Thermodynamic model for the solubility of Ba(SeO , SO
, SO ) precipitates
) precipitates
Rai, D.*; Felmy, A. R.*; Moore, D. A.*; Kitamura, Akira 
 ; Yoshikawa, Hideki; Doi, Reisuke
; Yoshikawa, Hideki; Doi, Reisuke 
 ; Yoshida, Yasushi*
; Yoshida, Yasushi*
The solubility of Ba(SeO , SO
, SO ) precipitates was determined as a function of the BaSeO
) precipitates was determined as a function of the BaSeO mole fractions, ranging from 0.0015 to 0.3830, and time with an equilibration period extending to as long as 302 days. Equilibrium/steady state conditions in this system are reached in
mole fractions, ranging from 0.0015 to 0.3830, and time with an equilibration period extending to as long as 302 days. Equilibrium/steady state conditions in this system are reached in 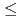 65 days. Pitzer's ion interaction model was used to calculate solid and aqueous phase activity coefficients. Thermodynamic analyses showed that the data do not satisfy Gibbs-Duhem equation, thereby demonstrating that a single-solid solution phase does not control both the selenate and sulfate concentrations. Our extensive data with [Ba], [SeO
65 days. Pitzer's ion interaction model was used to calculate solid and aqueous phase activity coefficients. Thermodynamic analyses showed that the data do not satisfy Gibbs-Duhem equation, thereby demonstrating that a single-solid solution phase does not control both the selenate and sulfate concentrations. Our extensive data with [Ba], [SeO ], and [SO
], and [SO ] can be explained with the formation of an ideal BaSeO
] can be explained with the formation of an ideal BaSeO solid solution phase that controls the selenium concentrations and a slightly disordered/less-crystalline BaSO
solid solution phase that controls the selenium concentrations and a slightly disordered/less-crystalline BaSO (s) that controls the sulfate concentrations. In these experiments the BaSO
(s) that controls the sulfate concentrations. In these experiments the BaSO component of the solid solution phase never reaches thermodynamic equilibrium with the aqueous phase. Thermodynamic interpretations of the data show that both the ideal BaSeO
component of the solid solution phase never reaches thermodynamic equilibrium with the aqueous phase. Thermodynamic interpretations of the data show that both the ideal BaSeO solid solution phase and less-crystalline BaSO
solid solution phase and less-crystalline BaSO (s) phase are in equilibrium with each other in the entire range of BaSeO
(s) phase are in equilibrium with each other in the entire range of BaSeO mole fractions investigated in this study.
mole fractions investigated in this study.