Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
安達 基泰; 前川 京子*; 松澤 由美子*; 斎藤 嘉朗*; 黒木 良太
no journal, ,
CYP2C9は、シトクロームP450に属し、ヒトの薬物代謝に関わる重要な酵素の一つである。本研究では、野生型CYP2C9及びそのSNPに由来する*30による薬物代謝の分子機構を解明するため、それらのCYP2C9と薬物との複合体のX線結晶構造解析を実施した。CYP2C9は、N末端領域を削除した遺伝子を大腸菌で発現および精製したものを、結晶化試料として用いた。ロサルタンとの複合体の結晶化は、共結晶化法により実施した。得られた結晶を用いて、放射光によるX線回折実験を行い、それぞれ2.1 分解能の回折データを取得した。立体構造解析の結果、結合した薬物の電子密度が観測された。
分解能の回折データを取得した。立体構造解析の結果、結合した薬物の電子密度が観測された。
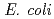 and their activities toward arachidonic acid
and their activities toward arachidonic acid 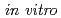
前川 京子*; 松澤 由美子*; 安達 基泰; 黒木 良太; 斎藤 嘉朗*
no journal, ,
Cytochrome P450 2C9 (CYP2C9) is a polymorphic enzyme responsible for the oxidative metabolism of up to 15% of the drugs that undergo phase I metabolism. We previously reported that 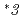 and
and 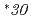 , which were observed at allele frequencies of about 3% and 0.2% in Japanese, respectively. This study was aimed to access their catalytic activities toward endogenous arachidonic acid
, which were observed at allele frequencies of about 3% and 0.2% in Japanese, respectively. This study was aimed to access their catalytic activities toward endogenous arachidonic acid 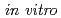 . The N-terminal modified and C-terminal His-tagged wild-type CYP2C9, CYP2C9
. The N-terminal modified and C-terminal His-tagged wild-type CYP2C9, CYP2C9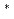 3 and CYP2C9
3 and CYP2C9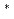 30 were expressed in
30 were expressed in 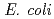 Rosetta2 cells. After cells were lysed, proteins were extracted from membranes using Cymal-5 and purified using Ni-affinity and CM-Sepharose ion exchange columns. Both variants exhibited significantly lower Luciferein-H hydroxylation activities than the wild-type. Arachidonic acid were metabolized into bioactive lipids such as 14,15-epoxyeicosatrienoic acid and 19-hydroxyeicosatetraenoic acid, but their production by variant enzymes was much lower than those by the wild-type.
Rosetta2 cells. After cells were lysed, proteins were extracted from membranes using Cymal-5 and purified using Ni-affinity and CM-Sepharose ion exchange columns. Both variants exhibited significantly lower Luciferein-H hydroxylation activities than the wild-type. Arachidonic acid were metabolized into bioactive lipids such as 14,15-epoxyeicosatrienoic acid and 19-hydroxyeicosatetraenoic acid, but their production by variant enzymes was much lower than those by the wild-type.