Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tamada, Taro; Honjo, Eijiro; Kuroki, Ryota
Nihon Kessho Gakkai-Shi, 48(6), p.429 - 435, 2006/12
The crystal structure of the signaling complex of human granulocyte colony-stimulating factor (GCSF) and ligand binding region of human GCSF receptor (GCSF-R) has been determined to 2.8  resolution. The GCSF:GCSF-R complex formed a 2:2 stoichiometry via a cross-over interaction between the Ig-like domains of GCSF-R and GCSF. The conformation of the complex is quite different to that between human GCSF and the CRH domain of mouse GCSF-R. This cross-over homodimerization necessary for GCSF-R activation is consistent with previously reported thermodynamic and mutational studies.
resolution. The GCSF:GCSF-R complex formed a 2:2 stoichiometry via a cross-over interaction between the Ig-like domains of GCSF-R and GCSF. The conformation of the complex is quite different to that between human GCSF and the CRH domain of mouse GCSF-R. This cross-over homodimerization necessary for GCSF-R activation is consistent with previously reported thermodynamic and mutational studies.
Tamada, Taro; Honjo, Eijiro; Maeda, Yoshitake*; Okamoto, Tomoyuki*; Ishibashi, Matsujiro*; Tokunaga, Masao*; Kuroki, Ryota
Proceedings of the National Academy of Sciences of the United States of America, 103(9), p.3135 - 3140, 2006/02
Times Cited Count:95 Percentile:84.33(Multidisciplinary Sciences)A crystal structure of the signaling complex between human granulocyte colony-stimulating factor (GCSF), and a ligand binding region of GCSF receptor (GCSF-R), has been determined to 2.8 resolution. The GCSF:GCSF-R complex formed a 2:2 stoichiometry via a cross-over interaction between the Ig-like domains of GCSF-R and GCSF. The conformation of the complex is quite different to that between human GCSF and the CRH domain of mouse GCSF-R, but similar to that of the interleukin-6 (IL-6)/gp130 signaling complex. The Ig-like domain cross-over structure necessary for GCSF-R activation is consistent with previously reported thermodynamic and mutational analyses.
resolution. The GCSF:GCSF-R complex formed a 2:2 stoichiometry via a cross-over interaction between the Ig-like domains of GCSF-R and GCSF. The conformation of the complex is quite different to that between human GCSF and the CRH domain of mouse GCSF-R, but similar to that of the interleukin-6 (IL-6)/gp130 signaling complex. The Ig-like domain cross-over structure necessary for GCSF-R activation is consistent with previously reported thermodynamic and mutational analyses.
Okamura, Masachika*; Tanaka, Atsushi; Momose, Masaki*; Umemoto, Naoyuki*; Silva, J.*; Toguri, Toshihiro*
Floriculture, Ornamental and Plant Biotechnology; Advances and Topical Issues, Vol.1, p.619 - 628, 2006/00
no abstracts in English
Honjo, Eijiro; Tamada, Taro; Maeda, Yoshitake*; Koshiba, Takumi*; Matsukura, Yasuko*; Okamoto, Tomoyuki*; Ishibashi, Matsujiro*; Tokunaga, Masao*; Kuroki, Ryota
Acta Crystallographica Section F, 61(8), p.788 - 790, 2005/08
Times Cited Count:7 Percentile:55.27(Biochemical Research Methods)Granulocyte colony-stimulating factor (GCSF) receptor receives signals for regulating the proliferation and differentiation of the precursor cells of granulocytes. The complex composed of two GCSFs and two GCSF receptors was crystallized. The crystal of the complex was grown in 1.0 M sodium formate and 0.1 M sodium acetate (pH4.6). It belongs to the space group 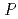 4
4 2
2 2 (or its enantiomorph
2 (or its enantiomorph 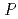 4
4 2
2 2) with unit cell dimensions of
2) with unit cell dimensions of 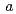 =
= 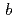 = 110.1
= 110.1  ,
,  = 331.8
= 331.8  . However, the diffraction data from the crystal beyond 5
. However, the diffraction data from the crystal beyond 5  resolution could not be collected. Since the heterogeneity of GCSF receptor seems to interrupt growth of good quality crystals, the GCSF receptor was fractionated by achromatography. Crystals of GCSF/fractionated GCSF receptor complex were grown as a new crystal form in 0.2 M ammonium phosphate. The new crystal diffracts beyond 3.0
resolution could not be collected. Since the heterogeneity of GCSF receptor seems to interrupt growth of good quality crystals, the GCSF receptor was fractionated by achromatography. Crystals of GCSF/fractionated GCSF receptor complex were grown as a new crystal form in 0.2 M ammonium phosphate. The new crystal diffracts beyond 3.0  resolution and belongs to space group
resolution and belongs to space group 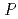 3
3 21 (or its enantiomorph
21 (or its enantiomorph 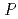 3
3 21) with unit-cell parameters
21) with unit-cell parameters 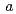 =
= 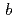 = 134.8,
= 134.8,  = 105.7
= 105.7  .
.
Okamura, Masachika*; Yasuno, Noriko*; Otsuka, Masako*; Tanaka, Atsushi; Shikazono, Naoya; Hase, Yoshihiro
Nuclear Instruments and Methods in Physics Research B, 206, p.574 - 578, 2003/05
Times Cited Count:91 Percentile:97.71(Instruments & Instrumentation)Resent studies indicate that the ion beams have higher mutation frequency than low-LET radiations in plants but the difference in mutation spectrum still remains to be characterized. We investigated the efficiency of ion-beam irradiation combined with tissue culture in obtaining floral mutants. Leaves collected from carnation plants, cultivar Vital (cherry pink flowers with frilly petals), were irradiated with carbon ions or X-rays. They were cultured till the shoots regenerated. Sixteen mutants were obtained from 705 regenerated plants by carbon-ion irradiation. Those mutants were rich in variety, i.e., pink, dark pink, light pink, salmon, red, complex- and striped-color, and round and Dianthus-type petals were obtained. In contrast, 7 mutants obtained from 556 regenerated plants by X-rays were only pink, light pink and red. These results indicate that the ion beams could induce wider variety of flower-color and shape mutant than X-rays, and also indicate that the combined method of ion-beam irradiation with tissue culture is useful to obtain commercial varieties in a short time.
Tamada, Taro
no journal, ,
no abstracts in English
Tamada, Taro
no journal, ,
no abstracts in English
Kuroki, Ryota; Tamada, Taro; Honjo, Eijiro; Maeda, Yoshitake*
no journal, ,
Receptor proteins expressed in the surface of the cell are known to be important drug targets for several diseases. The receptor transfer the signal originated from the binding of ligand proteins in to the signal cascades located inside of the cell. The signal transduction usually induced by the association of plural receptor molecules, thus the ligand molecules that help association of receptors are considered to be drug candidates. There are several association schemes are known. For example, the erythropoietin receptors associate with its ligand in 2:1 manner, the granulocyte colony stimulating factor receptor active complex with its ligand in 2:2 manners, and so on. Therefore, it is quite important to investigate the active scheme of receptor-ligand complex to understand how the receptor molecule start signal transduction. In this symposium, we show several examples of the measurements aimed to understand the activation scheme of drug target receptors.
Tamada, Taro
no journal, ,
no abstracts in English
Arai, Shigeki; Tamada, Taro; Maeda, Yoshitake*; Kuroki, Ryota
no journal, ,
The mouse antibody TN1 recognizes the human thrombopoietin (hTPO) that primarily stimulates megakaryocytopoiesis and platelet production. The TN1 neutralize the TPO activity preventing from the homo-dimerization of its receptor. In order to investigate the structural change of the TN1 upon antigen binding, the crystal structure of TN-1 Fab was determined to 2.1 resolution and was compared with that of TN1-Fab / hTPO complex (1V7M). It was found that each domain comprising TN1-Fab was strikingly similar (rmsd
resolution and was compared with that of TN1-Fab / hTPO complex (1V7M). It was found that each domain comprising TN1-Fab was strikingly similar (rmsd  0.6
0.6 ) between the antigen bound and unbound forms of TN1-Fab including three complementarity determining regions. The relative locations of the variable- and constant-regions of the TPO unbound form of TN1-Fab was slightly shifted from those of TN1-Fab / hTPO complex, which may be caused by difference of their crystal packing. These results indicate that the TN1-Fab need not accompany the conformational changes upon antigen binding.
) between the antigen bound and unbound forms of TN1-Fab including three complementarity determining regions. The relative locations of the variable- and constant-regions of the TPO unbound form of TN1-Fab was slightly shifted from those of TN1-Fab / hTPO complex, which may be caused by difference of their crystal packing. These results indicate that the TN1-Fab need not accompany the conformational changes upon antigen binding.
Okamura, Masachika*; Momose, Masaki*; Umemoto, Naoyuki*; Toguri, Toshihiro*; Tanaka, Atsushi; Hase, Yoshihiro; Yamaguchi, Hiroyasu*; Morishita, Toshikazu*
no journal, ,
no abstracts in English
Tamada, Taro
no journal, ,
no abstracts in English
Arai, Shigeki; Tamada, Taro; Maeda, Yoshitake*; Kuroki, Ryota
no journal, ,
The mouse antibody TN1 recognizes the human thrombopoietin (hTPO) that primarily stimulates megakaryocytopoiesis and platelet production. In order to clarify the mechanism of the neutralizing activity of the TN1 antibody, the crystal structure of TN1-Fab was determined to 2.1  resolution and was compared with that of TN1-Fab / hTPO complex (PDB id 1V7M and 1V7N). It was found that only side chain level "Induced-fit" upon the antigen binding was enough for TN1 to recognize hTPO. On the other hand, the relative angle of the variable- and constant-regions of the hTPO unbound form of TN1-Fab was slightly shifted from those of TN1-Fab / hTPO complex (rms deviation = 2.4
resolution and was compared with that of TN1-Fab / hTPO complex (PDB id 1V7M and 1V7N). It was found that only side chain level "Induced-fit" upon the antigen binding was enough for TN1 to recognize hTPO. On the other hand, the relative angle of the variable- and constant-regions of the hTPO unbound form of TN1-Fab was slightly shifted from those of TN1-Fab / hTPO complex (rms deviation = 2.4  for all C
for all C atoms of Fab). In this presentation, we will explain the details of the conformational change of the crystal structure of TN1-Fab fragment with and without binding of hTPO.
atoms of Fab). In this presentation, we will explain the details of the conformational change of the crystal structure of TN1-Fab fragment with and without binding of hTPO.
Kuroki, Ryota; Honjo, Eijiro; Maeda, Yoshitake*; Tamada, Taro
no journal, ,
We have succeeded in preparation and crystallization of 2:2 complex between human GCSF (hGCSF) and the Ig-like and CRH domains of human GCSF-R (hGCSF-R) and determined its tertiary structure by X-ray crystallography at 2.8 A resolution. The signaling 2:2 complex is formed via cross-over interactions between the Ig-like domain of hGCSF-R and the neighboring hGCSF, forming a two-fold axis of crystallographic symmetry. This conformation is quite different from that of the heterogeneous mGCSF-R complex, and more closely resembles the 2:2:2 active assembly of human interleukin-6 (IL-6), human IL-6  -receptor and human gp130 (which is a shared signal transducing receptor for several cytokines), and the 2:2 assembly of viral IL-6 and human gp130. The Ig-like domain cross-over structure necessary for GCSF-R activation is consistent with previously reported thermodynamic and mutational analyses.
-receptor and human gp130 (which is a shared signal transducing receptor for several cytokines), and the 2:2 assembly of viral IL-6 and human gp130. The Ig-like domain cross-over structure necessary for GCSF-R activation is consistent with previously reported thermodynamic and mutational analyses.
Tamada, Taro; Arai, Shigeki; Shoyama, Yoshinari; Honjo, Eijiro; Kuroki, Ryota
no journal, ,
no abstracts in English