Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Shindo, Manami*; Ueoku, Aya*; Okamura, Wakana*; Kikuchi, Shin; Yamazaki, Atsushi*; Koga, Nobuyoshi*
Thermochimica Acta, 749, p.180021_1 - 180021_14, 2025/07
Kikuchi, Shin; Koga, Nobuyoshi*
Journal of Thermal Analysis and Calorimetry, 150(1), p.585 - 590, 2025/01
Times Cited Count:0 Percentile:0.00(Thermodynamics)Teshigawara, Makoto; Lee, Y.*; Tatsumoto, Hideki*; Hartl, M.*; Aso, Tomokazu; Iverson, E. B.*; Ariyoshi, Gen; Ikeda, Yujiro*; Hasegawa, Takumi*
Nuclear Instruments and Methods in Physics Research B, 557, p.165534_1 - 165534_10, 2024/12
Times Cited Count:0 Percentile:0.00(Instruments & Instrumentation)At Japanese Spallation Neutron Source in J-PARC, the para-hydrogen fraction was measured by using Raman spectroscopy in-situ for an integrated beam power of 9.4 MW h at 1 MW operation, to evaluate the functionality of the ferric oxyhydroxide catalyst. This result showed that full functionality of the catalyst was retained up to the 1 MW operation. We attempted to study the effect of neutron scattering driven para to ortho-hydrogen back-conversion rate in the absence of the catalyst effect with a bypass line without catalyst. The measured increase of ortho-hydrogen fraction was 0.44% for an integrated beam power of 2.4 MW
h at 1 MW operation, to evaluate the functionality of the ferric oxyhydroxide catalyst. This result showed that full functionality of the catalyst was retained up to the 1 MW operation. We attempted to study the effect of neutron scattering driven para to ortho-hydrogen back-conversion rate in the absence of the catalyst effect with a bypass line without catalyst. The measured increase of ortho-hydrogen fraction was 0.44% for an integrated beam power of 2.4 MW h at 500 kW operation, however, which was considered to be due to not only to neutron collisions in cold moderators but also to the high ortho-hydrogen fraction of initially static liquid hydrogen in the bypass line and passive exudation of quasi-static hydrogen in the catalyst vessel to the main loop.
h at 500 kW operation, however, which was considered to be due to not only to neutron collisions in cold moderators but also to the high ortho-hydrogen fraction of initially static liquid hydrogen in the bypass line and passive exudation of quasi-static hydrogen in the catalyst vessel to the main loop.
Shindo, Manami*; Ueoku, Aya*; Okamura, Wakana*; Kikuchi, Shin; Yamazaki, Atsushi*; Koga, Nobuyoshi*
Thermochimica Acta, 738, p.179801_1 - 179801_12, 2024/08
Times Cited Count:1 Percentile:0.00(Thermodynamics)Iwata, Takuma*; Kosa, Towa*; Nishioka, Yukimi*; Owada, Kiyotaka*; Sumida, Kazuki; Annese, E.*; Kakoki, Masaaki*; Kuroda, Kenta*; Iwasawa, Hideaki*; Arita, Masashi*; et al.
Scientific Reports (Internet), 14, p.127_1 - 127_8, 2024/01
Times Cited Count:7 Percentile:89.50(Multidisciplinary Sciences)Zhang, A.*; Deng, K.*; Sheng, J.*; Liu, P.*; Kumar, S.*; Shimada, Kenya*; Jiang, Z.*; Liu, Z.*; Shen, D.*; Li, J.*; et al.
Chinese Physics Letters, 40(12), p.126101_1 - 126101_8, 2023/12
Times Cited Count:11 Percentile:83.08(Physics, Multidisciplinary)Hattori, Koichi*; Suenaga, Daiki*; Suzuki, Kei; Yasui, Shigehiro*
Physical Review B, 108(24), p.245110_1 - 245110_11, 2023/12
Times Cited Count:2 Percentile:13.37(Materials Science, Multidisciplinary)We develop a mean-field theory of a novel Kondo effect emerging in systems without a Fermi surface, which instead emerges under strong magnetic fields. We determine the magnitude of the Kondo condensate, which is a particle pairing composed of conducting Dirac fermions and localized impurities. We focus on the competition between the Kondo effect and the energy gap formation that stems from the pairing among the Dirac fermions leading to the dynamical chiral symmetry breaking. We find that this competition induces a quantum critical point. We also investigate finite-temperature effects. This system at vanishing fermion density can be studied with Monte Carlo lattice simulations, which do not suffer from the sign problem.
Sumida, Kazuki; Sakuraba, Yuya*; Kimura, Akio*
Kotai Butsuri, 58(3), p.117 - 137, 2023/03
no abstracts in English
Hirata, Sakiko*; Kusaka, Ryoji; Meiji, Shogo*; Tamekuni, Seita*; Okudera, Kosuke*; Hamada, Shoken*; Sakamoto, Chihiro*; Honda, Takumi*; Matsushita, Kosuke*; Muramatsu, Satoru*; et al.
Inorganic Chemistry, 62(1), p.474 - 486, 2023/01
Times Cited Count:3 Percentile:28.83(Chemistry, Inorganic & Nuclear)Iwasaki, Shun*; Sakai, Yasuhiro*; Kikuchi, Shin; Koga, Nobuyoshi*
Journal of Thermal Analysis and Calorimetry, 147(11), p.6309 - 6322, 2022/06
Times Cited Count:12 Percentile:52.19(Thermodynamics)The influence of atmospheric water vapor on the multistep thermal decomposition of perlite concrete was investigated in detail by separating the component reaction steps using kinetic deconvolution analysis (KDA). The retardation of thermal decomposition of Ca(OH) and the acceleration of thermal decomposition of CaCO
and the acceleration of thermal decomposition of CaCO were identified as specific effect of atmospheric water vapor.
were identified as specific effect of atmospheric water vapor.
 K
K (Zn
(Zn Mn
Mn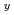 )
) As
As studied by X-ray magnetic circular dichroism and resonant inelastic X-ray scattering
studied by X-ray magnetic circular dichroism and resonant inelastic X-ray scatteringSuzuki, Hakuto*; Zhao, G.*; Okamoto, Jun*; Sakamoto, Shoya*; Chen, Z.-Y.*; Nonaka, Yosuke*; Shibata, Goro; Zhao, K.*; Chen, B.*; Wu, W.-B.*; et al.
Journal of the Physical Society of Japan, 91(6), p.064710_1 - 064710_5, 2022/06
Times Cited Count:0 Percentile:0.00(Physics, Multidisciplinary) on the thermal decomposition of perlite concrete
on the thermal decomposition of perlite concreteSakai, Yasuhiro*; Iwasaki, Shun*; Kikuchi, Shin; Koga, Nobuyoshi*
Journal of Thermal Analysis and Calorimetry, 147(10), p.5801 - 5813, 2022/05
Times Cited Count:14 Percentile:59.42(Thermodynamics)The influence of atmospheric CO on the multistep thermal decomposition of perlite concrete was investigated in detail by separating the component reaction steps using kinetic deconvolution analysis (KDA). The carbonation of Ca(OH)
on the multistep thermal decomposition of perlite concrete was investigated in detail by separating the component reaction steps using kinetic deconvolution analysis (KDA). The carbonation of Ca(OH) and the retardation of the thermal decomposition of CaCO
and the retardation of the thermal decomposition of CaCO were identified as specific effect of atmospheric CO
were identified as specific effect of atmospheric CO .
.
Kikuchi, Shin; Koga, Nobuyoshi*
Journal of Thermal Analysis and Calorimetry, 147(7), p.4635 - 4643, 2022/04
Times Cited Count:2 Percentile:9.57(Thermodynamics)In a sodium-cooled fast reactor (SFR), sodium-concrete reaction (SCR) may probably occur when liquid sodium from cooling system spilled into the floor may lead to fail the steel liner as protector of the structural concretes. The structural concretes of SFR comprises siliceous concreate as main body of reactor structure and perlite concrete placed between the steel liner and the siliceous concrete serving as a protector and an insulator, respectively. Therefore, the reaction behaviour between the perlite concrete and liquid sodium in the early stage of SCR should be focused. In this study, for the first step of elucidation on SCR, thermal behaviour of sodium-calcium hydroxide reaction was investigated using a differential scanning calorimetry (DSC). It was revealed that the reaction between Na(l) and Ca(OH) (s) initiates at approximately 550 K, producing the product layer composed of CaO(s) and NaOH(s), which is evident from XRD pattern of solid product after DSC measurement. The kinetic consideration of the cited reaction was carried out to obtain the activation energy.
(s) initiates at approximately 550 K, producing the product layer composed of CaO(s) and NaOH(s), which is evident from XRD pattern of solid product after DSC measurement. The kinetic consideration of the cited reaction was carried out to obtain the activation energy.
 reaction; A Physicogeometrical kinetic approach toward the safety assessment of Na-cooled fast reactors
reaction; A Physicogeometrical kinetic approach toward the safety assessment of Na-cooled fast reactorsKoga, Nobuyoshi*; Kikuchi, Shin
Industrial & Engineering Chemistry Research, 61(7), p.2759 - 2770, 2022/02
Times Cited Count:2 Percentile:10.49(Engineering, Chemical) MnGa thin films underneath a protection layer
MnGa thin films underneath a protection layerKono, Takashi*; Kakoki, Masaaki*; Yoshikawa, Tomoki*; Wang, X.*; Sumida, Kazuki; Muro, Takayuki*; Goto, Kazuki*; Sakuraba, Yuya*; Umetsu, Rie*; Kimura, Akio*
Physical Review B, 104(19), p.195112_1 - 195112_8, 2021/11
Times Cited Count:4 Percentile:24.33(Materials Science, Multidisciplinary)Angle-resolved photoelectron spectroscopy utilizing soft X-ray synchrotron radiation was applied to Heusler-type Co MnGa thin films that have a 1-nm Al capping layer. The bulk Fermi surfaces and band structures varied along the out-of-plane momentum, stemming from the three-dimensional crystal structure, in the absence of any in situ surface treatment. In addition, there were characteristic intersecting bands (Weyl cones), with crossing points near the Fermi level, which were consistent with computed results. The Weyl cones are of bulk origin and are responsible for the high anomalous Nernst and the anomalous Hall coefficients. A close comparison of the experimental band structures in Co
MnGa thin films that have a 1-nm Al capping layer. The bulk Fermi surfaces and band structures varied along the out-of-plane momentum, stemming from the three-dimensional crystal structure, in the absence of any in situ surface treatment. In addition, there were characteristic intersecting bands (Weyl cones), with crossing points near the Fermi level, which were consistent with computed results. The Weyl cones are of bulk origin and are responsible for the high anomalous Nernst and the anomalous Hall coefficients. A close comparison of the experimental band structures in Co MnGe and Co
MnGe and Co MnGa indicated that the rigid band picture is valid in both alloys and that fine carrier tuning is possible by replacing Ga with Ge to improve the anomalous conductivity.
MnGa indicated that the rigid band picture is valid in both alloys and that fine carrier tuning is possible by replacing Ga with Ge to improve the anomalous conductivity.
 VAl/Fe layered films showing exchange bias
VAl/Fe layered films showing exchange biasKubota, Takahide*; Shimada, Yusuke*; Tsuchiya, Tomoki*; Yoshikawa, Tomoki*; Ito, Keita*; Takeda, Yukiharu; Saito, Yuji; Konno, Toyohiko*; Kimura, Akio*; Takanashi, Koki*
Nanomaterials (Internet), 11(7), p.1723_1 - 1723_11, 2021/07
Times Cited Count:2 Percentile:10.80(Chemistry, Multidisciplinary) Te
Te -based topological insulators
-based topological insulatorsSumida, Kazuki; Ishida, Yukiaki*; G dde, J.*; H
dde, J.*; H fer, U.*; Shin, S.*; Kimura, Akio*
fer, U.*; Shin, S.*; Kimura, Akio*
Progress in Surface Science, 96(2), p.100628_1 - 100628_15, 2021/05
Times Cited Count:4 Percentile:2.91(Chemistry, Physical)Sumida, Kazuki; Ishida, Yukiaki*; Kimura, Akio*
Nihon Butsuri Gakkai-Shi, 75(12), p.756 - 760, 2020/12
Sumida, Kazuki; Sakuraba, Yuya*; Masuda, Keisuke*; Kono, Takashi*; Kakoki, Masaaki*; Goto, Kazuki*; Zhou, W.*; Miyamoto, Koji*; Miura, Yoshio*; Okuda, Taichi*; et al.
Communications Materials (Internet), 1, p.89_1 - 89_9, 2020/11
Segawa, Tomoomi; Kawaguchi, Koichi; Ishii, Katsunori; Suzuki, Masahiro; Fukasawa, Tomonori*; Fukui, Kunihiro*
Funtai Kogakkai-Shi, 57(9), p.485 - 494, 2020/09
In the spent fuel reprocessing process, a mixed solution of uranyl nitrate and plutonium nitrate is converted into mixed oxide powder by the microwave heating. To evaluate the applicability to the industrial-scale and acquire the characteristics data of the microwave heating denitration of various metal nitrate aqueous solutions based on the knowledge studied in the development of laboratory-scale basic experiments, the microwave heating characteristics and metal oxide powder properties were investigated using cerium nitrate, cobalt nitrate and copper nitrate aqueous solutions. The progress rate of the denitration reaction was depended on the position, and the denitration reaction proceeded faster at the periphery than at the center. The morphologies of the synthesized products were porous and hard dry solid with cerium nitrate aqueous solution, foamed dry solid with cobalt nitrate aqueous solution, and powdery particles with copper nitrate aqueous solution. The denitration ratio and average particle size of the synthesized products increased in the order of the cerium nitrate aqueous solution, the cobalt nitrate aqueous solution, and the copper nitrate aqueous solution. The numerical simulations revealed that the periphery of the bottom surface of the metal nitrate aqueous solution was heated by microwaves. This results consistent with the experimental results in which the denitration reaction started from the periphery of the metal nitrate aqueous solution.