A Theoretical cell-killing model to evaluate oxygen enhancement ratios at DNA damage and cell survival endpoints in radiation therapy
Matsuya, Yusuke 
 ; Sato, Tatsuhiko
; Sato, Tatsuhiko 

 ; Nakamura, Rui*; Naijo, Shingo*; Date, Hiroyuki*
; Nakamura, Rui*; Naijo, Shingo*; Date, Hiroyuki*
Radio-resistance induced under low oxygen pressure plays an important role in malignant progression in fractionated radiotherapy. For the general approach to predict cell killing under hypoxia, cell-killing models (e.g., the Linear-Quadratic model) have to be fitted to 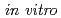 experimental survival data for both normoxia and hypoxia to obtain the oxygen enhancement ratio (OER). However, model parameters for every oxygen condition needs to be considered by model-fitting approaches. This is inefficient for fractionated irradiation planning. Here, we present an efficient model for fractionated radiotherapy the integrated microdosimetric-kinetic model including cell-cycle distribution and the OER at DNA double-strand break endpoint. The cell survival curves described by this model can reproduce the
experimental survival data for both normoxia and hypoxia to obtain the oxygen enhancement ratio (OER). However, model parameters for every oxygen condition needs to be considered by model-fitting approaches. This is inefficient for fractionated irradiation planning. Here, we present an efficient model for fractionated radiotherapy the integrated microdosimetric-kinetic model including cell-cycle distribution and the OER at DNA double-strand break endpoint. The cell survival curves described by this model can reproduce the 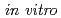 experimental survival data for both acute and chronic low oxygen concentrations. The OER
experimental survival data for both acute and chronic low oxygen concentrations. The OER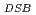 used for calculating cell survival agrees well with experimental DSB ratio of normoxia to hypoxia. This work provides biological effective dose (BED) under various oxygen conditions including its uncertainty, which can contribute to creating fractionated regimens for multi-fractionated radiotherapy. If the oxygen concentration in a tumor can be quantified by medical imaging, the present model will make it possible to estimate the cell-killing and BED under hypoxia in more realistic intravital situations.
used for calculating cell survival agrees well with experimental DSB ratio of normoxia to hypoxia. This work provides biological effective dose (BED) under various oxygen conditions including its uncertainty, which can contribute to creating fractionated regimens for multi-fractionated radiotherapy. If the oxygen concentration in a tumor can be quantified by medical imaging, the present model will make it possible to estimate the cell-killing and BED under hypoxia in more realistic intravital situations.