Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Uchida, Shunsuke*; Morishima, Yusuke*; Hirose, Tatsuya*; Miyazawa, Takahiro*; Sato, Tomonori; Sato, Yoshiyuki*; Wada, Yoichi*
Journal of Nuclear Science and Technology, 44(5), p.758 - 766, 2007/05
Times Cited Count:5 Percentile:37.19(Nuclear Science & Technology)no abstracts in English
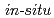 measurements of oxide film electric resistance in high temperature water
measurements of oxide film electric resistance in high temperature waterUchida, Shunsuke; Sato, Tomonori; Kakinuma, Nagao*; Miyazawa, Takahiro*; Sato, Yoshiyuki*; M kel
kel , K.*
, K.*
ECS Transactions, 2(25), p.39 - 50, 2007/00
no abstracts in English
Sato, Tomonori; Uchida, Shunsuke; Tsukada, Takashi; Morishima, Yusuke*; Miyazawa, Takahiro*; Sato, Yoshiyuki*
Proceedings of International Conference on Water Chemistry of Nuclear Reactor Systems 2006 (CD-ROM), 6 Pages, 2006/10
To evaluate ECP response of stainless steel in H O
O condition, the polarization of stainless steel in highly purified water at elevated temperature was measured. In this study a temporary Pt reference electrode, which was fixed at a natural potential vs. the Ag/AgCl reference electrode, was applied for polarization measurements. Stepwise voltage was supplied to a working electrode vs. the temporary reference electrode. As a result, the followings were concluded. (1) It was confirmed that both anodic and cathodic current densities increased as [H
condition, the polarization of stainless steel in highly purified water at elevated temperature was measured. In this study a temporary Pt reference electrode, which was fixed at a natural potential vs. the Ag/AgCl reference electrode, was applied for polarization measurements. Stepwise voltage was supplied to a working electrode vs. the temporary reference electrode. As a result, the followings were concluded. (1) It was confirmed that both anodic and cathodic current densities increased as [H O
O ] increased from 10 ppb to 100 ppb. It might be caused by the contribution of anodic reaction of H
] increased from 10 ppb to 100 ppb. It might be caused by the contribution of anodic reaction of H O
O as well as cathodic reaction of H
as well as cathodic reaction of H O
O . (2) Neither anodic nor cathodic polarization curves change as [O
. (2) Neither anodic nor cathodic polarization curves change as [O ] increase from 200 ppb to 2400 ppb. It might be caused by the mitigation of iron dissolution and cathodic reaction of O
] increase from 200 ppb to 2400 ppb. It might be caused by the mitigation of iron dissolution and cathodic reaction of O by the formation of protective oxide layer and lower oxide dissolution.
by the formation of protective oxide layer and lower oxide dissolution.
Uchida, Shunsuke; Sato, Tomonori; Tsukada, Takashi; Sato, Yoshiyuki*; Miyazawa, Takahiro*; Terachi, Takumi*; Wada, Yoichi*; Hosokawa, Hideyuki*
Proceedings of International Conference on Water Chemistry of Nuclear Reactor Systems 2006 (CD-ROM), 6 Pages, 2006/10
no abstracts in English
Miyazawa, Takahiro*; Terachi, Takumi*; Uchida, Shunsuke*; Sato, Tomonori; Tsukada, Takashi; Sato, Yoshiyuki*; Wada, Yoichi*; Hosokawa, Hideyuki*
Journal of Nuclear Science and Technology, 43(8), p.884 - 895, 2006/08
Times Cited Count:65 Percentile:96.73(Nuclear Science & Technology)In order to understand corrosion behavior of stainless steel in BWR reactor water conditions, characteristics of oxide films on stainless steel specimens exposed to H O
O and O
and O in high temperature water were determined by multilateral surface analyses. The following points were experimentally confirmed. (1) Oxide layers were divided into inner and outer layers: Outer layers of the specimen exposed to 100ppb H
in high temperature water were determined by multilateral surface analyses. The following points were experimentally confirmed. (1) Oxide layers were divided into inner and outer layers: Outer layers of the specimen exposed to 100ppb H O
O consisted of larger corundum type hematite particles, while inner layersconsisted of very fine Ni rich spinel type magnetite. (2) Outer oxide layers consisted of oxide particles, whose density and size were changed by oxidant concentration. The average diameter decreased with [O
consisted of larger corundum type hematite particles, while inner layersconsisted of very fine Ni rich spinel type magnetite. (2) Outer oxide layers consisted of oxide particles, whose density and size were changed by oxidant concentration. The average diameter decreased with [O ] and [H
] and [H O
O ]. (3) A larger dissolution rate at higher [H
]. (3) A larger dissolution rate at higher [H O
O ] resulted in a thinner oxide film with smaller particles.
] resulted in a thinner oxide film with smaller particles.
Uchida, Shunsuke*; Sato, Tomonori; Morishima, Yusuke*; Hirose, Tatsuya*; Miyazawa, Takahiro*; Kakinuma, Nagao*; Sato, Yoshiyuki*; Usui, Naoshi*; Wada, Yoichi*
Proceedings of 12th International Conference on Environmental Degradation of Materials in Nuclear Power Systems-Water Reactors (CD-ROM), p.19 - 29, 2005/00
Static and dynamic responses of stainless steel specimens exposed to H O
O and O
and O in high temperature water were evaluated by analyzing ECP and FDCI (frequency dependent complex impedance). The oxide films on the specimens were characterized by multilateral surface analyses, e.g., LRS, SIMS, XPS and direct electric resistance measurement. As a result of evaluation, it was confirmed that (1) corrosive condition of BWR normal water chemistry (NWC) was simulated by 100 ppb H
in high temperature water were evaluated by analyzing ECP and FDCI (frequency dependent complex impedance). The oxide films on the specimens were characterized by multilateral surface analyses, e.g., LRS, SIMS, XPS and direct electric resistance measurement. As a result of evaluation, it was confirmed that (1) corrosive condition of BWR normal water chemistry (NWC) was simulated by 100 ppb H O
O without co-existing O
without co-existing O , while that of hydrogen water chemistry (HWC) was simulated by 10 ppb H
, while that of hydrogen water chemistry (HWC) was simulated by 10 ppb H O
O , (2) ECP under HWC was as high as that under NWC, while dissolution rate of oxide film under HWC was much lower than that under NWC, (3) combination effects of electric resistance and dissolution rate of oxide caused same level ECP for both NWC and HWC, and (4) distinct weight loss of the specimen exposed to 100 ppb H
, (2) ECP under HWC was as high as that under NWC, while dissolution rate of oxide film under HWC was much lower than that under NWC, (3) combination effects of electric resistance and dissolution rate of oxide caused same level ECP for both NWC and HWC, and (4) distinct weight loss of the specimen exposed to 100 ppb H O
O was observed.
was observed.
Sato, Tomonori; Uchida, Shunsuke; Kakinuma, Nagao*; Miyazawa, Takahiro*; Sato, Yoshiyuki*; Tsukada, Takashi
no journal, ,
no abstracts in English
 O
O effects on corrosion of structural materials, 25; Dependency of electric resistance of the oxide film
effects on corrosion of structural materials, 25; Dependency of electric resistance of the oxide filmSato, Tomonori; Uchida, Shunsuke; Kakinuma, Nagao*; Miyazawa, Takahiro*; Sato, Yoshiyuki*; Tsukada, Takashi
no journal, ,
no abstracts in English