Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Iwasa, Toma; Arima, Tatsumi*
JAEA-Technology 2021-036, 23 Pages, 2022/03
Knowledge on the liquefaction (thermal decomposition and melting) temperatures of MA-bearing nitride fuels for transmutation by accelerator-driven system is essential for elucidation of the fuel behaviors under abnormal condition and for the safety analysis. A melting temperature measurement system for refractory materials was developed based on a laser spot heating method, which is expected to measure in a very short time with a small amount of sample, and demonstration tests using refractory metals and zirconium nitride were performed. As the results, it was found that this melting temperature measurement system can be applicable up to the temperatures around 3000 K which is close to the thermal decomposition temperature of nitride fuels and we confirmed the technical feasibility of this system for future application to small specimens of transuranium nitrides.
Kawaguchi, Munemichi; Miyahara, Shinya; Uno, Masayoshi*
Journal of Nuclear Science and Technology, 56(6), p.513 - 520, 2019/06
Times Cited Count:2 Percentile:20.94(Nuclear Science & Technology)This study revealed melting points and thermal conductivities of four samples generated by sodium-concrete reaction (SCR). We prepared the samples using two methods such as firing mixtures of sodium and grinded concrete powder, and sampling depositions after the SCR experiments. In the former, the mixing ratios were determined from the past experiment. The latter simulated the more realistic conditions such as the temperature history and the distribution of Na and concrete. The thermogravimetry-differential thermal analyzer (TG-DTA) measurement showed the melting points were 865-942 C, but those of the samples containing metallic Na couldn't be clarified. In the two more realistic samples, the compression moldings in a furnace were observed. The observation revealed the softening temperature was 800-840
C, but those of the samples containing metallic Na couldn't be clarified. In the two more realistic samples, the compression moldings in a furnace were observed. The observation revealed the softening temperature was 800-840 C and the melting point was 840-850
C and the melting point was 840-850 C, which was 10-20
C, which was 10-20 C lower than the TG-DTA results. The thermodynamics calculation of FactSage 7.2 revealed the temperature of the onset of melting was caused by melting of the some components such as Na
C lower than the TG-DTA results. The thermodynamics calculation of FactSage 7.2 revealed the temperature of the onset of melting was caused by melting of the some components such as Na SiO
SiO and/or Na
and/or Na SiO
SiO . Moreover, the thermal conductivity was
. Moreover, the thermal conductivity was  =1-3W/m-K, which was comparable to xNa
=1-3W/m-K, which was comparable to xNa O-1-xSiO
O-1-xSiO (x=0.5, 0.33, 0.25), and those at 700
(x=0.5, 0.33, 0.25), and those at 700 C were explained by the equation of
C were explained by the equation of 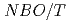 .
.
Kikuchi, Taiji; Yamada, Hirokazu*; Saito, Takashi; Nakamichi, Masaru; Tsuchiya, Kunihiko; Kawamura, Hiroshi
JAERI-Tech 2004-026, 28 Pages, 2004/03
Iirradiation capsule for irradiation test of tritium breeder and inner capsule for pebble bed of tritium breeder is inserted. Post irradiation examination of tritium breeder will be performed after irradiation test. On cutting of irradiation capsule, sweep gas line should be sealed to prevent the tritium gas release or inflow of water to sweep gas line. However, general valve and plug cannot apply to sweep gas line sealing because of the effect of neutron irradiation or limited space in irradiation capsule. Therefore, sealing plug for sweep gas line sealing has to be developed. This report shows the development of sealing plug for sweep gas line sealing and operating procedure of sealing plug in irradiation capsule.
Katayama, Yoshinori
Journal of Non-Crystalline Solids, 312-314, p.8 - 14, 2002/10
Times Cited Count:29 Percentile:83.93(Materials Science, Ceramics)Recently, we have found a sharp pressure-induced structural change between two characteristic structures of liquid phosphorus by an in-situ high-temperature high-presusre x-ray diffraction method using a JAERI beamline, BL14B1, at SPring-8. Our observations --- existence of molecular and polymeric liquids, sharp and reversible change between them and coexistence of two structures during the change --- strongly support that the structural change is a first-order liquid-liquid phase transition. It has been confirmed that the two liquid phases have different densities by a determination of melting curve by x-ray diffraction and by a density measurement by means of x-ray absorption. The structure of liquid P under high pressure resembles that of liquid arsenic.
Nakamura, Jinichi
Saishin Kaku Nenryo Kogaku; Kodoka No Genjo To Tembo, p.93 - 98, 2001/06
no abstracts in English
Fukui, Toshiki; Nakashio, Nobuyuki; Isobe, Motoyasu; Otake, Atsushi*; Wakui, Takuji*; Hirabayashi, Takakuni*; Nakashima, Mikio
JAERI-Review 2000-033, 82 Pages, 2001/02
no abstracts in English
 plasma
plasmaOkoshi, Kiyonori; Saito, Yuichi; Tajima, Satoshi
Kagaku To Kogyo, 52(11), p.1409 - 1412, 1999/11
no abstracts in English
Ishii, Tetsuya; Hirosawa, Takashi
PNC TN9410 97-075, 20 Pages, 1997/08
Fuel melting temperature is one of the major thermodynamical properties that is used for determining the design criteria on fuel temperature during irradiation in FBR. In general, it is necessary to evaluate the correlation of fuel melting temperature to confirm that the fuel temperature must be kept below the fuel melting temperature during irradiation at any conditions. The correlations of the melting temperature of uranium-plutonium mixed oxide (MOX) fuel, typical FBR fuel, used to be estimated and formulized based on the measured values reported in 1960's and has been applied to the design. At present, some experiments have been accumulated with improved experimental techniques. And it reveals that the recent measured melting temperatures does not agree well to the data reported in 1960's and that some of the 1960's data should be modified by taking into account of the recent measurements. In this study, the experience of melting temperature up to now are summarized and evaluated in order to make the fuel pin design more reliable. The effect of plutonium content, oxygen to metal ratio and burnup on MOX fuel melting was examined based on the recent data under the UO - PuO
- PuO - PuO
- PuO ideal solution model, and then formulized. The correlation obtained in this work is as-follows; T = T
ideal solution model, and then formulized. The correlation obtained in this work is as-follows; T = T +
+  T
T +
+  T
T +
+  T
T ----(A) T
----(A) T = 3120
= 3120  T
T = -5.7537
= -5.7537 PU + 1.3631
PU + 1.3631 10
10
 PU
PU + 1.7952
+ 1.7952 10
10
 PU
PU
 T
T = -1.41
= -1.41  PU
PU  (2.00 - OP)/0.39 OP : OP =
(2.00 - OP)/0.39 OP : OP =  /(0.01
/(0.01 PU)
PU)  T
T = -5.0
= -5.0 BU/10000 where T is the melting temperature (degree of K), PU is the weight fraction of PuO
BU/10000 where T is the melting temperature (degree of K), PU is the weight fraction of PuO in the mixed oxide fuel, OM is the oxygen to metal ratio, and BU is the burnup in the unit of MWd/MTM. respectively.
in the mixed oxide fuel, OM is the oxygen to metal ratio, and BU is the burnup in the unit of MWd/MTM. respectively.  T
T (plutonium content),
(plutonium content),  T
T (O/M Ratio),
(O/M Ratio),  ...
...
Yamahara, Takeshi; Nishino, Yasuharu; Amano, Hidetoshi;
IAEA-TECDOC-822, 0, p.43 - 54, 1995/09
no abstracts in English
Akino, Norio; Takase, Kazuyuki; Kubo, Shinji; *; *; *
Dai-31-Kai Nihon Dennetsu Shimpojiumu Koen Rombunshu, 0, p.589 - 591, 1994/00
no abstracts in English
Nature, 325, p.43 - 44, 1987/00
Times Cited Count:2 Percentile:18.56(Multidisciplinary Sciences)no abstracts in English
; ;
J.Appl.Polym.Sci., 32, p.5669 - 5671, 1986/00
Times Cited Count:7 Percentile:68.33(Polymer Science)no abstracts in English
; ; *
Nihon Genshiryoku Gakkai-Shi, 24(12), p.980 - 988, 1982/00
Times Cited Count:1 Percentile:21.73(Nuclear Science & Technology)no abstracts in English
; *; *; ; Araki, Kunio;
JAERI-M 9247, 28 Pages, 1980/12
no abstracts in English
Shiraishi, K.
Nihon Genshiryoku Gakkai-Shi, 21(6), p.471 - 479, 1979/00
Times Cited Count:0no abstracts in English
*; *; Kuriyama, Isamu
Reports on Progress in Polymer Physics in Japan, 19, p.169 - 172, 1976/00
no abstracts in English
Yamaki, J.; ; ; ;
JAERI-M 6250, 76 Pages, 1975/10
no abstracts in English
 fuel
fuelKoizumi, Masumichi; Furuya, Hirotaka
PNC TN841 74-22, 30 Pages, 1974/08
no abstracts in English
Kagaku No Ryoiki, 27(6), p.533 - 534, 1973/06
no abstracts in English
Koizumi, Masumichi; Kashima, Sadamitsu; *; *; Komatsu, Junji*; Matsumoto, Kenichi*
PNC TN841 71-38, 8 Pages, 1971/11
no abstracts in English