Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Matsuda, Shohei; Nakashima, Nobuaki*; Yokoyama, Keiichi; Taniguchi, Seiji*; Chosrowjan, H.*; Somekawa, Toshihiro*; Yatsuhashi, Tomoyuki*
Chemical Physics Letters, 802, p.139759_1 - 139759_6, 2022/09
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)no abstracts in English
Matsuda, Shohei; Yokoyama, Keiichi; Yaita, Tsuyoshi; Kobayashi, Toru; Kaneta, Yui; Simonnet, M.; Sekiguchi, Tetsuhiro; Honda, Mitsunori; Shimojo, Kojiro; Doi, Reisuke; et al.
Science Advances (Internet), 8(20), p.eabn1991_1 - eabn1991_11, 2022/05
Times Cited Count:6 Percentile:58.16(Multidisciplinary Sciences)no abstracts in English
Nakashima, Nobuaki*; Yatsuhashi, Tomoyuki*; Sakota, Kenji*; Iwakura, Izumi*; Hashimoto, Sena*; Yokoyama, Keiichi; Matsuda, Shohei
Chemical Physics Letters, 752, p.137570_1 - 137570_5, 2020/08
Times Cited Count:1 Percentile:5(Chemistry, Physical)Photo-redox reactions between Eu and Eu
and Eu ions are induced by laser irradiation in alcoholic solution. Efficiency, wavelength dependence, and laser-power dependence are investigated with three different lasers. Nano second laser pulses at a wavelength of 308 nm is found to cause one-photon redox reactions with a quantum yield around 0.5. Nano second laser pulses at a wavelength of 394 nm induces two-photon reduction of Eu
ions are induced by laser irradiation in alcoholic solution. Efficiency, wavelength dependence, and laser-power dependence are investigated with three different lasers. Nano second laser pulses at a wavelength of 308 nm is found to cause one-photon redox reactions with a quantum yield around 0.5. Nano second laser pulses at a wavelength of 394 nm induces two-photon reduction of Eu to form Eu
to form Eu . When the pulse energy is 5 mJ, the quantum yield is measured to be 0.015. Although the quantum yield is one order of magnitude lower than that of the one photon reduction, reduction phenomena can be easily observed under the moderate laser field strength. Because of the two-photon nature, there should be a room to improve the efficiency by increasing the laser field strength.
. When the pulse energy is 5 mJ, the quantum yield is measured to be 0.015. Although the quantum yield is one order of magnitude lower than that of the one photon reduction, reduction phenomena can be easily observed under the moderate laser field strength. Because of the two-photon nature, there should be a room to improve the efficiency by increasing the laser field strength.
Saeki, Morihisa; Taguchi, Tomitsugu; Nakashima, Nobuaki*; Oba, Hironori
Journal of Photochemistry and Photobiology A; Chemistry, 299, p.189 - 193, 2015/02
Times Cited Count:14 Percentile:35.41(Chemistry, Physical)Imaizumi, Tomomi; Miyauchi, Masaru; Ito, Masayasu; Watahiki, Shunsuke; Nagata, Hiroshi; Hanakawa, Hiroki; Naka, Michihiro; Kawamata, Kazuo; Yamaura, Takayuki; Ide, Hiroshi; et al.
JAEA-Technology 2011-031, 123 Pages, 2012/01
The number of research reactors in the world is decreasing because of their aging. However, the planning to introduce the nuclear power plants is increasing in Asian countries. In these Asian countries, the key issue is the human resource development for operation and management of nuclear power plants after constructed them, and also the necessity of research reactor, which is used for lifetime extension of LWRs, progress of the science and technology, expansion of industry use, human resources training and so on, is increasing. From above backgrounds, the Neutron Irradiation and Testing Reactor Center began to discuss basic concept of a multipurpose low-power research reactor for education and training, etc. This design study is expected to contribute not only to design tool improvement and human resources development in the Neutron Irradiation and Testing Reactor Center but also to maintain and upgrade the technology on research reactors in nuclear power-related companies. This report treats the activities of the working group from July 2010 to June 2011 on the multipurpose low-power research reactor in the Neutron Irradiation and Testing Reactor Center and nuclear power-related companies.
Kameo, Yutaka; Fujiwara, Asako; Watanabe, Koichi; Kono, Nobuaki; Nakashima, Mikio
Nihon Genshiryoku Gakkai Wabun Rombunshi, 4(3), p.187 - 193, 2005/09
no abstracts in English
Oyama, Yukio; *; Watanabe, Yukinobu*; Kawano, Toshihiko*; Numajiri, M.*; Ueki, Kotaro*; *; Yamano, Naoki*; Kosako, Kazuaki*; *; et al.
JAERI-Review 98-020, 130 Pages, 1998/11
no abstracts in English
Izawa, Yasukazu*; *; Suto, O.; Wada, Yukio; Nakashima, Nobuaki*
Reza Kenkyu, 26(1), p.112 - 119, 1998/01
None
Saeki, Morihisa; Taguchi, Tomitsugu; Matsumura, Daiju; Nakashima, Nobuaki*; Oba, Hironori
no journal, ,
no abstracts in English
Nakashima, Nobuaki*; Yamanaka, Kenichi*; Yatsuhashi, Tomoyuki*; Taniguchi, Seiji*; Saeki, Morihisa; Oba, Hironori
no journal, ,
no abstracts in English
Saeki, Morihisa; Taguchi, Tomitsugu; Matsumura, Daiju; Nakashima, Nobuaki*; Oba, Hironori
no journal, ,
no abstracts in English
Saeki, Morihisa; Matsumura, Daiju; Yomogida, Takumi; Taguchi, Tomitsugu; Tsuji, Takuya; Kusano, Shogo*; Miyazaki, Tatsuya*; Takao, Koichiro*; Oba, Hironori; Nakashima, Nobuaki*
no journal, ,
Recently, we found that photo-induced particle formation of Pd was promoted by coexistence of molybdenum anion in aqueous solution. In this study, we performed in-situ time-resolved XAFS measurement in the photoreduction of Pd
was promoted by coexistence of molybdenum anion in aqueous solution. In this study, we performed in-situ time-resolved XAFS measurement in the photoreduction of Pd in presence of various concentration of molybdenum anion. The effect of molybdenum addition on photo-induced particle formation of Pd will be discussed based on the relationship between the photoreduction rate of Pd
in presence of various concentration of molybdenum anion. The effect of molybdenum addition on photo-induced particle formation of Pd will be discussed based on the relationship between the photoreduction rate of Pd and the concentration of molybdenum anion.
and the concentration of molybdenum anion.
Saeki, Morihisa; Matsumura, Daiju; Yomogida, Takumi; Taguchi, Tomitsugu; Tsuji, Takuya; Kusano, Shogo*; Miyazaki, Tatsuya*; Takao, Koichiro*; Oba, Hironori; Nakashima, Nobuaki*
no journal, ,
no abstracts in English
Saeki, Morihisa*; Oba, Hironori*; Taguchi, Tomitsugu*; Yokoyama, Jun*; Asai, Shiho; Yomogida, Takumi; Hanzawa, Yukiko; Nakashima, Nobuaki*
no journal, ,
We are developing separation of platinum-group metals from a solution of high-level radioactive waste by laser-induced particle formation. In this report, we will show method and proof-of-principle experiments of the separation using the laser-induced particle formation.
Yokoyama, Keiichi; Nakashima, Nobuaki*; Yaita, Tsuyoshi
no journal, ,
We propose a new concept for separating f-electron elements like lanthanides or actinides. These elements have similar chemical property because only the inner shell electronic configuration is different from each other. Therefore, it is considered that chemical techniques alone is hard to complete precise mutual separation between these elements. Meanwhile, the f-f transitions exhibit drastic difference for each element in their photo absorption spectra owing to the inner shell character. There is a possibility to realize precise mutual separation by applying these f-f transitions to the separation processes. In the conference, we introduce and discuss our target and a technical feature in our study plan.
Nakashima, Nobuaki*; Yokoyama, Keiichi; Yatsuhashi, Tomoyuki*; Sakota, Kenji*; Iwakura, Izumi*
no journal, ,
Studies on photo-reduction of trivalent europium, Eu(III), is revisited by nanosecond lasers at various wavelengths with a Ti:Sapphire, excimer, and dye lasers. We measured the quantum yields, wavelength dependence, and power dependence. As a result, we found that the photo-reduction is induced by a two-photon absorption with a relatively high efficiency as to be observed directly with eyes. Also, the quantum yield of one-photon reduction at 308 nm was determined to be 0.6. These results indicate that highly efficient and selective separation techniques are possible for f-electron elements using multi-photon absorption process induced by laser light.
 by multiphoton excitation in alcohol solutions
by multiphoton excitation in alcohol solutionsMatsuda, Shohei; Nakashima, Nobuaki*; Yokoyama, Keiichi; Yatsuhashi, Tomoyuki*; Chosrowjan, H.*; Taniguchi, Seiji*; Somekawa, Toshihiro*
no journal, ,
The photoredox reactions of lanthanides as a photochemical separative technique proposed in the 1970s are expected to be applied for recovery and recycling of rare metals and nuclear fuel reprocessing. In this study, Eu ions in methanol and ethanol solutions were reduced to Eu
ions in methanol and ethanol solutions were reduced to Eu ions upon irradiation with nanosecond laser pulses. Eu
ions upon irradiation with nanosecond laser pulses. Eu was observed when the laser wavelength was tuned to f-f transition bands at 394 and 465 nm. The formation rate of Eu
was observed when the laser wavelength was tuned to f-f transition bands at 394 and 465 nm. The formation rate of Eu was proportional to the square of the incident laser intensity, indicating that the photoreduction is induced by a two-photon process. The efficiency of the photoreduction in ethanol was higher than that in methanol.
was proportional to the square of the incident laser intensity, indicating that the photoreduction is induced by a two-photon process. The efficiency of the photoreduction in ethanol was higher than that in methanol.
Matsuda, Shohei; Nakashima, Nobuaki*; Yokoyama, Keiichi; Yatsuhashi, Tomoyuki*; Chosrowjan, H.*; Taniguchi, Seiji*; Somekawa, Toshihiro*
no journal, ,
The photochemical reactions of lanthanide and actinide ions are expected to be utilized for recovery and recycling of rare earths, and nuclear fuel reprocessing. Heading towards research for implementation of photochemical purification, a unified evaluation of the efficiency of multiphoton processes is required. In this study, we have evaluated the fluence dependence of photoreduction of Eu(III) more precisely than before by measuring the beam profile of the excitation laser. The reduction rate of Eu(III) Eu(II) is proportional to the square of the laser fluence, indicating that the photoreduction of Eu(III) is induced by a two-photon process.
Eu(II) is proportional to the square of the laser fluence, indicating that the photoreduction of Eu(III) is induced by a two-photon process.
Matsuda, Shohei; Yokoyama, Keiichi; Yaita, Tsuyoshi; Kobayashi, Toru; Nakashima, Nobuaki*
no journal, ,
One of the spectroscopic properties for lanthanides and actinides is that they have f-f transitions in the visible region of their spectra. Those absorption appears at specific wavelength and has narrow line width even in the liquid phase owing to shielded f electrons. It has been reported that reduction reactions of Sm , Eu
, Eu , and Yb
, and Yb are induced by multiphoton excitation through each f-f transition depending on conditions. This photochemical process may be applicable to mutual separation of f elements. In this study, we tried to optically control the oxidation state of americium. As a result, we found that trivalent americium (Am
are induced by multiphoton excitation through each f-f transition depending on conditions. This photochemical process may be applicable to mutual separation of f elements. In this study, we tried to optically control the oxidation state of americium. As a result, we found that trivalent americium (Am ) in nitric acid solution is oxidized to pentavalent one (AmO
) in nitric acid solution is oxidized to pentavalent one (AmO
 ) during nanosecond pulsed-laser irradiation at 503 nm corresponding to the
) during nanosecond pulsed-laser irradiation at 503 nm corresponding to the  L
L
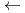
 F
F transition of Am
transition of Am . Identification and quantification of the reactants and products were performed by absorption spectroscopy. Excitation wavelength dependence and fluence dependence of the oxidation suggest that it is initiated by a resonance-enhanced multiphoton process.
. Identification and quantification of the reactants and products were performed by absorption spectroscopy. Excitation wavelength dependence and fluence dependence of the oxidation suggest that it is initiated by a resonance-enhanced multiphoton process.
Matsuda, Shohei; Yokoyama, Keiichi; Yaita, Tsuyoshi; Kobayashi, Toru; Nakashima, Nobuaki*
no journal, ,
We have recently realized the oxidation of trivalent americium (Am ) in nitric acid by multiphoton excitation via an f-f transition. In this study, an effect of nitrate ion (NO
) in nitric acid by multiphoton excitation via an f-f transition. In this study, an effect of nitrate ion (NO
 ) on the Am
) on the Am photooxidation have been investigated. Our results of NO
photooxidation have been investigated. Our results of NO
 activity dependence of the Am
activity dependence of the Am photooxidation and density functional theory calculation indicate that the complex of Am
photooxidation and density functional theory calculation indicate that the complex of Am with NO
with NO
 , in particular one-nitrate complex, contributes to it.
, in particular one-nitrate complex, contributes to it.