Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
村上 昌雄*; 菱川 良夫*; 宮島 悟史*; 岡崎 良子; Sutherland, K.*; 阿部 光幸*; Bulanov, S. V.; 大道 博行; Esirkepov, T. Z.; Koga, J. K.; et al.
AIP Conference Proceedings 1024, p.275 - 300, 2008/08
光医療研究連携センターで進める事業の中心的治療器の開発の概念について考えを提示する。
飯田 靖彦*; 花岡 宏史*; 片渕 竜也*; 渡辺 智; 石岡 典子; 松橋 信平; 織内 昇*; 樋口 徹也*; 宮久保 満之*; 遠藤 啓吾*
JAEA-Review 2006-042, JAEA Takasaki Annual Report 2005, P. 165, 2007/02
Radioimmunotherapy (RIT) becomes one of the most promising treatments for cancer therapy. Recently, because various radioisotopes are being used for RIT, the efficient tailor-made therapy is thought to be possible. The high energy  -particle-emitter, such as
-particle-emitter, such as  Y (2.28 MeV), seems particularly well suited for the treatment of relatively large tumor masses, but the middle energy
Y (2.28 MeV), seems particularly well suited for the treatment of relatively large tumor masses, but the middle energy  -particles, such as
-particles, such as 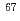 Cu (0.18-0.58 MeV), may have an advantage in treating small lesions. In this study, we prepared
Cu (0.18-0.58 MeV), may have an advantage in treating small lesions. In this study, we prepared 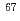 Cu and evaluated its potential of application for RIT in tumor bearing mice. Our results of tumor localization studies show that
Cu and evaluated its potential of application for RIT in tumor bearing mice. Our results of tumor localization studies show that 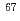 Cu was expected to be suitable for RIT.
Cu was expected to be suitable for RIT.
飯田 靖彦*; 花岡 宏史*; 吉岡 弘樹*; 宮久保 満之*; Paudyal, B.*; 樋口 徹也*; 織内 昇*; 遠藤 啓吾*; 片渕 竜也*; 石岡 典子; et al.
no journal, ,
近年、がん診断における F標識グルコース(
F標識グルコース( F-FDG)の有用性が明らかとなり、多数の施設においてPETによる画像診断が行われるようになっている。しかしながら、現在PETに用いられている放射性同位元素はいずれも半減期が短く、その利用には制限があるのが現状である。そこで、製薬会社において製造し、医療現場への供給が可能な半減期を有する新規PET用核種の製造とそれを用いた放射性薬剤の開発・有効性の確認に関する研究を進めている。本シンポジウムにおいては、新規核種として開発を進めている、
F-FDG)の有用性が明らかとなり、多数の施設においてPETによる画像診断が行われるようになっている。しかしながら、現在PETに用いられている放射性同位元素はいずれも半減期が短く、その利用には制限があるのが現状である。そこで、製薬会社において製造し、医療現場への供給が可能な半減期を有する新規PET用核種の製造とそれを用いた放射性薬剤の開発・有効性の確認に関する研究を進めている。本シンポジウムにおいては、新規核種として開発を進めている、 Cu,
Cu, 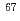 Cu,
Cu, 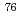 Br,
Br,  Luについて、それぞれの核種の特徴及び研究開発の進捗を紹介する。
Luについて、それぞれの核種の特徴及び研究開発の進捗を紹介する。
 Cu for immuno-PET; In vivo measurement of radiation dose for radioimmunotherapy
Cu for immuno-PET; In vivo measurement of radiation dose for radioimmunotherapy飯田 靖彦*; 花岡 宏史*; 片渕 竜也*; 渡辺 智; 石岡 典子; 松橋 信平; 織内 昇*; 樋口 徹也*; 宮久保 満之*; 遠藤 啓吾*
no journal, ,
Radioimmunotherapy (RIT) becomes one of the most promising treatments for cancer therapy. Recently, RIT becomes available for clinical use and shows high efficacy, but it has some adverse effects for radiation to normal tissues and its therapeutic window is limited. So, assessment of radiation dose deliver to both tumor and normal tissues is very important for RIT. PET is superior in quantitative measurement and it can estimate radiation dose directly. Although representative positron emitter has short half-life, which is not good for labeling of antibody,  Cu has appropriate property, T
Cu has appropriate property, T = 12.7 hr, and it may have a great potential for immuno-PET. In this study, we prepared a radioimmunoconjugate, NuB2, anti-CD20 monoclonal antibody (mAb), labeled with
= 12.7 hr, and it may have a great potential for immuno-PET. In this study, we prepared a radioimmunoconjugate, NuB2, anti-CD20 monoclonal antibody (mAb), labeled with  Cu, and evaluated potential for in vivo quantitative measurement of radiation dose for RIT. NuB2 was labeled with
Cu, and evaluated potential for in vivo quantitative measurement of radiation dose for RIT. NuB2 was labeled with  Cu. For in vivo studies, SCID mice bearing CD20
Cu. For in vivo studies, SCID mice bearing CD20 tumor were used. After administration, biodistribution and PET imaging were performed. For the results of biodistribution,
tumor were used. After administration, biodistribution and PET imaging were performed. For the results of biodistribution,  Cu-NuB2 was highly accumulated to CD20
Cu-NuB2 was highly accumulated to CD20 tumor, and PET images could show the same results. From these date, the use of
tumor, and PET images could show the same results. From these date, the use of  Cu immuno-PET has potential for accurately measurement of radiation dose for RIT.
Cu immuno-PET has potential for accurately measurement of radiation dose for RIT.
 Cu for quantitative evaluation of radioimmunotherapy
Cu for quantitative evaluation of radioimmunotherapy飯田 靖彦*; 花岡 宏史*; 片渕 竜也*; 渡辺 智; 石岡 典子; 松橋 信平; 織内 昇*; 樋口 徹也*; 宮久保 満之*; 遠藤 啓吾*
no journal, ,
 Cu is one of the most attractive candidate emitter for radioimmunotherapy (RIT).
Cu is one of the most attractive candidate emitter for radioimmunotherapy (RIT).  Cu releases positron emitter (0.653 MeV, 18%) as well as middle energy
Cu releases positron emitter (0.653 MeV, 18%) as well as middle energy  -particles (0.578 MeV, 37%), so it can be applied for diagnostic imaging with PET. PET is superior in quantitative measurement and suitable for evaluation of therapeutic effect with RIT. PET can also directly estimate radiation dose of RIT. In this study, we prepared a radioimmunoconjugate, NuB2, anti-CD20 monoclonal antibody (mAb), labeled with
-particles (0.578 MeV, 37%), so it can be applied for diagnostic imaging with PET. PET is superior in quantitative measurement and suitable for evaluation of therapeutic effect with RIT. PET can also directly estimate radiation dose of RIT. In this study, we prepared a radioimmunoconjugate, NuB2, anti-CD20 monoclonal antibody (mAb), labeled with  Cu, and evaluated its potential for PET imaging with RIT. NuB2 was labeled with
Cu, and evaluated its potential for PET imaging with RIT. NuB2 was labeled with  Cu. For in vivo studies, SCID mice bearing CD20
Cu. For in vivo studies, SCID mice bearing CD20 tumor were used. After administration, biodistribution and PET imaging were performed. For the results of biodistribution,
tumor were used. After administration, biodistribution and PET imaging were performed. For the results of biodistribution,  Cu-NuB2 was highly accumulated to CD20
Cu-NuB2 was highly accumulated to CD20 tumor, and PET images could show the same results. From these date, the use of
tumor, and PET images could show the same results. From these date, the use of  Cu for PET imaging has potential for accurately evaluation of therapeutic effect of RIT.
Cu for PET imaging has potential for accurately evaluation of therapeutic effect of RIT.
村上 昌雄*; 出水 祐介*; 丹羽 康江*; 永山 伸一*; 前田 拓也*; 馬場 理師*; 宮脇 大輔*; 寺嶋 千貴*; 有村 健*; 美馬 正幸*; et al.
no journal, ,
The Hyogo Ion Beam Medical Center was established in May 2001, a leading project of the "Hyogo Cancer Strategy". The accelerator is a synchrotron that can accelerate proton and carbon ion beams at a maximum of 230 and 320 MeV/u, respectively, and the maximum ranges in water are 300 and 200 mm, respectively. Three irradiation rooms installed with 45-degree, horizontal/vertical, and horizontal fixed ports can be used for carbon ion radiation therapy, and 2 gantry rooms can be additionally used for proton beams. Particle beam radiation therapy had been performed in 2,639 patients as of the end of March 2009. The diseases treated were prostate cancer, head and neck tumors, liver cancer, lung cancer, and bone soft tissue tumors, in decreasing order of frequency, and these 5 major diseases accounted for 87% of the cases. As the current problems of particle beam radiation therapy, the effect of the differential use of proton and carbon ion beams is unclear, adverse events, such as skin disorders, may occur due to the limitation of the broad beam method, and the necessity to install large-scale devices is an obstacle to its dissemination. We are aiming at the development and clinical application of a laser-driven proton radiotherapy device in cooperation with the Japan Atomic Energy Agency.
Bolton, P.; 阿部 光幸*; 赤城 卓*; Nuesslin, F.*; 堀 利彦; 岩下 芳久*; 河西 俊一; 近藤 公伯; 前田 拓也; Molls, M.*; et al.
no journal, ,
The rapid advancement of high power laser technology combined with laser-accelerated ion yields from intense laser-plasma interactions sustains a strong interest in the development of integrated laser-driven ion accelerator systems (ILDIAS) that can be used for laser-driven ion beam radiotherapy (L-IBRT). A prime motivation is the promise of significantly reduced size and cost that would afford much greater patient access. Bunch duration of several nanoseconds and high peak current (with a low duty factor) make laser-driven ion irradiation unique. En route to a compact laser-driven "clinical" radiotherapeutic facility it is essential to develop multipurpose "preclinical" or test beamlines that can be used for (1) testing suitable transport optics, diagnostics and control instrumentation, (2) medical and radiobiological studies at the cellular level and in tissue to validate the radiobiological effectiveness of laser-driven ion beam radiotherapy and (3) applications to nonmedical science and technology. Preclinical and clinical beamline development calls for delivering proton energies near 50 MeV and at least 80 MeV respectively with beam energy spread and dose accuracy at one to few percent levels. Control instrumentation and diagnostics capable of single bunch resolution at required repetition rates will be essential to verify and optimize ILDIAS machine performance and capability. In the context of L-IBRT we will discuss the ILDIAS concept as well as beam delivery and instrumentation requirements.