Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
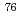 Br-
Br- -methyl-phenylalanine for tumor PET imaging
-methyl-phenylalanine for tumor PET imagingHanaoka, Hirofumi*; Ohshima, Yasuhiro; Suzuki, Yurika*; Yamaguchi, Aiko*; Watanabe, Shigeki; Uehara, Tomoya*; Nagamori, Shushi*; Kanai, Yoshikatsu*; Ishioka, Noriko; Tsushima, Yoshito*; et al.
Journal of Nuclear Medicine, 56(5), p.791 - 797, 2015/05
Times Cited Count:18 Percentile:62.37(Radiology, Nuclear Medicine & Medical Imaging)Chinone, Shumpei*; Hanaoka, Yasushi*; Tokuhiro, Koji*; Nakatsubo, Koichi*; Amano, Masayuki*; Hase, Yoshihiro; Tanaka, Atsushi; Narumi, Issei
JAEA-Review 2006-042, JAEA Takasaki Annual Report 2005, P. 90, 2007/02
no abstracts in English
 Re]Organorhenium-labeled antibody fragments with renal enzyme-cleavable linkage for low renal radioactivity levels
Re]Organorhenium-labeled antibody fragments with renal enzyme-cleavable linkage for low renal radioactivity levelsUehara, Tomoya*; Koike, Miho*; Nakata, Hideo*; Hanaoka, Hirofumi*; Iida, Yasuhiko*; Hashimoto, Kazuyuki; Akizawa, Hiromichi*; Endo, Keigo*; Arano, Yasushi*
Bioconjugate Chemistry, 18(1), p.190 - 198, 2007/01
Times Cited Count:20 Percentile:55.17(Biochemical Research Methods)Renal localization of radiolabeled antibody fragments constitutes a problem in targeted imaging and radiotherapy. To estimate the applicability of the molecular design to metallic radionuclides, [ Re]tricarbonyl(cyclopentadienylcarbonate)rhenium ([
Re]tricarbonyl(cyclopentadienylcarbonate)rhenium ([ Re]CpTR-COOH) was conjugated with maleoyl-glycyl-lysine to prepare [
Re]CpTR-COOH) was conjugated with maleoyl-glycyl-lysine to prepare [ Re]CpTR-GK. The cleavage of the glycyl-lysine linkage of the compound generates a glycine conjugate of [
Re]CpTR-GK. The cleavage of the glycyl-lysine linkage of the compound generates a glycine conjugate of [ Re]CpTR-Gly. [
Re]CpTR-Gly. [ Re]CpTR-GK was conjugated to thiolated Fab fragments to prepare [
Re]CpTR-GK was conjugated to thiolated Fab fragments to prepare [ Re]CpTR-GK-Fab. The biodistribution of radioactivity after injection of [
Re]CpTR-GK-Fab. The biodistribution of radioactivity after injection of [ Re]CpTR-GK-Fab was compared with that of [
Re]CpTR-GK-Fab was compared with that of [ Re]CpTR-Fab. [
Re]CpTR-Fab. [ Re]CpTR-GK-Fab exhibited significantly lower renal radioactivity levels than did [
Re]CpTR-GK-Fab exhibited significantly lower renal radioactivity levels than did [ Re]CpTR-Fab. The analysis of urine samples collected for 6 h postinjection of [
Re]CpTR-Fab. The analysis of urine samples collected for 6 h postinjection of [ Re]CpTR-GK-Fab showed that [
Re]CpTR-GK-Fab showed that [ Re]CpTR-Gly was the major radiometabolite. In tumor-bearing mice, [
Re]CpTR-Gly was the major radiometabolite. In tumor-bearing mice, [ Re]CpTR-GK-Fab significantly reduced renal radioactivity levels without impairing the radioactivity levels in tumor. These findings indicate that the molecular design of radioparmaceuticals labeled with metallic radionuclides can be useful by using a radiometal chelate of high inertness and by designing a radiometabolite of high urinary excretion when released from antibody fragments following cleavage of a glycyl-lysine linkage. This study also indicates that a change in chemical structure of a radiolabel attached to a glycyl-lysine linkage significantly affected enzymes involved in the hydrolysis reaction.
Re]CpTR-GK-Fab significantly reduced renal radioactivity levels without impairing the radioactivity levels in tumor. These findings indicate that the molecular design of radioparmaceuticals labeled with metallic radionuclides can be useful by using a radiometal chelate of high inertness and by designing a radiometabolite of high urinary excretion when released from antibody fragments following cleavage of a glycyl-lysine linkage. This study also indicates that a change in chemical structure of a radiolabel attached to a glycyl-lysine linkage significantly affected enzymes involved in the hydrolysis reaction.
Ogawa, Kazuma*; Mukai, Takahiro*; Arano, Yasushi*; Ono, Masahiro*; Hanaoka, Hirofumi*; Ishino, Seigo*; Hashimoto, Kazuyuki; Nishimura, Hiroshi*; Saji, Hideo*
Bioconjugate Chemistry, 16(4), p.751 - 757, 2005/07
Times Cited Count:62 Percentile:86.83(Biochemical Research Methods)Rhenium-186-1-hydroxyethylidene-1,1-diphosphonate ( Re-HEDP) has been used for the palliation of metastatic bone pain. Delayed blood clearance and high gastric uptake of radioactivity have been observed upon injection, due to the instability of
Re-HEDP) has been used for the palliation of metastatic bone pain. Delayed blood clearance and high gastric uptake of radioactivity have been observed upon injection, due to the instability of  Re-HEDP in vivo. In this study, on the basis of the concept of bifunctional radiopharmaceuticals, we designed a stable
Re-HEDP in vivo. In this study, on the basis of the concept of bifunctional radiopharmaceuticals, we designed a stable  Re-mercaptoacetylglycylglycylglycine (MAG3) complex-conjugated bisphosphonate (
Re-mercaptoacetylglycylglycylglycine (MAG3) complex-conjugated bisphosphonate ( Re-MAG3-HBP). After purification by HPLC,
Re-MAG3-HBP). After purification by HPLC,  Re-MAG3-HBP was synthesized with a radiochemical purity of over 95%. In biodistribution experiments, the radioactivity level of
Re-MAG3-HBP was synthesized with a radiochemical purity of over 95%. In biodistribution experiments, the radioactivity level of Re-MAG3-HBP in bone was significantly higher than that of
Re-MAG3-HBP in bone was significantly higher than that of  Re-HEDP. Blood clearance of
Re-HEDP. Blood clearance of  Re-MAG3-HBP was faster than that of
Re-MAG3-HBP was faster than that of  Re-HEDP. In addition, the gastric accumulation of
Re-HEDP. In addition, the gastric accumulation of  Re-MAG3-HBP radioactivity was lower than that of
Re-MAG3-HBP radioactivity was lower than that of  Re-HEDP. In conclusion,
Re-HEDP. In conclusion,  Re-MAG3-HBP is expected to be a useful radiopharmaceutical for the palliation of metastatic bone pain.
Re-MAG3-HBP is expected to be a useful radiopharmaceutical for the palliation of metastatic bone pain.
Ogawa, Kazuma*; Mukai, Takahiro*; Arano, Yasushi*; Hanaoka, Hirofumi*; Hashimoto, Kazuyuki; Nishimura, Hiroshi*; Saji, Hideo*
Journal of Labelled Compounds and Radiopharmaceuticals, 47(11), p.753 - 761, 2004/11
Times Cited Count:29 Percentile:61.59(Biochemical Research Methods)no abstracts in English
Chinone, Shumpei*; Hanaoka, Yasushi*; Tokuhiro, Koji*; Nakatsubo, Koichi*; Amano, Masayuki*; Hase, Yoshihiro; Tanaka, Atsushi; Narumi, Issei
no journal, ,
no abstracts in English
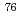 Br-labeled antibody fragments for reduction of non-target radioactivity
Br-labeled antibody fragments for reduction of non-target radioactivityHanaoka, Hirofumi*; Watanabe, Shigeki; Watanabe, Satoshi; Ohshima, Yasuhiro; Uehara, Tomoya*; Akizawa, Hiromichi*; Iida, Yasuhiko*; Ishioka, Noriko; Arano, Yasushi*; Endo, Keigo*
no journal, ,
no abstracts in English
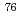 Br-labeled amino acid derivative for PET imaging of tumor
Br-labeled amino acid derivative for PET imaging of tumorHanaoka, Hirofumi*; Watanabe, Shigeki; Tominaga, Hideyuki*; Ohshima, Yasuhiro; Watanabe, Satoshi; Yamada, Keiichi*; Arano, Yasushi*; Ishioka, Noriko; Endo, Keigo*
no journal, ,
no abstracts in English
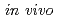 evaluation of
evaluation of 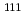 In labeled hippuric acid derivative with high urinary excretion
In labeled hippuric acid derivative with high urinary excretionSuzuki, Hiroyuki; Kanai, Ayaka*; Uehara, Tomoya*; Hanaoka, Hirofumi*; Arano, Yasushi*
no journal, ,
 At]astato-
At]astato- -methyl-L-phenylalanine (2-AAMP) as a novel radiopharmaceutical for internal radiotherapy
-methyl-L-phenylalanine (2-AAMP) as a novel radiopharmaceutical for internal radiotherapyOhshima, Yasuhiro; Suzuki, Hiroyuki*; Hanaoka, Hirofumi*; Watanabe, Shigeki; Watanabe, Satoshi; Watanabe, Naoyuki*; Tsushima, Yoshito*; Endo, Keigo*; Arano, Yasushi*; Ishioka, Noriko
no journal, ,
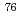 Br-bromo-
Br-bromo- -methyl-L-phenylalanine as a novel
-methyl-L-phenylalanine as a novel 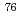 Br-labeled PET tracer
Br-labeled PET tracerOhshima, Yasuhiro; Hanaoka, Hirofumi*; Suzuki, Yurika*; Yamaguchi, Aiko*; Watanabe, Shigeki; Uehara, Tomoya*; Nagamori, Shushi*; Kanai, Yoshikatsu*; Ishioka, Noriko; Tsushima, Yoshito*; et al.
no journal, ,
no abstracts in English
 At labeled
At labeled  -methyl-L-phenylalanine
-methyl-L-phenylalanineSuzuki, Hiroyuki*; Ohshima, Yasuhiro; Hanaoka, Hirofumi*; Watanabe, Shigeki; Watanabe, Satoshi; Sasaki, Ichiro; Sakashita, Tetsuya; Arano, Yasushi*; Ishioka, Noriko
no journal, ,
no abstracts in English