile:70.26(Multidisciplinary Sciences)Awual, M. R.; Kobayashi, Toru; Shiwaku, Hideaki; Miyazaki, Yuji; Motokawa, Ryuhei; Suzuki, Shinichi; Okamoto, Yoshihiro; Yaita, Tsuyoshi
Chemical Engineering Journal, 225, p.558 - 566, 2013/06
Times Cited Count:196 Percentile:98.69(Engineering, Environmental)Awual, M. R.; Kobayashi, Toru; Miyazaki, Yuji; Motokawa, Ryuhei; Shiwaku, Hideaki; Suzuki, Shinichi; Okamoto, Yoshihiro; Yaita, Tsuyoshi
Journal of Hazardous Materials, 252-253, p.313 - 320, 2013/05
Times Cited Count:183 Percentile:97.66(Engineering, Environmental) -butyl phosphate/
-butyl phosphate/ -octane mixtures by X-ray and neutron scattering in a wide
-octane mixtures by X-ray and neutron scattering in a wide 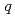 range
rangeMotokawa, Ryuhei; Suzuki, Shinichi; Ogawa, Hiroki*; Antonio, M. R.*; Yaita, Tsuyoshi
Journal of Physical Chemistry B, 116(4), p.1319 - 1327, 2012/02
Times Cited Count:36 Percentile:65.56(Chemistry, Physical)Koizumi, Satoshi; Iwase, Hiroki; Suzuki, Junichi; Oku, Takayuki; Motokawa, Ryuhei; Sasao, Hajime*; Tanaka, Hirokazu; Yamaguchi, Daisuke; Shimizu, Hirohiko; Hashimoto, Takeji
Physica B; Condensed Matter, 385-386(2), p.1000 - 1006, 2006/11
Times Cited Count:39 Percentile:80.68(Physics, Condensed Matter)no abstracts in English
Suzuki, Shinichi; Yaita, Tsuyoshi; Motokawa, Ryuhei; Koizumi, Satoshi; Okamoto, Yoshihiro; Shiwaku, Hideaki
no journal, ,
no abstracts in English
Suzuki, Shinichi; Motokawa, Ryuhei; Okamoto, Yoshihiro; Shiwaku, Hideaki; Yaita, Tsuyoshi
no journal, ,
no abstracts in English
 /tributylphosphine-octane; Small-angle scattering study of the third phase formation
/tributylphosphine-octane; Small-angle scattering study of the third phase formationMotokawa, Ryuhei; Suzuki, Shinichi; Yaita, Tsuyoshi
no journal, ,
no abstracts in English
 -butyl phosphate and zirconium ion in extracted solution
-butyl phosphate and zirconium ion in extracted solutionMotokawa, Ryuhei; Suzuki, Shinichi; Yaita, Tsuyoshi
no journal, ,
In the solvent extraction of Zr(NO )
) -HNO
-HNO /Octane-Tri-
/Octane-Tri- -butyl phosphate (TBP), zirconic ions (Zr
-butyl phosphate (TBP), zirconic ions (Zr ) in aqueous phase are extracted to organic phase with involving a complex formation among Zr
) in aqueous phase are extracted to organic phase with involving a complex formation among Zr , NO
, NO , and TBP, i.e., Zr(NO
, and TBP, i.e., Zr(NO )
) (TBP)
(TBP) . In this solvent extraction process the organic phase is separated into two organic phases: light organic phase and heavy organic phase, which is in particular called as the third phase, when the solution temperature deceases over a critical temperature. In this study, we investigated the temperature dependence of microscopic structure, formed by TBP, Zr
. In this solvent extraction process the organic phase is separated into two organic phases: light organic phase and heavy organic phase, which is in particular called as the third phase, when the solution temperature deceases over a critical temperature. In this study, we investigated the temperature dependence of microscopic structure, formed by TBP, Zr , and NO
, and NO , in diluent by employing small-angle neutron and X-ray scattering (SANS and SAXS) techniques. As a result, we found that the correlation length of the thermal concentration fluctuation of the organic phase strongly contributes to the third phase formation with varying temperature.
, in diluent by employing small-angle neutron and X-ray scattering (SANS and SAXS) techniques. As a result, we found that the correlation length of the thermal concentration fluctuation of the organic phase strongly contributes to the third phase formation with varying temperature.