Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Bh in the
Bh in the  Cm(
Cm(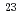 Na,5
Na,5 )
) Bh reaction and its decay properties
Bh reaction and its decay propertiesHaba, Hiromitsu*; Fan, F.*; Kaji, Daiya*; Kasamatsu, Yoshitaka*; Kikunaga, Hidetoshi*; Komori, Yukiko*; Kondo, Narumi*; Kudo, Hisaaki*; Morimoto, Koji*; Morita, Kosuke*; et al.
Physical Review C, 102(2), p.024625_1 - 024625_12, 2020/08
Times Cited Count:6 Percentile:58.52(Physics, Nuclear)Kudo, Atsunari; Kurabayashi, Kazuaki; Yanagibashi, Futoshi; Sasaki, Shunichi; Sato, Takehiko; Fujimoto, Ikuo; Obu, Tomoyuki
Proceedings of 2017 International Congress on Advances in Nuclear Power Plants (ICAPP 2017) (CD-ROM), 6 Pages, 2017/04
The Co-processing process is the extraction process to recover Pu/U mixed product solution with given Pu/U ratio for improving of nuclear proliferation resistance. In addition, Np is also recovered with U and Pu because Np is one of minor actinides and a long-lived radionuclide and Np has the extractability into TBP solvent. Development of its flowsheet achieves to decrease environmental effect of waste materials. The orientation of development about Co-processing process is to demonstrate of reprocessing the future spent fuels from a LWR, a LWR-MOX hybrid, and a FR-MOX with one cycle. We demonstrated by use of miniature reflux-type centrifugal contactors at the partitioning unit. The test conditions of the Pu/U ratio in the loaded solvents were 1%, 3%, and 5% considering the composition of spent fuels. We used the HAN as the reductant of Np (VI) for back extraction. The results of these tests were very good. We got the prospect of U, Pu, and Np Co-processing flowsheet.
Eichler, R.*; Asai, Masato; Brand, H.*; Chiera, N. M.*; Di Nitto, A.*; Dressler, R.*; D llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
EPJ Web of Conferences, 131, p.07005_1 - 07005_7, 2016/12
Times Cited Count:3 Percentile:72.98(Chemistry, Inorganic & Nuclear)In recent years gas-phase chemical studies assisted by physical pre-separation allowed for the productions and investigations of fragile single molecular species of superheavy elements. The latest highlight is the formation of very volatile hexacarbonyl compound of element 106, Sg(CO) . Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO)
. Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO) , W(CO)
, W(CO) , and Sg(CO)
, and Sg(CO) .
.
 and W(CO)
and W(CO)
Usoltsev, I.*; Eichler, R.*; Wang, Y.*; Even, J.*; Yakushev, A.*; Haba, Hiromitsu*; Asai, Masato; Brand, H.*; Di Nitto, A.*; D llmann, Ch. E.*; et al.
llmann, Ch. E.*; et al.
Radiochimica Acta, 104(3), p.141 - 151, 2016/03
Times Cited Count:31 Percentile:94.91(Chemistry, Inorganic & Nuclear)Conditions of the production and decomposition of hexacarbonyl complexes of short-lived Mo and W isotopes were investigated to study thermal stability of the heaviest group 6 hexacarbonyl complex Sg(CO) . A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO)
. A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO) and W(CO)
and W(CO) , indicating that the first bond dissociation energy of Sg(CO)
, indicating that the first bond dissociation energy of Sg(CO) could be determined with this technique.
could be determined with this technique.
 Nb and
Nb and 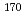 Ta for chemical studies of element 105, Db, using the GARIS gas-jet system
Ta for chemical studies of element 105, Db, using the GARIS gas-jet systemHuang, M.*; Haba, Hiromitsu*; Murakami, Masashi*; Asai, Masato; Kaji, Daiya*; Kanaya, Jumpei*; Kasamatsu, Yoshitaka*; Kikunaga, Hidetoshi*; Kikutani, Yuki*; Komori, Yukiko*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 304(2), p.845 - 849, 2015/05
Times Cited Count:3 Percentile:25.64(Chemistry, Analytical)A technique to utilize radioisotopes of Nb and Ta was developed for chemical studies of element 105, Db, by coupling a gas-jet transport system to the RIKEN gas-filled recoil ion separator (GARIS). The short-lived  Nb and
Nb and 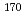 Ta were produced with nuclear reactions using a
Ta were produced with nuclear reactions using a  F beam whose energy was the same as that to produce
F beam whose energy was the same as that to produce  Db. Then, they were separated with GARIS and extracted to a chemistry laboratory with the gas-jet transport system. By changing only magnetic field of GARIS and inserting an energy degrader and a shutter for recoil ions, we could deliver the
Db. Then, they were separated with GARIS and extracted to a chemistry laboratory with the gas-jet transport system. By changing only magnetic field of GARIS and inserting an energy degrader and a shutter for recoil ions, we could deliver the  Nb and
Nb and 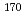 Ta to a chemistry device for
Ta to a chemistry device for  Db without changing other experimental conditions.
Db without changing other experimental conditions.
Even, J.*; Ackermann, D.*; Asai, Masato; Block, M.*; Brand, H.*; Di Nitto, A.*; D llmann, Ch. E.*; Eichler, R.*; Fan, F.*; Haba, Hiromitsu*; et al.
llmann, Ch. E.*; Eichler, R.*; Fan, F.*; Haba, Hiromitsu*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(3), p.2457 - 2466, 2015/03
Times Cited Count:15 Percentile:77.35(Chemistry, Analytical)Rapid In situ synthesis of metal carbonyl complexes has been demonstrated using short-lived isotopes produced in nuclear fission and fusion reactions. The short-lived isotopes with high recoil energy directly react with carbon-monoxides and form carbonyl complexes. Only highly volatile complexes were fast transported in a gas stream to counting and chemistry devices. Short-lived Mo, Tc, Ru, Rh, W, Re, Os, and Ir were found to form volatile carbonyl complexes, while no volataile complex of Hf and Ta were detected. This technique has been applied to a chemical investigation of the superheavy element Sg (atomic number 106), and will be applicable to various fields of nuclear science with short-lived transition metal isotopes.
Even, J.*; Yakushev, A.*; D llmann, Ch. E.*; Haba, Hiromitsu*; Asai, Masato; Sato, Tetsuya; Brand, H.*; Di Nitto, A.*; Eichler, R.*; Fan, F. L.*; et al.
llmann, Ch. E.*; Haba, Hiromitsu*; Asai, Masato; Sato, Tetsuya; Brand, H.*; Di Nitto, A.*; Eichler, R.*; Fan, F. L.*; et al.
Science, 345(6203), p.1491 - 1493, 2014/09
Times Cited Count:64 Percentile:83.15(Multidisciplinary Sciences)A new superheavy element complex, a seaborgium carbonyl, has been successfully synthesized, and its adsorption property has been studied using a cryo-thermochromatography and  -detection apparatus COMPACT. Nuclear reaction products of short-lived
-detection apparatus COMPACT. Nuclear reaction products of short-lived  Sg preseparated with a gas-filled recoil ion separator GARIS at RIKEN were directly injected into a gas cell filled with He/CO mixture gas, and chemical reaction products of volatile carbonyl complexes were trasported to COMPACT. The Sg carbonyl complex detected with COMPACT was found to be very volatile with adsorption enthalpy of
Sg preseparated with a gas-filled recoil ion separator GARIS at RIKEN were directly injected into a gas cell filled with He/CO mixture gas, and chemical reaction products of volatile carbonyl complexes were trasported to COMPACT. The Sg carbonyl complex detected with COMPACT was found to be very volatile with adsorption enthalpy of 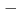 50 kJ/mol, from which we have concluded that this complex should be a Sg hexacarbonyl Sg(CO)
50 kJ/mol, from which we have concluded that this complex should be a Sg hexacarbonyl Sg(CO) . This is the first synthesis of organometallic compounds of transactinide elements for which only simple inorganic comounds have been synthesized so far.
. This is the first synthesis of organometallic compounds of transactinide elements for which only simple inorganic comounds have been synthesized so far.
 Db in the
Db in the  Cm(
Cm( F,5
F,5 )
) Db reaction and decay properties of
Db reaction and decay properties of  Db and
Db and 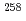 Lr
LrHaba, Hiromitsu*; Huang, M.*; Kaji, Daiya*; Kanaya, Jumpei*; Kudo, Yuki*; Morimoto, Koji*; Morita, Kosuke*; Murakami, Masashi*; Ozeki, Kazutaka*; Sakai, Ryutaro*; et al.
Physical Review C, 89(2), p.024618_1 - 024618_11, 2014/02
Times Cited Count:25 Percentile:82.19(Physics, Nuclear)Park, J.*; Takayama, Toshio*; Asano, Masaharu; Maekawa, Yasunari; Kudo, Kazuaki*; Takayama, Toshio*
Polymer, 54(17), p.4570 - 4577, 2013/08
Times Cited Count:6 Percentile:19.27(Polymer Science)Graft-type sulfonated polybenzimidazole was prepared through radiation-induced graft polymerization of styrenes into an alicyclic polybenzimidazole film and subsequent sulfonation. The alicyclic polybenzimidazole, ChPBI, was prepared from 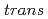 -1,4-cyclohexanedicarboxylic acid and 3,3'-diaminobenzidine using typical polycondensation. Anisotropic domains with a size of several tens of micrometers were found in the ChPBI films casted from LiCl-containing
-1,4-cyclohexanedicarboxylic acid and 3,3'-diaminobenzidine using typical polycondensation. Anisotropic domains with a size of several tens of micrometers were found in the ChPBI films casted from LiCl-containing  ,
, -dimethylacetamide. Irradiation of the ChPBI membranes with a 220 kGy dose of
-dimethylacetamide. Irradiation of the ChPBI membranes with a 220 kGy dose of  -rays created radical species with mean lifetimes of two days. The treatment of this membrane with a 50/50 (v/v) mixture of 1-propanol and styrene produced polystyrene graft chains. Sulfonation of the resulting grafted membrane with ClSOH
-rays created radical species with mean lifetimes of two days. The treatment of this membrane with a 50/50 (v/v) mixture of 1-propanol and styrene produced polystyrene graft chains. Sulfonation of the resulting grafted membrane with ClSOH OH occurred selectively on the polystyrene grafts. The sulfonated films showed proton conductivity on the order of 10
OH occurred selectively on the polystyrene grafts. The sulfonated films showed proton conductivity on the order of 10 to 10
to 10 S/cm with an ion exchange capacity between 2.1 and 2.9 mmol/g. SEM-EDX analysis of the membrane indicated the presence of macrophase separated domains up to 1
S/cm with an ion exchange capacity between 2.1 and 2.9 mmol/g. SEM-EDX analysis of the membrane indicated the presence of macrophase separated domains up to 1  m in diameter. The proton conductivity of the membrane did not decrease for 600 h at 120
m in diameter. The proton conductivity of the membrane did not decrease for 600 h at 120 C in liquid water.
C in liquid water.
 SO
SO /HNO
/HNO mixed solution
mixed solutionLi, Z.*; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Sato, Tetsuya; Sato, Nozomi; Kikuchi, Takahiro; Nagame, Yuichiro; Sch del, M.; Pershina, V.*; et al.
del, M.; Pershina, V.*; et al.
Radiochimica Acta, 100(3), p.157 - 164, 2012/03
Times Cited Count:14 Percentile:68.93(Chemistry, Inorganic & Nuclear) Sg in the
Sg in the  Cm(
Cm(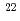 Ne,5
Ne,5 )
) Sg reaction and decay properties of two isomeric states in
Sg reaction and decay properties of two isomeric states in  Sg
SgHaba, Hiromitsu*; Kaji, Daiya*; Kudo, Yuki*; Morimoto, Koji*; Morita, Kosuke*; Ozeki, Kazutaka*; Sakai, Ryutaro*; Sumita, Takayuki*; Yoneda, Akira*; Kasamatsu, Yoshitaka*; et al.
Physical Review C, 85(2), p.024611_1 - 024611_11, 2012/02
Times Cited Count:53 Percentile:91.21(Physics, Nuclear)Two isomeric states in  Sg, i.e,
Sg, i.e,  Sg
Sg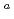 and
and  Sg
Sg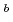 were produced in the
were produced in the  Cm(
Cm(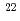 Ne,5
Ne,5 ) reaction. Decay properties of
) reaction. Decay properties of  Sg
Sg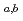 were investigated with a rotating-wheel apparatus for
were investigated with a rotating-wheel apparatus for  and spontaneous fission (SF) spectrometry under low background condition attained by a gas-jet transport system coupled to the RIKEN gas-filled recoil ion separator. Based on genetically correlated
and spontaneous fission (SF) spectrometry under low background condition attained by a gas-jet transport system coupled to the RIKEN gas-filled recoil ion separator. Based on genetically correlated  -
- (-
(- ) and
) and  -SF decay chains, 18 and 24 events were assigned to
-SF decay chains, 18 and 24 events were assigned to  Sg
Sg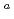 and
and  Sg
Sg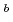 , respectively. The half-life and
, respectively. The half-life and  -particle energy of
-particle energy of  Sg
Sg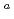 were measured to be
were measured to be 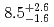 s and
s and 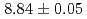 MeV, respectively, and those of
MeV, respectively, and those of  Sg
Sg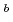 were
were 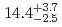 s and
s and 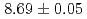 MeV.
MeV.
Ishii, Yasuo; Toyoshima, Atsushi; Tsukada, Kazuaki; Asai, Masato; Li, Z.*; Nagame, Yuichiro; Miyashita, Sunao*; Mori, Tomotaka*; Suganuma, Hideo*; Haba, Hiromitsu*; et al.
Bulletin of the Chemical Society of Japan, 84(9), p.903 - 911, 2011/09
Times Cited Count:18 Percentile:49.26(Chemistry, Multidisciplinary)The cation-exchange behavior of element 104, rutherfordium (Rf), was investigated together with its lighter group-4 homologues Zr and Hf, and the tetravalent pseudo-homologue Th in HF/HNO mixed solution. The results demonstrate that distribution coefficients (
mixed solution. The results demonstrate that distribution coefficients (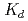 ) of Rf in HF/0.10 M HNO
) of Rf in HF/0.10 M HNO decrease with increasing concentration of the fluoride ion [F
decrease with increasing concentration of the fluoride ion [F ], indicating the consecutive formation of fluorido complexes of Rf. We also measured the
], indicating the consecutive formation of fluorido complexes of Rf. We also measured the 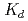 values of Rf and the homologues as a function of the hydrogen ion concentration [H
values of Rf and the homologues as a function of the hydrogen ion concentration [H ]. The log
]. The log 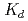 values decrease linearly with an increase of log [H
values decrease linearly with an increase of log [H ] with slopes between -2.1 and -2.5. This indicates that these elements are likely to form the same chemical compounds: mixture of [MF]
] with slopes between -2.1 and -2.5. This indicates that these elements are likely to form the same chemical compounds: mixture of [MF] and [MF
and [MF ]
] (M = Rf, Zr, Hf and Th) in the studied solution. It is also ascertained that sequence in the fluoride complex formation is Zr
(M = Rf, Zr, Hf and Th) in the studied solution. It is also ascertained that sequence in the fluoride complex formation is Zr  Hf
Hf  Rf
Rf  Th.
Th.
Toyoshima, Atsushi; Kasamatsu, Yoshitaka*; Tsukada, Kazuaki; Asai, Masato; Ishii, Yasuo; Tome, Hayato*; Nishinaka, Ichiro; Sato, Tetsuya; Nagame, Yuichiro; Sch del, M.; et al.
del, M.; et al.
Journal of Nuclear and Radiochemical Sciences, 11(1), p.7 - 11, 2010/06
The extraction behavior of rutherfordium (Rf) into trioctylphosphine oxide (TOPO) from 2.0 - 7.0 M HCl solution was studied together with that of the homologues Zr and Hf. The extracted yields of Rf, Zr, and Hf increased with an increase of HCl concentration, and the sequence of their extraction was Zr  Hf
Hf  Rf. It is suggested that the stability of the RfCl
Rf. It is suggested that the stability of the RfCl
 2(TOPO) complex is lower than that of the corresponding species of the homologues.
2(TOPO) complex is lower than that of the corresponding species of the homologues.
Kasamatsu, Yoshitaka*; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Li, Z.; Ishii, Yasuo; Tome, Hayato*; Sato, Tetsuya; Kikuchi, Takahiro; Nishinaka, Ichiro; et al.
Chemistry Letters, 38(11), p.1084 - 1085, 2009/10
Times Cited Count:16 Percentile:48.84(Chemistry, Multidisciplinary)We report on the characteristic anion-exchange behavior of the superheavy element dubnium (Db) with atomic number Z = 105 in HF/HNO solution at the fluoride ion concentration [F
solution at the fluoride ion concentration [F ] = 0.003 M. The result clearly demonstrates that the fluoro complex formation of Db is significantly different from that of the group-5 homologue Ta in the 6th period of the periodic table while the behavior of Db is similar to that of the lighter homologue Nb in the 5th period.
] = 0.003 M. The result clearly demonstrates that the fluoro complex formation of Db is significantly different from that of the group-5 homologue Ta in the 6th period of the periodic table while the behavior of Db is similar to that of the lighter homologue Nb in the 5th period.
 Bh and
Bh and  Db produced in the
Db produced in the  Cm +
Cm + 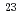 Na reaction
Na reactionMorita, Kosuke*; Morimoto, Koji*; Kaji, Daiya*; Haba, Hiromitsu*; Ozeki, Kazutaka*; Kudo, Yuki*; Sato, Nozomi*; Sumita, Takayuki*; Yoneda, Akira*; Ichikawa, Takatoshi*; et al.
Journal of the Physical Society of Japan, 78(6), p.064201_1 - 064201_6, 2009/06
Times Cited Count:30 Percentile:78.29(Physics, Multidisciplinary)Decay properties of an isotope  Bh and its daughter nucleus
Bh and its daughter nucleus  Db produced by the
Db produced by the  Cm(
Cm(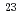 Na,5
Na,5 ) reaction were studied by using a gas-filled recoil separator coupled with a position-sensitive semiconductor detector.
) reaction were studied by using a gas-filled recoil separator coupled with a position-sensitive semiconductor detector.  Bh was clearly identified from the correlation of the known nuclide,
Bh was clearly identified from the correlation of the known nuclide,  Db. The obtained decay properties of
Db. The obtained decay properties of  Bh and
Bh and  Db are consistent with those observed in the
Db are consistent with those observed in the 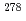 113 chain, which provided further confirmation of the discovery of
113 chain, which provided further confirmation of the discovery of 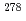 113.
113.
Tsukada, Kazuaki; Haba, Hiromitsu*; Asai, Masato; Toyoshima, Atsushi; Akiyama, Kazuhiko*; Kasamatsu, Yoshitaka; Nishinaka, Ichiro; Ichikawa, Shinichi; Yasuda, Kenichiro; Miyamoto, Yutaka; et al.
Radiochimica Acta, 97(2), p.83 - 89, 2009/02
Times Cited Count:21 Percentile:77.63(Chemistry, Inorganic & Nuclear)Anion-exchange chromatography of element 105, dubnium (Db), produced in the  Cm(
Cm( F, 5n)
F, 5n) Db reaction is investigated together with the homologues Nb and Ta, and the pseudo-homologue Pa in 13.9 M hydrofluoric acid (HF) solution. The distribution coefficient (K
Db reaction is investigated together with the homologues Nb and Ta, and the pseudo-homologue Pa in 13.9 M hydrofluoric acid (HF) solution. The distribution coefficient (K ) of Db on an anion-exchange resin is successfully determined by running cycles of the 1702 chromatographic column separations. The result clearly indicates that the adsorption of Db on the resin is significantly different from that of the homologues and that the adsorption of anionic fluoro complexes of these elements decreases in the sequence of Ta
) of Db on an anion-exchange resin is successfully determined by running cycles of the 1702 chromatographic column separations. The result clearly indicates that the adsorption of Db on the resin is significantly different from that of the homologues and that the adsorption of anionic fluoro complexes of these elements decreases in the sequence of Ta  Nb
Nb  Db
Db  Pa.
Pa.
Ishii, Yasuo; Toyoshima, Atsushi; Tsukada, Kazuaki; Asai, Masato; Tome, Hayato; Nishinaka, Ichiro; Nagame, Yuichiro; Miyashita, Sunao*; Mori, Tomotaka*; Suganuma, Hideo*; et al.
Chemistry Letters, 37(3), p.288 - 289, 2008/03
Times Cited Count:21 Percentile:54.95(Chemistry, Multidisciplinary)We have investigated cation-exchange behavior of Rf together with the lighter homologues of the group-4 elements Zr and Hf, and the tetravalent pseudo-homologue Th, in HF/HNO solution using Automated Ion exchange separation apparatus coupled with the Detection system for Alpha spectroscopy (AIDA). The
solution using Automated Ion exchange separation apparatus coupled with the Detection system for Alpha spectroscopy (AIDA). The 
 values of Zr, Hf, Th and Rf in HF/0.1 M HNO
values of Zr, Hf, Th and Rf in HF/0.1 M HNO were decreased with increasing the concentration of the fluoride ion [F
were decreased with increasing the concentration of the fluoride ion [F ], indicating the formation of the fluoride complexes. The sequence of the fluoride complexation strength is Zr
], indicating the formation of the fluoride complexes. The sequence of the fluoride complexation strength is Zr  Hf
Hf  Rf
Rf  Th.
Th.
 solutions
solutionsToyoshima, Atsushi; Haba, Hiromitsu*; Tsukada, Kazuaki; Asai, Masato; Akiyama, Kazuhiko*; Goto, Shinichi*; Ishii, Yasuo; Nishinaka, Ichiro; Sato, Tetsuya; Nagame, Yuichiro; et al.
Radiochimica Acta, 96(3), p.125 - 134, 2008/03
Times Cited Count:29 Percentile:84.99(Chemistry, Inorganic & Nuclear)Formation of an anionic fluoride-complex of element 104, rutherfordium (Rf) produced in the  Cm(
Cm( O,5n)
O,5n) Rf reaction was studied by an anion-exchange method based on an atom-at-a-time scale. It was found that the hexafluoro complex of Rf, [RfF
Rf reaction was studied by an anion-exchange method based on an atom-at-a-time scale. It was found that the hexafluoro complex of Rf, [RfF ]
] , was formed in the studied fluoride ion concentrations of 0.0005 - 0.013 M. Formation of [RfF
, was formed in the studied fluoride ion concentrations of 0.0005 - 0.013 M. Formation of [RfF ]
] was significantly different from that of the homologues Zr and Hf, [ZrF
was significantly different from that of the homologues Zr and Hf, [ZrF ]
] and [HfF
and [HfF ]
] ; the evaluated formation constant of [RfF
; the evaluated formation constant of [RfF ]
] is at least one-order of magnitude smaller than those of [ZrF
is at least one-order of magnitude smaller than those of [ZrF ]
] and [HfF
and [HfF ]
] .
.
Nishinaka, Ichiro; Tanikawa, Masashi*; Goto, Shinichi*; Nishio, Katsuhisa; Asai, Masato; Tsukada, Kazuaki; Nagame, Yuichiro; Kudo, Hisaaki*
Nuclear Physics A, 805, p.434 - 436, 2008/00
The aim of this work is to study how shell effects of fissioning nuclei and fission fragments are responsible for deformation and mass division during descent from the saddle point to scission. We studied in detail excitation energy dependence of mass and total kinetic energy distributions in the fission of  +
+ 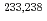 U at incident beam energies of 10.0, 11.5 and 13.0 MeV. For the asymmetric mass division in the fission for both uranium isotopes, the obtained yield at
U at incident beam energies of 10.0, 11.5 and 13.0 MeV. For the asymmetric mass division in the fission for both uranium isotopes, the obtained yield at 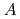
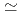 132 with the spherical shell of
132 with the spherical shell of  = 82 increases more largely with increasing excitation energy than that at
= 82 increases more largely with increasing excitation energy than that at 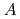
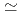 143 with deformed shell of
143 with deformed shell of  = 86-88. The present excitation energy dependence shows the opposite trend observed in the thermal neutron induced fission and cold fission of actinides. We will discuss the shell effects on excitation energy dependence of fragment mass and total kinetic energy distributions in fission.
= 86-88. The present excitation energy dependence shows the opposite trend observed in the thermal neutron induced fission and cold fission of actinides. We will discuss the shell effects on excitation energy dependence of fragment mass and total kinetic energy distributions in fission.
Haba, Hiromitsu*; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Ishii, Yasuo; Tome, Hayato; Sato, Tetsuya; Nishinaka, Ichiro; Ichikawa, Takatoshi; Ichikawa, Shinichi; et al.
Radiochimica Acta, 95(1), p.1 - 6, 2007/01
Times Cited Count:16 Percentile:70.17(Chemistry, Inorganic & Nuclear)no abstracts in English