Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Cs
CsUchiyama, Yusuke*; Tokunaga, Natsuki*; Azuma, Kohei*; Kamidaira, Yuki; Tsumune, Daisuke*; Iwasaki, Toshiki*; Yamada, Masatoshi*; Tateda, Yutaka*; Ishimaru, Takashi*; Ito, Yukari*; et al.
Science of the Total Environment, 816, p.151573_1 - 151573_13, 2022/04
Times Cited Count:7 Percentile:71.43(Environmental Sciences)no abstracts in English
 trapped and polarized in a CO
trapped and polarized in a CO matrix
matrixYamakawa, Koichiro; Ishibashi, Atsuki*; Namiyoshi, Toshinobu*; Azuma, Yuichi*; Arakawa, Ichiro*
Physical Review B, 102(4), p.041401_1 - 041401_5, 2020/07
Times Cited Count:2 Percentile:12.54(Materials Science, Multidisciplinary)Uchiyama, Yusuke*; Azuma, Kohei*; Odani, Sachika*; Iwasaki, Toshiki*; Tsumune, Daisuke*; Kamidaira, Yuki; Shimizu, Yasuyuki*; Onda, Yuichi*
Doboku Gakkai Rombunshu, B2 (Kaigan Kogaku) (Internet), 73(2), p.I_685 - I_690, 2017/10
no abstracts in English
 as the origin of volume collapse
as the origin of volume collapseYu, R.*; Hojo, Hajime*; Watanuki, Tetsu; Mizumaki, Masaichiro*; Mizokawa, Takashi*; Oka, Kengo*; Kim, H.*; Machida, Akihiko; Sakaki, Koji*; Nakamura, Yumiko*; et al.
Journal of the American Chemical Society, 137(39), p.12719 - 12728, 2015/10
Times Cited Count:34 Percentile:69.92(Chemistry, Multidisciplinary)no abstracts in English
 induced by intermetallic charge transfer
induced by intermetallic charge transferAzuma, Masaki*; Chen, W.*; Seki, Hayato*; Czapski, M.*; Olga, S.*; Oka, Kengo*; Mizumaki, Masaichiro*; Watanuki, Tetsu; Ishimatsu, Naoki*; Kawamura, Naomi*; et al.
Nature Communications (Internet), 2, p.347_1 - 347_5, 2011/06
Times Cited Count:359 Percentile:99.08(Multidisciplinary Sciences)no abstracts in English
 Mn
Mn O
O (NO
(NO ) oxynitrate comprising
) oxynitrate comprising 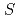 =3/2 honeycomb lattice
=3/2 honeycomb latticeSmirnova, O.*; Azuma, Masaki*; Kumada, Nobuhiro*; Kusano, Yoshihiro*; Matsuda, Masaaki; Shimakawa, Yuichi*; Takei, Takahiro*; Yonesaki, Yoshinori*; Kinomura, Nobukazu*
Journal of the American Chemical Society, 131(23), p.8313 - 8317, 2009/05
Times Cited Count:125 Percentile:91.9(Chemistry, Multidisciplinary)Neutron diffraction and FT-IR analysis revealed that the novel oxynitrate Bi Mn
Mn O
O (NO
(NO ) prepared by hydrothermal synthesis is of a new structural type including flat NO
) prepared by hydrothermal synthesis is of a new structural type including flat NO layers alternating with blocks of two PbSb
layers alternating with blocks of two PbSb O
O -like layers. Mn
-like layers. Mn (
(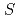 =3/2) forms a regular honeycomb lattice, and magnetic susceptibility data indicated two-dimensional magnetism. Despite its Weiss constant of
=3/2) forms a regular honeycomb lattice, and magnetic susceptibility data indicated two-dimensional magnetism. Despite its Weiss constant of 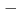 257K, no long-range ordering was observed down to 0.4K because of the magnetic frustration due to the competition between the nearest and the next-nearest antiferromagnetic interactions.
257K, no long-range ordering was observed down to 0.4K because of the magnetic frustration due to the competition between the nearest and the next-nearest antiferromagnetic interactions.
Azuma, Shunichi*; Ishii, Hiroshi*; Asai, Yasuhiro*; Kitagawa, Yuichi*; Wakita, Hiroshi*; Yamauchi, Tsuneo*; Asamori, Koichi
Geodynamics of Atotsugawa Fault System, p.173 - 179, 2007/00
no abstracts in English

Belik, A. A.*; Iikubo, Satoshi; Kodama, Katsuaki; Igawa, Naoki; Shamoto, Shinichi; Niitaka, Seiji*; Azuma, Masaki*; Shimakawa, Yuichi*; Takano, Mikio*; Izumi, Fujio*; et al.
Chemistry of Materials, 18(3), p.798 - 803, 2006/02
Times Cited Count:279 Percentile:98.62(Chemistry, Physical)The crystal and magnetic structures of polycrystalline BiCoO have been determined by the Rietveldmethod from neutron diffraction data measured at temperatures from 5 to 520 K. BiCoO
have been determined by the Rietveldmethod from neutron diffraction data measured at temperatures from 5 to 520 K. BiCoO (space groupP4mm; Z=1; a=3.72937(7)
(space groupP4mm; Z=1; a=3.72937(7)  and c=4.72382(15)
and c=4.72382(15)  at room temperature; tetragonality c/a=1.267) is isotypic with BaTiO
at room temperature; tetragonality c/a=1.267) is isotypic with BaTiO and PbTiO
and PbTiO in the whole temperature range. BiCoO
in the whole temperature range. BiCoO is an insulator with a Neeltemperature of 470 K. A possible model for antiferromagnetic order is proposed with a propagationvector of k=(1/2, 1/2, 0). In this model, magnetic moments of Co
is an insulator with a Neeltemperature of 470 K. A possible model for antiferromagnetic order is proposed with a propagationvector of k=(1/2, 1/2, 0). In this model, magnetic moments of Co ions are parallel to the c directionand align antiferromagnetically in the ab plane. The antiferromagnetic ab layers stack ferromagneticallyalong the c axis, forming a C-type antiferromagnetic structure. Refined magnetic moments at 5 and 300K are 3.24(2)
ions are parallel to the c directionand align antiferromagnetically in the ab plane. The antiferromagnetic ab layers stack ferromagneticallyalong the c axis, forming a C-type antiferromagnetic structure. Refined magnetic moments at 5 and 300K are 3.24(2)
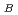 and 2.93(2)
and 2.93(2)
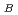 , respectively. The structure refinements revealed no deviation fromstoichiometry in BiCoO
, respectively. The structure refinements revealed no deviation fromstoichiometry in BiCoO . BiCoO
. BiCoO decomposed in air above 720 K to give Co
decomposed in air above 720 K to give Co O
O and sillenite-like Bi
and sillenite-like Bi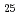 CoO
CoO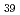 .
.

Mizumaki, Masaichiro*; Agui, Akane; Saito, Takashi*; Azuma, Masaki*; Shimakawa, Yuichi*; Takano, Mikio*; Uozumi, Takayuki*
no journal, ,
no abstracts in English