Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Takahashi, Masamitsu; Kozu, Miwa*; Sasaki, Takuo
Japanese Journal of Applied Physics, 55(4S), p.04EJ04_1 - 04EJ04_4, 2016/04
Times Cited Count:5 Percentile:24.79(Physics, Applied)Suzuki, Hidetoshi*; Nakata, Yuka*; Takahashi, Masamitsu; Ikeda, Kazuma*; Oshita, Yoshio*; Morohara, Osamu*; Geka, Hirotaka*; Moriyasu, Yoshitaka*
AIP Advances (Internet), 6(3), p.035303_1 - 035303_6, 2016/03
Times Cited Count:4 Percentile:19.99(Nanoscience & Nanotechnology) synchrotron X-ray diffraction
synchrotron X-ray diffractionSasaki, Takuo; Ishikawa, Fumitaro*; Takahashi, Masamitsu
Applied Physics Letters, 108(1), p.012102_1 - 012102_5, 2016/01
Times Cited Count:4 Percentile:14.94(Physics, Applied) X-ray diffraction
X-ray diffractionShimomura, Kenichi*; Suzuki, Hidetoshi*; Sasaki, Takuo; Takahashi, Masamitsu; Oshita, Yoshio*; Kamiya, Itaru*
Journal of Applied Physics, 118(18), p.185303_1 - 185303_7, 2015/11
Times Cited Count:9 Percentile:38.07(Physics, Applied)Takahashi, Masamitsu; Kozu, Miwa*; Sasaki, Takuo; Hu, W.*
Crystal Growth & Design, 15(10), p.4979 - 4985, 2015/10
Times Cited Count:14 Percentile:69.21(Chemistry, Multidisciplinary) synchrotron X-ray diffraction
synchrotron X-ray diffractionSasaki, Takuo; Takahashi, Masamitsu
Nihon Kessho Seicho Gakkai-Shi, 42(3), p.210 - 217, 2015/10
Takahashi, Masamitsu
Nihon Kessho Seicho Gakkai-Shi, 42(3), p.201 - 209, 2015/10
 three-dimensional X-ray reciprocal-space mapping of InGaAs multilayer structures grown on GaAs(001) by MBE
three-dimensional X-ray reciprocal-space mapping of InGaAs multilayer structures grown on GaAs(001) by MBESasaki, Takuo; Takahashi, Masamitsu; Suzuki, Hidetoshi*; Oshita, Yoshio*; Yamaguchi, Masafumi*
Journal of Crystal Growth, 425, p.13 - 15, 2015/09
Times Cited Count:4 Percentile:29.54(Crystallography) Co
Co O
O film
filmHu, W.*; Hayashi, Koichi*; Fukumura, Tomoteru*; Akagi, Kazuto*; Tsukada, Masaru*; Happo, Naohisa*; Hosokawa, Shinya*; Owada, Kenji; Takahashi, Masamitsu; Suzuki, Motohiro*; et al.
Applied Physics Letters, 106(22), p.222403_1 - 222403_5, 2015/06
Times Cited Count:39 Percentile:81.57(Physics, Applied)Biermanns, A.*; Dimakis, E.*; Davydok, A.*; Sasaki, Takuo; Geelhaar, L.*; Takahashi, Masamitsu; Pietsch, U.*
Nano Letters, 14(12), p.6878 - 6883, 2014/12
Times Cited Count:27 Percentile:72.71(Chemistry, Multidisciplinary)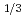 Nb
Nb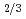 )O
)O
Hu, W.*; Hayashi, Koichi*; Owada, Kenji; Chen, J.*; Happo, Naohisa*; Hosokawa, Shinya*; Takahashi, Masamitsu; Bokov, A.*; Ye, Z.-G*
Physical Review B, 89(14), p.140103_1 - 140103_5, 2014/04
Times Cited Count:48 Percentile:85.62(Materials Science, Multidisciplinary) C discharge at RI facilities; A Comparison of collection and oxidation methods
C discharge at RI facilities; A Comparison of collection and oxidation methodsUeno, Yumi; Koarashi, Jun; Iwai, Yasunori; Sato, Junya; Takahashi, Teruhiko; Sawahata, Katsunori; Sekita, Tsutomu; Kobayashi, Makoto; Tsunoda, Masahiko; Kikuchi, Masamitsu
Hoken Butsuri, 49(1), p.39 - 44, 2014/03
The Japan Atomic Energy Agency has conducted a monthly monitoring of airborne  C discharge at the forth research building (RI facility) of the Tokai Research and Development Center. In the current monitoring,
C discharge at the forth research building (RI facility) of the Tokai Research and Development Center. In the current monitoring,  C, which exists in various chemical forms in airborne effluent, is converted into
C, which exists in various chemical forms in airborne effluent, is converted into  CO
CO with CuO catalyst and then collected using monoethanolamine (MEA) as CO
with CuO catalyst and then collected using monoethanolamine (MEA) as CO absorbent. However, this collection method has some issues on safety management because the CuO catalyst requires a high heating temperature (600
absorbent. However, this collection method has some issues on safety management because the CuO catalyst requires a high heating temperature (600 C) to ensure a high oxidation efficiency and the MEA is specified as a poisonous and deleterious substance. To establish a safer, manageable and reliable method for monitoring airborne
C) to ensure a high oxidation efficiency and the MEA is specified as a poisonous and deleterious substance. To establish a safer, manageable and reliable method for monitoring airborne  C discharge, we examined collection methods that use different CO
C discharge, we examined collection methods that use different CO absorbents (MEA and Carbo-Sorb E) and oxidation catalysts (CuO, Pt/Alumina and Pd/ZrO
absorbents (MEA and Carbo-Sorb E) and oxidation catalysts (CuO, Pt/Alumina and Pd/ZrO ). The results showed 100% CO
). The results showed 100% CO collection efficiency of MEA during a 30-day sampling period under the condition tested. In contrast, Carbo-Sorb E was found to be unsuitable for the monthly-long CO
collection efficiency of MEA during a 30-day sampling period under the condition tested. In contrast, Carbo-Sorb E was found to be unsuitable for the monthly-long CO collection because of its high volatile nature. Among the oxidation catalysts, the Pd/ZrO
collection because of its high volatile nature. Among the oxidation catalysts, the Pd/ZrO showed the highest oxidation efficiency for CH
showed the highest oxidation efficiency for CH at a lower temperature.
at a lower temperature.
Sasaki, Takuo; Norman, A. G.*; Romero, M. J.*; Al-Jassim, M. M.*; Takahashi, Masamitsu; Kojima, Nobuaki*; Oshita, Yoshio*; Yamaguchi, Masafumi*
Physica Status Solidi (C), 10(11), p.1640 - 1643, 2013/11
Times Cited Count:4 Percentile:83.55(Physics, Applied)Nishi, Toshiaki*; Sasaki, Takuo; Ikeda, Kazuma*; Suzuki, Hidetoshi*; Takahashi, Masamitsu; Shimomura, Kenichi*; Kojima, Nobuaki*; Oshita, Yoshio*; Yamaguchi, Masafumi*
AIP Conference Proceedings 1556, p.14 - 17, 2013/09
Times Cited Count:0 Percentile:0.01(Energy & Fuels)Takahashi, Masamitsu; Nakata, Yuka*; Suzuki, Hidetoshi*; Ikeda, Kazuma*; Kozu, Miwa; Hu, W.; Oshita, Yoshio*
Journal of Crystal Growth, 378, p.34 - 36, 2013/09
Times Cited Count:5 Percentile:42.72(Crystallography)Takahashi, Masamitsu
Journal of Crystal Growth, 401, p.372 - 375, 2013/09
Times Cited Count:3 Percentile:21.49(Crystallography)Hu, W.; Takahashi, Masamitsu; Kozu, Miwa*; Nakata, Yuka*
Journal of Physics; Conference Series, 425(20), p.202010_1 - 202010_4, 2013/03
Times Cited Count:1 Percentile:53.29(Instruments & Instrumentation)Takahashi, Masamitsu
Journal of the Physical Society of Japan, 82(2), p.021011_1 - 021011_14, 2013/02
Times Cited Count:10 Percentile:53.63(Physics, Multidisciplinary)Hu, W.; Suzuki, Hidetoshi*; Sasaki, Takuo*; Kozu, Miwa*; Takahashi, Masamitsu
Journal of Applied Crystallography, 45(5), p.1046 - 1053, 2012/10
Times Cited Count:12 Percentile:72.05(Chemistry, Multidisciplinary)Takahashi, Masamitsu
Hyomen Kagaku, 33(9), p.507 - 512, 2012/09
Surface structures of GaAs(001) under growth conditions have been investigated using in situ X-ray diffraction. The atomic arrangements of GaAs(001) surface stabilized at an elevated temperature under as pressure were quantitatively determined in a molecular-beam epitaxy chamber integrated with an X-ray diffractometer. With the help of high angular resolution of synchrotron radiation, disordered structures appearing in the transition from (2 4) to other phases were clarified. Energy tunability of synchrotron X-rays allowed for element-specific analysis of the c(4
4) to other phases were clarified. Energy tunability of synchrotron X-rays allowed for element-specific analysis of the c(4 4) structure, providing direct evidence for Ga-As heterodimer formation.
4) structure, providing direct evidence for Ga-As heterodimer formation.