Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -Ga
-Ga O
O (001)
(001)Tsai, Y. H.*; Kobata, Masaaki; Fukuda, Tatsuo; Tanida, Hajime; Kobayashi, Toru; Yamashita, Yoshiyuki*
Applied Physics Letters, 124(11), p.112105_1 - 112105_5, 2024/03
Nankawa, Takuya; Sekine, Yurina; Matsumura, Daiju; Hiroi, Kosuke; Takata, Shinichi; Kamiya, Yoshimi*; Honda, Takayuki*
Langmuir, 40(11), p.5725 - 5730, 2024/03
Times Cited Count:0The chemical reaction between Fe and lacquer has been used to create the black color lacquer since ancient times. Here, the chemical state of Fe ions in black lacquer was investigated by using X-ray absorption near edge structure (XANES), extended X-ray absorption fine structure (EXAFS), and Fourier transform-infrared (FT-IR) spectroscopy. Fe(II) or Fe(III) was added to the lacquer paste to prepare black lacquer films by air drying, heating, or UV irradiation. The XANES spectral features of all the film samples were similar, meaning that the Fe ions in the samples existed in the trivalent state regardless of the oxidation state of the initially added Fe. The corresponding Fourier transforms of the EXAFS spectra around the Fe K-edge were used to investigate Fe sites in the lacquer films. The spectra of all the film samples were similar shapes, but the peak intensities decreased in the order air dried  heated
heated  UV irradiated films. This result indicates that heating and UV irradiation made the coordination structure of Fe in the lacquer non-uniform, and that heating caused the greatest non-uniformity. The complementary use of XANES, XAFS, and FT-IR spectroscopy is highly effective for non-destructive analysis of black lacquer in precious cultural artifacts.
UV irradiated films. This result indicates that heating and UV irradiation made the coordination structure of Fe in the lacquer non-uniform, and that heating caused the greatest non-uniformity. The complementary use of XANES, XAFS, and FT-IR spectroscopy is highly effective for non-destructive analysis of black lacquer in precious cultural artifacts.
Miura, Yasufumi*; Suzuki, Kenji*; Morooka, Satoshi; Shobu, Takahisa
Quantum Beam Science (Internet), 8(1), p.1_1 - 1_14, 2024/03
Yoshimoto, Masataka*; Tamura, Kazuhisa; Watanabe, Kenta*; Shimizu, Keisuke*; Horisawa, Yuhei*; Kobayashi, Takeshi*; Tsurita, Hanae*; Suzuki, Kota*; Kanno, Ryoji*; Hirayama, Masaaki*
Sustainable Energy & Fuels (Internet), 8(6), p.1236 - 1244, 2024/03
Times Cited Count:0Photo-rechargeable systems, which can efficiently convert and store solar energy into chemical energy within single devices, are essential to harness sunlight effectively. Photo-(de)intercalation plays a pivotal role in the functionality of photorechargeable systems. Nevertheless, the photo-(de)intercalation process has not been conclusively confirmed owing to potential interference from side reactions, such as the decomposition of liquid electrolytes and the elution of electrode materials. In this study, we successfully demonstrated photo-responsive Li -deintercalation using an all-solid-state thin-film battery comprised of epitaxially-grown anatase TiO
-deintercalation using an all-solid-state thin-film battery comprised of epitaxially-grown anatase TiO doped with Nb (a-TiO
doped with Nb (a-TiO :Nb) as the cathode. Under light irradiation, Li
:Nb) as the cathode. Under light irradiation, Li -deintercalation occurred and was subsequently reversibly intercalated into a-TiO
-deintercalation occurred and was subsequently reversibly intercalated into a-TiO :Nb during discharge.
:Nb during discharge.
Watanabe, Kenta*; Horisawa, Yuhei*; Yoshimoto, Masataka*; Tamura, Kazuhisa; Suzuki, Kota*; Kanno, Ryoji*; Hirayama, Masaaki*
Nano Letters, 24(6), p.1916 - 1922, 2024/02
Times Cited Count:1 Percentile:0.02(Chemistry, Multidisciplinary)Electrochemistry has extended from reactions at solid/liquid interfaces to those at solid/solid interfaces. In this study, we achieve the stable photoelectrochemical reaction at the semiconductor-electrode/solid-electrolyte interface in Nb-doped anatase-TiO (a-TiO
(a-TiO :Nb)/Li
:Nb)/Li PO
PO (LPO)/Li all-solid-state cell. The oxidative currents of a-TiO
(LPO)/Li all-solid-state cell. The oxidative currents of a-TiO :Nb/LPO/Li increase upon light irradiation when a-TiO
:Nb/LPO/Li increase upon light irradiation when a-TiO :Nb is located at a potential that is more positive than its flat-band potential. The photoelectrochemical reaction at the semiconductor/solid-electrolyte interface is driven by the same principle as that at semiconductor/liquid-electrolyte interfaces. Thus, we extend photoelectrochemistry to all-solid-state systems composed of solid/solid interfaces.
:Nb is located at a potential that is more positive than its flat-band potential. The photoelectrochemical reaction at the semiconductor/solid-electrolyte interface is driven by the same principle as that at semiconductor/liquid-electrolyte interfaces. Thus, we extend photoelectrochemistry to all-solid-state systems composed of solid/solid interfaces.
 Cl dissociation, CH
Cl dissociation, CH abstraction, and Cl adsorption from the dissociative scattering of supersonic CH
abstraction, and Cl adsorption from the dissociative scattering of supersonic CH Cl on Cu(111) and Cu(410)
Cl on Cu(111) and Cu(410)Makino, Takamasa*; Tsuda, Yasutaka; Yoshigoe, Akitaka; Di o, W. A.*; Okada, Michio*
o, W. A.*; Okada, Michio*
Applied Surface Science, 642, p.158568_1 - 158568_6, 2024/01
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)Ikeda, Kazutaka*; Sashida, Sho*; Otomo, Toshiya*; Oshita, Hidetoshi*; Honda, Takashi*; Hawai, Takafumi*; Saito, Hiraku*; Ito, Shinichi*; Yokoo, Tetsuya*; Sakaki, Koji*; et al.
International Journal of Hydrogen Energy, 51(Part A), p.79 - 87, 2024/01
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)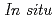 Ga K-edge XANES study of Ga-exchanged zeolites at high temperatures under different atmospheres including vacuum, CO, and pressurized H
Ga K-edge XANES study of Ga-exchanged zeolites at high temperatures under different atmospheres including vacuum, CO, and pressurized H
Huang, M.*; Kinjo, Tetsuya*; Yasumura, Shunsaku*; Toyao, Takashi*; Matsumura, Daiju; Saito, Hiroyuki*; Shimizu, Kenichi*; Namiki, Norikazu*; Maeno, Zen*
Catalysis Science & Technology, 13(23), p.6832 - 6838, 2023/12
Times Cited Count:0 Percentile:0(Chemistry, Physical)Okazaki, Hiroyuki*; Idesaki, Akira*; Koshikawa, Hiroshi*; Matsumura, Daiju; Ikeda, Takashi*; Yamamoto, Shunya*; Yamaki, Tetsuya*
Journal of Physical Chemistry C, 127(49), p.23628 - 23633, 2023/12
Times Cited Count:0 Percentile:0(Chemistry, Physical) Ge
Ge
Fujiwara, Hidenori*; Nakatani, Yasuhiro*; Aratani, Hidekazu*; Kanai-Nakata, Yuina*; Yamagami, Kohei*; Hamamoto, Satoru*; Kiss, Takayuki*; Sekiyama, Akira*; Tanaka, Arata*; Ebihara, Takao*; et al.
New Physics; Sae Mulli, 73(12), p.1062 - 1066, 2023/12
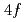 states in the electronic structure of the intermediate-valence superconductor CeIr
states in the electronic structure of the intermediate-valence superconductor CeIr
Fujimori, Shinichi; Kawasaki, Ikuto; Takeda, Yukiharu; Yamagami, Hiroshi; Sasabe, Norimasa*; Sato, Yoshiki*; Shimizu, Yusei*; Nakamura, Ai*; Maruya, A.*; Homma, Yoshiya*; et al.
Electronic Structure (Internet), 5(4), p.045009_1 - 045009_7, 2023/11
Tamura, Kazuhisa
Journal of Physical Chemistry C, 127(46), p.22733 - 22739, 2023/11
Times Cited Count:0 Percentile:0(Chemistry, Physical)The underpotential deposition of Bi on Au(111) electrode in 1 M HClO solution and 1-butyl-3-methylimidazolium tetrafluoroborate was investigated using visible light reflectance measurement and surface X-ray scattering techniques. The electrosorption valency of the UPD reaction of Bi was elucidated and it was found that in both 1 M HClO
solution and 1-butyl-3-methylimidazolium tetrafluoroborate was investigated using visible light reflectance measurement and surface X-ray scattering techniques. The electrosorption valency of the UPD reaction of Bi was elucidated and it was found that in both 1 M HClO and 1-butyl-3-methylimidazolium tetrafluoroborate the electrosorption valency was smaller than 3, but the detail process in the UPD reaction was different between in two electrolytes. The difference may be originated from the difference in the solvation status of Bi
and 1-butyl-3-methylimidazolium tetrafluoroborate the electrosorption valency was smaller than 3, but the detail process in the UPD reaction was different between in two electrolytes. The difference may be originated from the difference in the solvation status of Bi rather than the electrical double layer structure.
rather than the electrical double layer structure.
Matsumura, Daiju; Kimura, Yusaku*; Tsuji, Takuya; Mizuki, Junichiro*
SPring-8/SACLA Riyo Kenkyu Seikashu (Internet), 11(5), p.296 - 299, 2023/11
no abstracts in English
Yamaguchi, Akiko; Okumura, Masahiko; Takahashi, Yoshio*
Isotope News, (789), p.20 - 23, 2023/10
Radium is a radioactive element produced from uranium and thorium and is important for environmental contamination issues around uranium mines and for geological disposal. In addition, radium is used in radiometric dating and cancer therapy, making it important not only in environmental chemistry but also in many other fields, including geochemistry and nuclear medicine. However, because radium is a radioactive element with no stable isotopes, spectroscopic measurement of radium is difficult, and little information at the molecular level has been obtained so far. In this study, we have clarified the molecular-level information of hydrated radium for the first time in the world by combining extended X-ray absorption fine structure (EXAFS) measurements and first-principles molecular dynamics simulations.
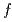 symmetry for anisotropic
symmetry for anisotropic 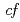 hybridization in the heavy-fermion superconductor CeNi
hybridization in the heavy-fermion superconductor CeNi Ge
Ge
Fujiwara, Hidenori*; Nakatani, Yasuhiro*; Aratani, Hidekazu*; Kanai, Yuina*; Yamagami, Kohei*; Hamamoto, Satoru*; Kiss, Takayuki*; Yamasaki, Atsushi*; Higashiya, Atsushi*; Imada, Shin*; et al.
Physical Review B, 108(16), p.165121_1 - 165121_10, 2023/10
Times Cited Count:0 Percentile:0(Materials Science, Multidisciplinary)Hirano, Tatsumi*; Maeda, Takehiro*; Murata, Tetsuyuki*; Yamamoto, Takahiro*; Matsubara, Eiichiro*; Shobu, Takahisa; Shiro, Ayumi*; Yasuda, Ryo*; Takamatsu, Daiko*
SPring-8/SACLA Riyo Kenkyu Seikashu (Internet), 11(5), p.345 - 353, 2023/10
no abstracts in English
Arai, Taiki*; Yoshigoe, Akitaka; Motohashi, Mitsuya*
Zairyo No Kagaku To Kogaku, 60(5), p.153 - 158, 2023/10
Si oxide films are currently widely used as insulating materials in electronic devices and biomaterials. The atomic bonding state of these films significantly influences the properties of each device, thus it is particularly necessary to understand and control the chemical bonding state between Si and O in the films. In this study, the Si oxide films formed by anodic oxidation on Si substrate surfaces in extremely low concentrations of HF solutions were analyzed by X-ray photoelectron spectroscopy mainly focusing on Si2p and F1s spectra. Although the HF concentration is in the order of ppm, the films contain percent order of F atoms, suggesting the formation of Si-F and Si-O-F bonds in the films. It was also found that the different depth profiles for F and O atoms was observed, indicating that the surface reaction processes seem to be different depending on each element.
Ikeda, Yoichi*; Umemoto, Yoshihiko*; Matsumura, Daiju; Tsuji, Takuya; Hashimoto, Yuki*; Kitazawa, Takafumi*; Fujita, Masaki*
Materials Transactions, 64(9), p.2254 - 2260, 2023/09
Times Cited Count:0Takeda, Yukiharu; Posp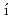
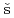 il, J.*; Yamagami, Hiroshi; Yamamoto, Etsuji; Haga, Yoshinori
il, J.*; Yamagami, Hiroshi; Yamamoto, Etsuji; Haga, Yoshinori
Physical Review B, 108(8), p.085129_1 - 085129_10, 2023/08
Times Cited Count:0 Percentile:0(Materials Science, Multidisciplinary)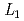 -edge XAFS measurement
-edge XAFS measurementTsuji, Takuya; Matsumura, Daiju; Kobayashi, Toru
SPring-8/SACLA Riyo Kenkyu Seikashu (Internet), 11(4), p.214 - 217, 2023/08
no abstracts in English