Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kitatsuji, Yoshihiro; Okugaki, Tomohiko*; Kasuno, Megumi*; Kubota, Hiroki*; Maeda, Koji*; Kimura, Takaumi; Yoshida, Zenko; Kihara, Sorin*
Journal of Chemical Thermodynamics, 43(6), p.844 - 851, 2011/06
Times Cited Count:8 Percentile:30.19(Thermodynamics)Standard Gibbs energies for transfer ( G
G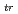
 ) of actinyl ions (AnO
) of actinyl ions (AnO
 ; z = 2 or 1; An: U, Np or Pu) between an aqueous solution and an organic solution were determined based on distribution method combined with voltammetry for ion transfer at the interface of two immiscible electrolyte solutions. The organic solutions examined were nitrobenzene, 1,2-dichloroethane, benzonitrile, acetophenone and 2-nitrophenyl octyl ether. Irrespective of the type of organic solutions,
; z = 2 or 1; An: U, Np or Pu) between an aqueous solution and an organic solution were determined based on distribution method combined with voltammetry for ion transfer at the interface of two immiscible electrolyte solutions. The organic solutions examined were nitrobenzene, 1,2-dichloroethane, benzonitrile, acetophenone and 2-nitrophenyl octyl ether. Irrespective of the type of organic solutions,  G
G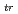
 of UO
of UO
 , NpO
, NpO
 and PuO
and PuO
 were nearly equal to each other and slightly larger than that of Mg
were nearly equal to each other and slightly larger than that of Mg . The
. The  G
G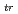
 of NpO
of NpO
 was extraordinary large compared with those of ordinary monovalent cations. The dependence of
was extraordinary large compared with those of ordinary monovalent cations. The dependence of  G
G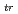
 of AnO
of AnO
 on the type of organic solutions was similar to that of H
on the type of organic solutions was similar to that of H or Mg
or Mg . The
. The  G
G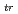
 of An
of An and An
and An were also discussed briefly.
were also discussed briefly.
Okugaki, Tomohiko*; Kitatsuji, Yoshihiro; Kasuno, Megumi*; Yoshizumi, Asuka*; Kubota, Hiroki*; Shibafuji, Yayoi*; Maeda, Koji*; Yoshida, Zenko; Kihara, Sorin*
Journal of Electroanalytical Chemistry, 629(1-2), p.50 - 56, 2009/04
A high performance electrochemical solvent extraction method was developed based on the quantitative transfer of ions from an aqueous, W, to an organic solution, O. It was realized by applying a potential difference between W and O. The cell was composed of a porous Teflon tube immersed with O, a Ag wire inserted into the tube, a Pt wire placed outside the tube. When W containing ions was forced to flow through the narrow gap between the tube and Ag wire, and potential was applied, a very rapid quantitative ion transfer was attained. When O containing extractant, more than 99% of U(VI) in W was extracted during the residence (e.g., 40 s) in the cell. The fundamental feature of the extraction system was investigated, taking into account the application of the system to the extraction of actinide, lanthanide, Sr or Cs ions. The use of a column electrode system connected before the extraction system was examined in order to adjust the oxidation state of the element to that desired.
 organic solution interface in chelate extraction
organic solution interface in chelate extractionUehara, Akihiro*; Kasuno, Megumi*; Okugaki, Tomohiko*; Kitatsuji, Yoshihiro; Shirai, Osamu*; Yoshida, Zenko; Kihara, Sorin*
Journal of Electroanalytical Chemistry, 604(2), p.115 - 124, 2007/06
The distribution ratio of a metal ion between an aqueous solution and an organic solution in solvent extraction with a chelating agent was evaluated by using physicochemical constants determined electrochemically such as standard Gibbs energies for transfers of the ions, the overall complex formation constants and the acid dissociation constants of the chelating agent. The distribution ratio thus evaluated agreed well with those determined by the distribution experiment.
Yoshizumi, Asuka*; Uehara, Akihiro*; Kasuno, Megumi*; Kitatsuji, Yoshihiro; Yoshida, Zenko; Kihara, Sorin*
Journal of Electroanalytical Chemistry, 581(2), p.275 - 283, 2005/08
no abstracts in English
 organic solution interface
organic solution interfaceUehara, Akihiro*; Yoshida, Zenko; Yoshida, Yumi*; Kitatsuji, Yoshihiro; Kasuno, Megumi*; Maeda, Koji*; Kihara, Sorin*
Journal of Electroanalytical Chemistry, 563(2), p.257 - 267, 2004/03
no abstracts in English
Kitatsuji, Yoshihiro; Okugaki, Tomohiko*; Kasuno, Megumi*; Maeda, Koji*; Kimura, Takaumi; Yoshida, Zenko; Kihara, Sorin*
no journal, ,
no abstracts in English