Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
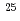 F from
F from 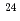 O
O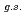 ?
?Tang, T. L.*; Uesaka, Tomohiro*; Kawase, Shoichiro; Beaumel, D.*; Dozono, Masanori*; Fujii, Toshihiko*; Fukuda, Naoki*; Fukunaga, Taku*; Galindo-Uribarri, A.*; Hwang, S. H.*; et al.
Physical Review Letters, 124(21), p.212502_1 - 212502_6, 2020/05
Times Cited Count:19 Percentile:72.96(Physics, Multidisciplinary)The structure of a neutron-rich  F nucleus is investigated by a quasifree (
F nucleus is investigated by a quasifree (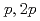 ) knockout reaction. The sum of spectroscopic factors of
) knockout reaction. The sum of spectroscopic factors of 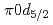 orbital is found to be 1.0
orbital is found to be 1.0  0.3. The result shows that the
0.3. The result shows that the  O core of
O core of  F nucleus significantly differs from a free
F nucleus significantly differs from a free 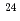 O nucleus, and the core consists of
O nucleus, and the core consists of  35%
35% 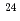 O
O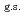 , and
, and  65% excited
65% excited 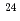 O. The result shows that the
O. The result shows that the 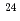 O core of
O core of 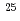 F nucleus significantly differs from a free
F nucleus significantly differs from a free 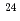 O nucleus. The result may infer that the addition of the
O nucleus. The result may infer that the addition of the 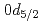 proton considerably changes the neutron structure in
proton considerably changes the neutron structure in 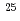 F from that in
F from that in 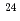 O, which could be a possible mechanism responsible for the oxygen dripline anomaly.
O, which could be a possible mechanism responsible for the oxygen dripline anomaly.
Nakano, Masanao; Fujii, Tomoko; Nagaoka, Mika; Inoue, Kazumi; Koike, Yuko; Yamada, Ryohei; Yoshii, Hideki*; Otani, Kazunori*; Hiyama, Yoshinori*; Kikuchi, Masaaki*; et al.
JAEA-Review 2019-045, 120 Pages, 2020/03
Based on the regulations (the safety regulation of Tokai Reprocessing Plant, the safety regulation of nuclear fuel material usage facilities, the radiation safety rule, the regulation about prevention from radiation hazards due to radioisotopes, which are related with the nuclear regulatory acts, the local agreement concerning with safety and environment conservation around nuclear facilities, the water pollution control law, and by law of Ibaraki Prefecture), the effluent control of liquid waste discharged from the Nuclear Fuel Cycle Engineering Laboratories of Japan Atomic Energy Agency has been performed. This report describes the effluent control results of the liquid waste in the fiscal year 2018. In this period, the concentrations and the quantities of the radioactivity in liquid waste discharged from the reprocessing plant, the plutonium fuel fabrication facilities, and the other nuclear fuel material usage facilities were much lower than the limits authorized by the above regulations.
Sakuma, Hiroshi*; Tachi, Yukio; Yotsuji, Kenji; Suehara, Shigeru*; Arima, Tatsumi*; Fujii, Naoki*; Kawamura, Katsuyuki*; Honda, Akira
Clays and Clay Minerals, 65(4), p.252 - 272, 2017/08
Times Cited Count:2 Percentile:15.19(Chemistry, Physical)Structure and stability of montmorillonite edge faces (110), (010), (100), and (130) of the layer charges y = 0.5 and 0.33 are investigated by the first-principles electronic calculations based on the density functional theory. Stacking and single layer models are tested for understanding the effect of stacking on the stability of montmorillonite edge faces. Most stacking layers stabilize the edge faces by making hydrogen bonds between the layers; therefore, the surface energy of stacking layers is reduced rather than the single layer model. This indicates that the surface energy of edge faces should be estimated depending on the swelling conditions. Lowest surface energies of (010) and (130) edge faces were realized by the presence of Mg ions on the edge faces. These edge faces have a strong adsorption site for cations due to local negative charge of the edges.
Matsue, Naoto*; Abidin, Z.*; Fujii, Naoki*; Oda, Chie; Honda, Akira
Nendo Kagaku, 52(2), p.62 - 70, 2014/00
Bentonite is expected to be used along with cement as engineered barrier components of TRU waste geological disposal system. There is a concern that the bentonite dissolved or altered owing to the contact with strong alkaline and high Ca concentration water derived from the reaction of the cement and groundwater. The first-principles quantum chemical calculations using the cluster model of montmorillonite, the main component of bentonite, were performed, and it was indicated that octahedral Al has higher solubility than octahedral Mg under strong alkaline condition and unsaturated Si-O-Al bond connecting tetrahedral and octahedral sheets at the edge of 110 and 010 planes cleaves easily. The adsorption of OH on tetrahedral Si atom caused dissolution of adjacent Si and Al atoms in addition to the Si atom. While, the dissociation of Si-OH and Al-OH
on tetrahedral Si atom caused dissolution of adjacent Si and Al atoms in addition to the Si atom. While, the dissociation of Si-OH and Al-OH groups and the difference in exchangeable cation species were shown to be less likely to affect the dissolution behavior of montmorillonite.
groups and the difference in exchangeable cation species were shown to be less likely to affect the dissolution behavior of montmorillonite.
 MnO
MnO (
(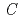 2/
2/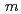 ) for a lithium-battery cathode in several charge-discharge potential ranges
) for a lithium-battery cathode in several charge-discharge potential rangesNakao, Yasuhiro*; Ozawa, Kiyoshi*; Fujii, Hiroki*; Mochiku, Takashi*; Iwai, Hideo*; Tsuchiya, Yoshinori*; Igawa, Naoki
Transactions of the Materials Research Society of Japan, 38(2), p.229 - 233, 2013/12
Li MnO
MnO powder with a monoclinic cell related to the space group of
powder with a monoclinic cell related to the space group of 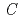 2/
2/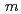 was synthesized by a simplified coprecipitation method. Its electrode properties as a lithium-battery cathode were investigated in the charge-discharge potential ranges of 2.0 - 4.6, 2.0 - 4.8, and 2.0 - 5.0 V. In all the potential ranges, the electrochemical capacities gradually increased during cycling, and the increased capacities significantly depended on the potential ranges. Compared to the discharge capacities for the potential ranges of 2.0 - 4.6 and 2.0 - 5.0 V, the former merely increased from 12.5 to 22.5 mAhg
was synthesized by a simplified coprecipitation method. Its electrode properties as a lithium-battery cathode were investigated in the charge-discharge potential ranges of 2.0 - 4.6, 2.0 - 4.8, and 2.0 - 5.0 V. In all the potential ranges, the electrochemical capacities gradually increased during cycling, and the increased capacities significantly depended on the potential ranges. Compared to the discharge capacities for the potential ranges of 2.0 - 4.6 and 2.0 - 5.0 V, the former merely increased from 12.5 to 22.5 mAhg between the 1st and 99th cycles, whereas the latter increased from 20.0 to 110.2 mAhg
between the 1st and 99th cycles, whereas the latter increased from 20.0 to 110.2 mAhg during the same cycling. The structural variation in Li
during the same cycling. The structural variation in Li MnO
MnO is induced by the cycling, the rhombohedral phase is detected during cycling in the potential range of 2.0 - 5.0 V.
is induced by the cycling, the rhombohedral phase is detected during cycling in the potential range of 2.0 - 5.0 V.
Fukui, Kunihiro*; Igawa, Yusuke*; Arimitsu, Naoki*; Suzuki, Masahiro; Segawa, Tomoomi; Fujii, Kanichi*; Yamamoto, Tetsuya*; Yoshida, Hideto*
Chemical Engineering Journal, 211-212, p.1 - 8, 2012/11
Times Cited Count:13 Percentile:39.33(Engineering, Environmental)The process for synthesizing metallic oxide powders by the microwave denitration method was investigated using hexahydrated nickel nitrate and trihydrated copper nitrate aqueous solutions, and the electrical field and the temperature distributions in the reactor were numerically simulated. Although CuO powder can be obtained from a trihydrated copper nitrate aqueous solution by the microwave denitration method, a hexahydrated nickel nitrate aqueous solution cannot be heated up to over 270  C by microwave irradiation. It was also found that the reaction routes for microwave heating are the same as those for conventional external heating. This finding indicates that the success of producing oxide particles by microwave denitration depends not only on the microwave absorptivity of the intermediate and the metallic oxide, but also on the temperature difference.
C by microwave irradiation. It was also found that the reaction routes for microwave heating are the same as those for conventional external heating. This finding indicates that the success of producing oxide particles by microwave denitration depends not only on the microwave absorptivity of the intermediate and the metallic oxide, but also on the temperature difference.
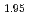 Mn
Mn Co
Co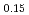 O
O (
(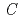 2/
2/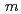 ) as a lithium-battery cathode
) as a lithium-battery cathodeOzawa, Kiyoshi*; Nakao, Yasuhiro*; Mochiku, Takashi*; Cheng, Z.*; Wang, L.*; Iwai, Hideo*; Tsuchiya, Yoshinori*; Fujii, Hiroki*; Igawa, Naoki
Journal of the Electrochemical Society, 159(3), p.A300 - A304, 2012/01
Times Cited Count:14 Percentile:44.40(Electrochemistry)A manganese-based solid solution with the composition of Li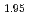 Mn
Mn Co
Co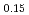 O
O was synthesized by a simplified coprecipitation method, and its electrochemical characteristics as a lithium-battery cathode were investigated. Rietveld refinement based on neutron diffraction data revealed that the material is assigned to an Li
was synthesized by a simplified coprecipitation method, and its electrochemical characteristics as a lithium-battery cathode were investigated. Rietveld refinement based on neutron diffraction data revealed that the material is assigned to an Li MnO
MnO -type structure model with a space group symmetry of
-type structure model with a space group symmetry of 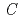 2/
2/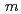 . In cycling of the cell in the potential range from 2.0 to 4.8 V at current densities of 30 mAhg
. In cycling of the cell in the potential range from 2.0 to 4.8 V at current densities of 30 mAhg , the discharge capacity characteristically increases from 46.3 to 196.5 mAhg
, the discharge capacity characteristically increases from 46.3 to 196.5 mAhg as the cycle increases from 1 to 11, and a discharge capacity above 175.5 mAhg
as the cycle increases from 1 to 11, and a discharge capacity above 175.5 mAhg is obtained between the 23rd and 58th cycles. The cyclic voltammogram and X-ray photoelectron spectroscopy measurements showed that the manganese redox reaction is progressively activated during the first ten-odd cycles.
is obtained between the 23rd and 58th cycles. The cyclic voltammogram and X-ray photoelectron spectroscopy measurements showed that the manganese redox reaction is progressively activated during the first ten-odd cycles.
Kimura, Akihiko*; Nagai, Yasuyoshi*; Fujii, Katsuhiko*; Nishiyama, Yutaka; Soneda, Naoki*
Nihon Genshiryoku Gakkai-Shi ATOMO , 50(10), p.630 - 633, 2008/10
, 50(10), p.630 - 633, 2008/10
no abstracts in English
Yamasaki, Chisato*; Murakami, Katsuhiko*; Fujii, Yasuyuki*; Sato, Yoshiharu*; Harada, Erimi*; Takeda, Junichi*; Taniya, Takayuki*; Sakate, Ryuichi*; Kikugawa, Shingo*; Shimada, Makoto*; et al.
Nucleic Acids Research, 36(Database), p.D793 - D799, 2008/01
Times Cited Count:52 Percentile:70.47(Biochemistry & Molecular Biology)Here we report the new features and improvements in our latest release of the H-Invitational Database, a comprehensive annotation resource for human genes and transcripts. H-InvDB, originally developed as an integrated database of the human transcriptome based on extensive annotation of large sets of fulllength cDNA (FLcDNA) clones, now provides annotation for 120 558 human mRNAs extracted from the International Nucleotide Sequence Databases (INSD), in addition to 54 978 human FLcDNAs, in the latest release H-InvDB. We mapped those human transcripts onto the human genome sequences (NCBI build 36.1) and determined 34 699 human gene clusters, which could define 34 057 protein-coding and 642 non-protein-coding loci; 858 transcribed loci overlapped with predicted pseudogenes.
Fujii, Naoki; Ichikawa, Yasuaki*
Oyo Rikigaku Rombunshu, 9, p.323 - 332, 2006/08
Diffusion in bentonite clay is investigated by the homogenization analysis, which related the microscale structural and chemical characteristics to the macroscale behavior. However, the diffusion coefficient of the cation by calculation is smaller than the effective diffusion coefficient by experiment. This difference is caused by the surface diffusion. In order to resolve the surface diffusion by numerical analysis, we formulated the phenomenon owing to the cation thickening model on the clay surface by the homogenization technique. The numerical result showed that the total diffusion increase with the increase in a distribution ratio of cation. The increase in the diffusion flux under the increase in a local concentration gradient causes surface diffusion.
Suzuki, Satoru; Sato, Haruo; Ishidera, Takamitsu; Fujii, Naoki*; Kawamura, Katsuyuki*
JNC TN8400 2001-031, 44 Pages, 2002/05
In order to quantify effect of temperature on diffusivity of deuterated water (HDO) in compacted sodium-bentonite, through-diffusion experiments were conducted at elevated tempemture from 298 to 333 K. Kunipia F (Na-montmorillonite content  98 wt. %; Kunimine Industly Co.) was compacted to a dry density of 0.9 and l.35 Mg/m
98 wt. %; Kunimine Industly Co.) was compacted to a dry density of 0.9 and l.35 Mg/m . Since smectite flakes were perpendicularly oriented to a direction of compaction, anisotropy of diffusivity was investigated parallel and normal to the preferred orientation of smectite. Effective diffusion coeficient D
. Since smectite flakes were perpendicularly oriented to a direction of compaction, anisotropy of diffusivity was investigated parallel and normal to the preferred orientation of smectite. Effective diffusion coeficient D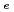 of HDO was larger for a diffusional direction parallel to the preferred orientation than normal to that for both dry densities. These results well agreed to the previously reported ones for tritiated water. Activation energies of D
of HDO was larger for a diffusional direction parallel to the preferred orientation than normal to that for both dry densities. These results well agreed to the previously reported ones for tritiated water. Activation energies of D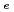 in compacted bentonite increased with increasing dry density in the range of 19 - 25 kJ/mol which was slightly larger than that in bulk water (18 kJ/mol). This relationship can be considered to be due to both the pore structure development and high activation energy of water (18-23 kJ/mol) in the vicinity of smectite surface (within 2 nm) on the basis of molecular dynamics simulations.
in compacted bentonite increased with increasing dry density in the range of 19 - 25 kJ/mol which was slightly larger than that in bulk water (18 kJ/mol). This relationship can be considered to be due to both the pore structure development and high activation energy of water (18-23 kJ/mol) in the vicinity of smectite surface (within 2 nm) on the basis of molecular dynamics simulations.
Suzuki, Satoru; Fujishima, Atsushi; Ueno, Kenichi; Ichikawa, Yasuaki*; Kawamura, Katsuyuki*; Fujii, Naoki*; Shibata, Masahiro; Sato, Haruo; Kitayama, Kazumi*
Nendo Kagaku, 41(2), p.43 - 57, 2001/12
None
Suzuki, Satoru; Fujishima, Atsushi; Ueno, Kenichi; Ichikawa, Yasuaki*; Kawamura, Katsuyuki*; Fujii, Naoki*; Shibata, Masahiro; Sato, Haruo
JNC TY8400 2001-003, 66 Pages, 2001/05
no abstracts in English
Segawa, Tomoomi; Suzuki, Masahiro; Fujii, Kanichi; Igawa, Yusuke*; Arimitsu, Naoki*; Yamamoto, Tetsuya*; Fukui, Kunihiro*; Yoshida, Hideto*
no journal, ,
The synthesis process of the denitration reaction by the microwave heating method was investigated using a nickel nitrate (Ni(NO )
) ) aqueous solution. NiO powder cannot be obtained from Ni(NO
) aqueous solution. NiO powder cannot be obtained from Ni(NO )
) aqueous solution by the microwave heating method because of low microwave absorption of the intermediate obtained from nitrate aqueous solution. In the present work, it was showed that Ni(NO
aqueous solution by the microwave heating method because of low microwave absorption of the intermediate obtained from nitrate aqueous solution. In the present work, it was showed that Ni(NO )
) aqueous solution with 6.0 g of NiO powder as a microwave absorber can be completely converted to NiO powder by microwave heating. NiO powder promotes the denitration reaction, and reduces the required reaction time with increasing the amount of NiO powder. Furthermore the adiabator was set around the reactor in order to equalize the temperature distribution in the reactor. It was also found that the denitration efficiency of Ni(NO
aqueous solution with 6.0 g of NiO powder as a microwave absorber can be completely converted to NiO powder by microwave heating. NiO powder promotes the denitration reaction, and reduces the required reaction time with increasing the amount of NiO powder. Furthermore the adiabator was set around the reactor in order to equalize the temperature distribution in the reactor. It was also found that the denitration efficiency of Ni(NO )
) aqueous solution to NiO powder can be improved by using the adiabator when the center of the reactor has the same temperature.
aqueous solution to NiO powder can be improved by using the adiabator when the center of the reactor has the same temperature.
Maekawa, Keisuke; Makino, Hitoshi; Umeki, Hiroyuki; Hioki, Kazumasa; Kaneko, Yu*; Takase, Hiroyasu*; Fujii, Naoki*
no journal, ,
no abstracts in English
Fujii, Naoki; Kawamura, Katsuyuki*
no journal, ,
no abstracts in English
Shinoda, Yoshihiko; Hayashizaki, Noriyosu*; Shioda, Akiko*; Yamano, Naoki*; Fujii, Yasuhiko*; Torii, Hiroyuki*
no journal, ,
The social research group of 21st century COE program carries out community experiments on mutual evoluation of companies in general, local community and nuclear technology. Oarai town of Ibaraki for community as the case study and it is the purpose of mutual evoluation to seek the nuclear technology utilizing Oarai town. In this study, we sent out questionnaire of awareness about Oarai town and energy issues toward visitor and analyzed it.
Kuwabara, Shogo*; Nara, Yoshitaka*; Kashiwaya, Koki*; Fujii, Hirokazu*; Zhao, Y.*; Matsui, Hiroya; Ozaki, Yusuke; Hiroyoshi, Naoki*
no journal, ,
This study aims to develop a healing method of fractured rock by injection of cement powder and carbonated water to form carbonated minerals. The in-situ hydraulic tests were carried out to confirm the effect of their injection into rock. The decrease of hydraulic conductivity was recognized after the injection of both cement powder and carbonated water, whereas the injection of only carbonated water did not induce the decrease of hydraulic conductivity.