Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Amamoto, Ippei; Mitamura, Naoki*; Tsuzuki, Tatsuya*; Takasaki, Yasushi*; Shibayama, Atsushi*; Yano, Tetsuji*; Nakada, Masami; Okamoto, Yoshihiro
Proceedings of 13th International Conference on Environmental Remediation and Radioactive Waste Management (ICEM 2010) (CD-ROM), p.503 - 508, 2010/10
The main objective of this development is to recycle the purified eutectic medium of the pyroreprocessing, delaying its disposal for as long as possible. We have introduced the simple filtration method to remove the rare earth element (REE) particles which were formed due to the conversion of REE chlorides to phosphates. Here, the iron phosphate glass is used as a filtration medium for the removal of FP particles. However, some soluble FP such as compounds of alkali-metals, alkaline-earth metals, etc. still remain in the eutectic medium. This time around, on an experimental basis, the iron phosphate glass has been used as a sorbent instead, to remove the soluble FP. We have obtained some positive results and have intention to incorporate it into the spent electrolyte recycle process as a part of the FP separation and immobilization system.
Amamoto, Ippei; Kofuji, Hirohide; Myochin, Munetaka; Takasaki, Yasushi*; Yano, Tetsuji*; Terai, Takayuki*
Nuclear Technology, 171(3), p.316 - 324, 2010/09
Times Cited Count:11 Percentile:59.05(Nuclear Science & Technology)The separation technique by phosphate conversion of FP from NaCl-KCl or NaCl-2CsCl as the medium of the spent electrolyte of pyroprocessing is being developed at RIAR in Russia. To ascertain the technical feasibility of applying this RIAR's method for recycling of spent electrolyte which composed of FP such as AL, ALE, and REE and 3LiCl-2KCl as the medium, preliminary study was undertaken at JAEA. This study occupies an auxiliary role which is forwarded as a part of FaCT Project of Japan. As a first approach, the thermodynamic analysis to understand the behaviours of FP in the electrolyte was carried out after literature research and acquisition of required thermodynamic properties. Preliminary experiment was then undertaken to observe the conversion behaviours of chlorides to phosphate. In this paper, these results are shown and are evaluated to discern the feasibility of the phosphate conversion method.
Amamoto, Ippei; Kofuji, Hirohide; Myochin, Munetaka; Tsuzuki, Tatsuya*; Takasaki, Yasushi*; Yano, Tetsuji*; Terai, Takayuki*
Proceedings of 12th International Conference on Environmental Remediation and Radioactive Waste Management (ICEM '09/DECOM '09) (CD-ROM), 9 Pages, 2009/10
This study is carried out to make the pyroprocessing hold a competitive advantage. As one of the measures is to reduce the volume of HLW, the phosphate conversion method is applied for removal of FP from the spent electrolyte in this paper. Though the removing target elements in the electrolyte are alkali metals, alkaline earth metals and lanthanoid elements, only lanthanoid elements and lithium form the insoluble phosphates by reaction with Li PO
PO . Therefore, as the first step, the precipitation experiment was carried out to observe the behaviours of elements which form the insoluble precipitates. Then the filtration was experimented to remove precipitates in the spent electrolyte using Fe
. Therefore, as the first step, the precipitation experiment was carried out to observe the behaviours of elements which form the insoluble precipitates. Then the filtration was experimented to remove precipitates in the spent electrolyte using Fe O
O -P
-P O
O glass system as a filtlation medium which is compatible material with the glassification. The result of separation of precipitates by filtration was effective and attained almost 100
glass system as a filtlation medium which is compatible material with the glassification. The result of separation of precipitates by filtration was effective and attained almost 100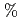 .
.
Kofuji, Hirohide; Amamoto, Ippei; Sasaki, Kazuya*; Yasumoto, Masaru*; Takasaki, Yasushi*; Myochin, Munetaka; Terai, Takayuki*
Denki Kagaku Oyobi Kogyo Butsuri Kagaku, 77(8), p.597 - 600, 2009/08
Times Cited Count:0 Percentile:0.01(Electrochemistry)The process flow of the phosphate conversion technique has been developed for the reduction of high-level radioactive waste (HLW) generated from the metal-electrorefining process. In this study, the results of thermodynamic calculations for the phosphate conversion reactions were examined by the basic experiments. The chlorides of rare earth elements (REE) turned out to be converted into phosphates easily. Furthermore, as the additive for the phosphate conversion reaction, high temperature behavior of lithium phosphate was evaluated to elucidate the thermodynamic property.
Amamoto, Ippei; Kofuji, Hirohide; Myochin, Munetaka; Takasaki, Yasushi*; Terai, Takayuki*
Journal of Nuclear Materials, 389(1), p.142 - 148, 2009/05
Times Cited Count:15 Percentile:68.83(Materials Science, Multidisciplinary)The spent electrolyte of the pyroprocessing by metal electrorefining method should be considered for recycling after removal of FP, such as AL (alkali metals), ALE (alkaline earth metals), and/or REE (rare-earth elements), to reduce the volume of HLW. Authors have been carrying out theoretical analysis and experiment showing the behaviours of phosphate precipitates so as to estimate the feasibility of the phosphate conversion method. From hitherto results, it was found that AL except Li, and ALE are unlikely formed phosphate precipitates. However, their conversion behaviours including REE were compatible with former presented theoretical analysis; in the case of lanthanum precipitate as one of REE, submicron-size particles could be observed while that of Li PO
PO was larger; under different conversion temperatures, the precipitates were apt to grow larger at higher temperature; etc..
was larger; under different conversion temperatures, the precipitates were apt to grow larger at higher temperature; etc..
Kofuji, Hirohide; Amamoto, Ippei; Yasumoto, Masaru*; Takasaki, Yasushi*; Sasaki, Kazuya*; Myochin, Munetaka; Terai, Takayuki*
no journal, ,
Phosphate conversion process for recycling spent electrolyte originated from pyrochemical reprocessing has been studied. Feasibility of the process has been evaluated thermodynamically based on basic experiments. Current situation of phosphate conversion experiments and the estimations of thermodynamic properties will be introduced in this presentation.
Kofuji, Hirohide; Amamoto, Ippei; Sasaki, Kazuya*; Yasumoto, Masaru*; Takasaki, Yasushi*; Myochin, Munetaka; Terai, Takayuki*
no journal, ,
Phosphate conversion process for recycling spent electrolyte originated from pyrochemical reprocessing has been studied. Feasibility of the process has been evaluated thermodynamically based on basic experiments. Current situation of phosphate conversion experiments and the estimations of thermodynamic properties will be introduced in this presentation.
Amamoto, Ippei; Kofuji, Hirohide; Myochin, Munetaka; Takasaki, Yasushi*; Yano, Tetsuji*; Terai, Takayuki*
no journal, ,
no abstracts in English
Amamoto, Ippei; Kofuji, Hirohide; Myochin, Munetaka; Tsuzuki, Tatsuya*; Mitamura, Naoki*; Takasaki, Yasushi*; Yano, Tetsuji*; Terai, Takayuki*
no journal, ,
no abstracts in English
Takasaki, Yasushi*; Amamoto, Ippei; Myochin, Munetaka; Shibayama, Atsushi*
no journal, ,
no abstracts in English
Amamoto, Ippei; Kofuji, Hirohide; Tsuzuki, Tatsuya*; Mitamura, Naoki*; Takasaki, Yasushi*; Shibayama, Atsushi*; Yano, Tetsuji*; Terai, Takayuki*
no journal, ,
no abstracts in English
Amamoto, Ippei; Kofuji, Hirohide; Mitamura, Naoki*; Tsuzuki, Tatsuya*; Motohashi, Takumi*; Takasaki, Yasushi*; Shibayama, Atsushi*; Yano, Tetsuji*
no journal, ,
no abstracts in English