Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -dioctylthiodiglycolamic acid; Effect of S donor on metal extraction
-dioctylthiodiglycolamic acid; Effect of S donor on metal extractionShimojo, Kojiro; Fujiwara, Iori*; Saito, Takumi*; Oshima, Tatsuya*
Analytical Sciences, 40(8), p.1429 - 1436, 2024/08
Times Cited Count:0 Percentile:0.00(Chemistry, Analytical)Extraction ability of  -dioctylthiodiglycolamic acid (T-DODGAA), a soft-base sulfur donor ligand with an amide group and a carboxylic acid connected by a thioether chain, for 56 metal ions have been comprehensively investigated and compared with that of N,N-dioctyldiglycolamic acid (DODGAA) with an etheric oxygen atom, a hard-base donor. The p
-dioctylthiodiglycolamic acid (T-DODGAA), a soft-base sulfur donor ligand with an amide group and a carboxylic acid connected by a thioether chain, for 56 metal ions have been comprehensively investigated and compared with that of N,N-dioctyldiglycolamic acid (DODGAA) with an etheric oxygen atom, a hard-base donor. The p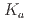 of the thiodiglycolamic acid framework was determined to be 3.71
of the thiodiglycolamic acid framework was determined to be 3.71  0.06 in water (0.1 M LiCl, 25
0.06 in water (0.1 M LiCl, 25 C ) by potentiometric titration, indicating that T-DODGAA is a slightly weaker acid than DODGAA (p
C ) by potentiometric titration, indicating that T-DODGAA is a slightly weaker acid than DODGAA (p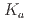 = 3.54
= 3.54  0.03). T-DODGAA can quantitatively extract various metal ions from the 56 metal ions through a proton-exchange reaction. T-DODGAA provided higher extraction performance than DODGAA for Hf(IV), Cr(III), Fe(III), Ni(II), Cu(II), Pd(II), Ag(I), Au(III), Hg(II), Al(III), and Ga(III), especially for soft metal ions. Furthermore, to demonstrate the practical feasibility of T-DODGAA for hydrometallurgy and metal recycling, we performed selective separation tests of rare metal ions such as Sc(III), Ni(II), Co(II), Pd(II), Au(III), In(III), and Ga(II) in metal-mixed extraction systems.
0.03). T-DODGAA can quantitatively extract various metal ions from the 56 metal ions through a proton-exchange reaction. T-DODGAA provided higher extraction performance than DODGAA for Hf(IV), Cr(III), Fe(III), Ni(II), Cu(II), Pd(II), Ag(I), Au(III), Hg(II), Al(III), and Ga(III), especially for soft metal ions. Furthermore, to demonstrate the practical feasibility of T-DODGAA for hydrometallurgy and metal recycling, we performed selective separation tests of rare metal ions such as Sc(III), Ni(II), Co(II), Pd(II), Au(III), In(III), and Ga(II) in metal-mixed extraction systems.
Shimojo, Kojiro; Fujiwara, Iori*; Oshima, Tatsuya*; Yokoyama, Keiichi; Yaita, Tsuyoshi
Analytical Sciences, 38(7), p.1003 - 1006, 2022/07
Times Cited Count:2 Percentile:13.92(Chemistry, Analytical)Liquid-liquid extraction of lanthanide (Ln) ions was investigated using  -dioctylthiodiglycolamic acid (DOTDGAA), which is a sulfur donor ligand with an amide group and a carboxyl group connected by a thioether chain. The extraction performance and selectivity of DOTDGAA for Ln ions were compared with those of
-dioctylthiodiglycolamic acid (DOTDGAA), which is a sulfur donor ligand with an amide group and a carboxyl group connected by a thioether chain. The extraction performance and selectivity of DOTDGAA for Ln ions were compared with those of  -dioctyldiglycolamic acid (DODGAA), which is also an oxygen donor ligand with a similar chemical structure, to assess the effect of the soft/hard donor atom on Ln separation. DOTDGAA quantitatively extracted all Ln ions while being selective toward light and middle Ln ions, in contrast to the selectivity of DODGAA for heavier Ln ions. Slope analysis demonstrated that the Ln
-dioctyldiglycolamic acid (DODGAA), which is also an oxygen donor ligand with a similar chemical structure, to assess the effect of the soft/hard donor atom on Ln separation. DOTDGAA quantitatively extracted all Ln ions while being selective toward light and middle Ln ions, in contrast to the selectivity of DODGAA for heavier Ln ions. Slope analysis demonstrated that the Ln transfer using DOTDGAA proceeded through a proton-exchange reaction, forming a 1:3 complex, Ln(DOTDGAA)
transfer using DOTDGAA proceeded through a proton-exchange reaction, forming a 1:3 complex, Ln(DOTDGAA) . The back-extraction of Ln ions from the extracting phase was successfully achieved under acidic conditions.
. The back-extraction of Ln ions from the extracting phase was successfully achieved under acidic conditions.
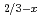 Li
Li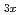 TiO
TiO (x = 0.13) studied by quasielastic neutron scattering
(x = 0.13) studied by quasielastic neutron scatteringMatsuura, Masato*; Fujiwara, Yasuyuki*; Moriwake, Hiroki*; Ohara, Koji*; Kawakita, Yukinobu
Physical Review B, 104(9), p.094305_1 - 094305_7, 2021/09
Times Cited Count:8 Percentile:42.22(Materials Science, Multidisciplinary)Sugita, Tsuyoshi; Fujiwara, Iori*; Okamura, Hiroyuki; Oshima, Tatsuya*; Baba, Yoshinari*; Naganawa, Hirochika; Shimojo, Kojiro
Solvent Extraction Research and Development, Japan, 24(2), p.61 - 69, 2017/05
We investigated an influence of amide group in diglycolamic acid-type extractants on extraction property of metal ions. The extraction characteristics of  -dodecyldiglycolamic acid (C
-dodecyldiglycolamic acid (C DGAA), with a secondary amide group, for 56 metal ions have been investigated, and compared with those of
DGAA), with a secondary amide group, for 56 metal ions have been investigated, and compared with those of  -dioctyldiglycolamic acid (DODGAA) with a tertiary amide group. Compared with DODGAA, C
-dioctyldiglycolamic acid (DODGAA) with a tertiary amide group. Compared with DODGAA, C DGAA has a poor extraction performance and separation ability for rare-earth metal ions, except for Sc(III). However, C
DGAA has a poor extraction performance and separation ability for rare-earth metal ions, except for Sc(III). However, C DGAA tended to provide better extraction for relatively small-sized metal ions than DODGAA. In addition, it was found that C
DGAA tended to provide better extraction for relatively small-sized metal ions than DODGAA. In addition, it was found that C DGAA enables the selective removal of Hg(II) from aqueous solutions containing various divalent metal ions.
DGAA enables the selective removal of Hg(II) from aqueous solutions containing various divalent metal ions.
Shimojo, Kojiro; Fujiwara, Iori*; Fujisawa, Kiyoshi*; Okamura, Hiroyuki; Sugita, Tsuyoshi; Oshima, Tatsuya*; Baba, Yoshinari*; Naganawa, Hirochika
Solvent Extraction Research and Development, Japan, 23(2), p.151 - 159, 2016/05
Liquid-liquid extraction of rare-earth (RE) cations has been investigated using  -dodecyldiglycolamic acid (C
-dodecyldiglycolamic acid (C DGAA) with a secondary amide group, and compared with that using
DGAA) with a secondary amide group, and compared with that using  -dioctyldiglycolamic acid (DODGAA) with a tertiary amide group. C
-dioctyldiglycolamic acid (DODGAA) with a tertiary amide group. C DGAA enables quantitative transfer of all RE cations from moderately acidic solution, while being selective toward the heavier RE cations, and performs better than typical carboxylic-acid-type extractants. However, C
DGAA enables quantitative transfer of all RE cations from moderately acidic solution, while being selective toward the heavier RE cations, and performs better than typical carboxylic-acid-type extractants. However, C DGAA provides low extraction performance and separation ability for RE cations compared with DODGAA because of the weaker basicity of the amide oxygen. Slope analysis demonstrated that RE
DGAA provides low extraction performance and separation ability for RE cations compared with DODGAA because of the weaker basicity of the amide oxygen. Slope analysis demonstrated that RE transfer with C
transfer with C DGAA proceeded through a proton-exchange reaction, forming a 1:3 complex, RE(C
DGAA proceeded through a proton-exchange reaction, forming a 1:3 complex, RE(C DGAA)
DGAA) . Structural characterization by X-ray diffraction revealed that three
. Structural characterization by X-ray diffraction revealed that three  -butyldiglycolamic acid (C
-butyldiglycolamic acid (C DGAA) molecules coordinated to the La
DGAA) molecules coordinated to the La central ion in a tridentate fashion and the La
central ion in a tridentate fashion and the La primary coordination sphere consisted of three oxygen atoms from the amide group, three oxygen atoms from the ether group, and three oxygen atoms from the carboxy group.
primary coordination sphere consisted of three oxygen atoms from the amide group, three oxygen atoms from the ether group, and three oxygen atoms from the carboxy group.
Tashiro, Koji*; Hanesaka, Makoto*; Yamamoto, Hiroko*; Wasanasuk, K.*; Jayaratri, P.*; Yoshizawa, Yoshinori*; Tanaka, Ichiro*; Niimura, Nobuo*; Kusaka, Katsuhiro*; Hosoya, Takaaki*; et al.
Kobunshi Rombunshu, 71(11), p.508 - 526, 2014/11
Times Cited Count:6 Percentile:20.64(Polymer Science)The crystal structure analysis of various polymer substances has been reviewed on the basis of wide-angle high-energy X-ray and neutron diffraction data. The progress in structural analytical techniques of polymer crystals have been reviewed at first. The structural models proposed so far were reinvestigated and new models have been proposed for various kinds of polymer crystals including polyethylene, poly(vinyl alcohol), poly(lactic acid) and its stereocomplex etc. The hydrogen atomic positions were also clarified by the quantitative analysis of wide-angle neutron diffraction data, from which the physical properties of polymer crystals have been evaluated theoretically. The bonded electron density distribution has been estimated for a polydiacetylene single crystal on the basis of the so-called X-N method or by the combination of structural information derived from X-ray and neutron diffraction data analysis. Some comments have been added about future developments in the field of structure-property relationship determination.
Ishihara, Ryo*; Fujiwara, Kunio*; Harayama, Takato*; Okamura, Yusuke*; Uchiyama, Shoichiro*; Sugiyama, Mai*; Someya, Takaaki*; Amakai, Wataru*; Umino, Satoshi*; Ono, Tsubasa*; et al.
Journal of Nuclear Science and Technology, 48(10), p.1281 - 1284, 2011/10
Times Cited Count:44 Percentile:93.87(Nuclear Science & Technology)Yanagie, Hironobu*; Kumada, Hiroaki*; Nakamura, Takemi; Higashi, Shushi*; Ikushima, Ichiro*; Morishita, Yasuyuki*; Shinohara, Atsuko*; Fujiwara, Mitsuteru*; Suzuki, Minoru*; Sakurai, Yoshinori*; et al.
Proceedings of 14th International Congress on Neutron Capture Therapy (ICNCT-14) (CD-ROM), p.157 - 160, 2010/10
Morita, Takami*; Niwa, Kentaro*; Fujimoto, Ken*; Kasai, Hiromi*; Yamada, Haruya*; Nishiuchi, Ko*; Sakamoto, Tatsuya*; Godo, Waichiro*; Taino, Seiya*; Hayashi, Yoshihiro*; et al.
Science of the Total Environment, 408(16), p.3443 - 3447, 2010/06
Times Cited Count:17 Percentile:37.12(Environmental Sciences)Iodine-131 ( I) was detected in brown algae collected off the Japanese coast. The maximum measured specific activity of
I) was detected in brown algae collected off the Japanese coast. The maximum measured specific activity of  I in brown algae was 0.37
I in brown algae was 0.37 0.010 Bq/kg-wet. Cesium-137 (
0.010 Bq/kg-wet. Cesium-137 ( Cs) was also detected in all brown algal samples used in this study. There was no correlation between specific activities of
Cs) was also detected in all brown algal samples used in this study. There was no correlation between specific activities of  I and
I and  Cs in these seaweeds. Low specific activity and minimal variability of
Cs in these seaweeds. Low specific activity and minimal variability of  Cs in brown algae indicated that past nuclear weapon tests were the source of
Cs in brown algae indicated that past nuclear weapon tests were the source of  Cs. Although nuclear power facilities are known to be pollution sources of
Cs. Although nuclear power facilities are known to be pollution sources of  I, there was no relationship between the sites where
I, there was no relationship between the sites where  I was detected and the locations of nuclear power facilities. Most of the sites where
I was detected and the locations of nuclear power facilities. Most of the sites where  I was detected were near big cities with large populations. On the basis of the results, we suggest that the likely pollution source of
I was detected were near big cities with large populations. On the basis of the results, we suggest that the likely pollution source of  I, detected in brown seaweeds, is not nuclear power facilities, but nuclear medicine procedures.
I, detected in brown seaweeds, is not nuclear power facilities, but nuclear medicine procedures.
Kawamura, Takumi*; Ito, Kei; Sakai, Takaaki; Nakajima, Masayoshi; Fujiwara, Koji; Ohshima, Hiroyuki
Nihon Genshiryoku Gakkai Wabun Rombunshi, 7(3), p.297 - 307, 2008/09
In a vitrification process of high level radio-active liquid wastes, platinum group particles are mixed into molted glass and convected with the glass flow in vitrification melters. Though behaviors of the particles in the melters should be investigated to establish efficient melter operation, difficulties on observations in the high-radioactive melters have prevented enough investigation. In this paper, we present a numerical analysis method to evaluate the particles behaviors. Since thermal convection of the molten glass, electrical potential and particle distribution are interacting each other, we coupled these physical behaviors to evaluate the melter status with high accuracy. The simulation results for ten batches of operation showed that an effective discharge of the particles can be achieved as well as the mock-up experiments did. These results imply that our numerical method can evaluate transient behaviors of the platinum group particles in vitrification melters.
Umeda, Koji; Oi, Takao; Osawa, Hideaki; Oyama, Takuya; Oda, Chie; Kamei, Gento; Kuji, Masayoshi*; Kurosawa, Hideki; Kobayashi, Yasushi; Sasaki, Yasuo; et al.
JAEA-Review 2007-050, 82 Pages, 2007/12
This report shows the annual report which shows the summarized results and topic outline of each project on geological disposal technology in the fiscal year of 2006.
Tashiro, Koji*; Hanesaka, Makoto*; Ohara, Takashi; Ozeki, Tomoji*; Kitano, Toshiaki*; Nishu, Takashi*; Kurihara, Kazuo; Tamada, Taro; Kuroki, Ryota; Fujiwara, Satoru; et al.
Polymer Journal, 39(12), p.1253 - 1273, 2007/12
Times Cited Count:21 Percentile:53.47(Polymer Science)no abstracts in English
Takamura, Shuichi*; Kado, Shinichiro*; Fujii, Takashi*; Fujiyama, Hiroshi*; Takabe, Hideaki*; Adachi, Kazuo*; Morimiya, Osamu*; Fujimori, Naoji*; Watanabe, Takayuki*; Hayashi, Yasuaki*; et al.
Kara Zukai, Purazuma Enerugi No Subete, P. 164, 2007/03
no abstracts in English
Umeda, Koji; Osawa, Hideaki; Nohara, Tsuyoshi; Sasao, Eiji; Fujiwara, Osamu; Asamori, Koichi; Nakatsuka, Noboru
Genshiryoku Bakkuendo Kenkyu, 11(2), p.97 - 112, 2005/03
This paper describes an overview of geotectonic events in Japan from the point of view of the long-term stability of geological environment. Moreover, the current status of research/prediction technologies developed by JNC is summarized.
Matsumoto, Fumiko*; Makino, Koji*; Maeda, Kayo*; Patzelt, H.*; Maeda, Yuichiro*; Fujiwara, Satoru
Journal of Molecular Biology, 342(4), p.1209 - 1221, 2004/09
Times Cited Count:19 Percentile:29.09(Biochemistry & Molecular Biology)Regulation of skeletal and cardiac muscle contraction is associated with the thin filament-based proteins, troponin C (TnC), TnI, TnT, tropomyosin, and actin. Knowledge of  structures of these proteins is indispensable for elucidating the molecular mechanism of this Ca
structures of these proteins is indispensable for elucidating the molecular mechanism of this Ca -sensitive regulation. Here the structure of TnC within the thin filaments was investigated with neutron scattering, combined with selective deuteration and the contrast matching technique. Deuterated TnC was prepared, reconstituted into the native thin filaments, and neutron scattering patterns of these reconstituted thin filaments containing deuterated TnC were measured under the condition where non-deuterated components were rendered 'invisible' to neutrons. The obtained scattering curves arising only from deuterated TnC were analyzed by model calculations using the Monte Carlo method. The results showed that upon binding of Ca
-sensitive regulation. Here the structure of TnC within the thin filaments was investigated with neutron scattering, combined with selective deuteration and the contrast matching technique. Deuterated TnC was prepared, reconstituted into the native thin filaments, and neutron scattering patterns of these reconstituted thin filaments containing deuterated TnC were measured under the condition where non-deuterated components were rendered 'invisible' to neutrons. The obtained scattering curves arising only from deuterated TnC were analyzed by model calculations using the Monte Carlo method. The results showed that upon binding of Ca ,
,  radius of gyration of TnC changed from 23 AA to 24 AA , and the radial position of TnC within the thin filament changed from 53 AA to 49 AA .
radius of gyration of TnC changed from 23 AA to 24 AA , and the radial position of TnC within the thin filament changed from 53 AA to 49 AA .
Umeda, Koji; Nohara, Tsuyoshi; Fujiwara, Osamu; Asamori, Koichi; Kinoshita, Hirohisa; Nakatsuka, Noboru
JNC TN7400 2004-004, 58 Pages, 2004/08
This report summarizes databases for the long-term stability of geological environment in Japan.
Tashiro, Koji*; Tanaka, Ichiro*; Ohara, Takashi; Niimura, Nobuo*; Fujiwara, Satoru; Kamae, Toshiya*
Macromolecules, 37(11), p.4109 - 4117, 2004/06
Times Cited Count:22 Percentile:56.25(Polymer Science)Two-dimensional wide-angle neutron diffraction patterns have been measured successfully for uniaxially oriented fully deuterated and fully hydrogeneous polyethylene samples by using an imaging plate system (BIX-3) installed at the Japan Atomic Energy Research Institute. The detailed data analysis allowed us to extract the positions of hydrogen atoms in the crystal lattice accurately. The results were compared quantitatively with those obtained from the X-ray and electron diffraction experiments as well as the computer simulation result.
 (
(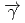 ,
,
 )
)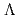 and
and  (
(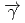 ,
,
 )
)
 reactions for
reactions for 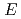
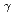 =1.5-2.4 GeV
=1.5-2.4 GeVZegers, R. G. T.*; Sumihama, Mizuki*; Ahn, D. S.*; Ahn, J. K.*; Akimune, Hidetoshi*; Asano, Yoshihiro; Chang, W. C.*; Dat , S.*; Ejiri, Hiroyasu*; Fujimura, Hisako*; et al.
, S.*; Ejiri, Hiroyasu*; Fujimura, Hisako*; et al.
Physical Review Letters, 91(9), p.092001_1 - 092001_4, 2003/08
Times Cited Count:128 Percentile:94.59(Physics, Multidisciplinary)no abstracts in English
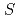 = +1 Baryon resonance in photoproduction from the neutron
= +1 Baryon resonance in photoproduction from the neutronNakano, Takashi*; Ahn, D. S.*; Ahn, J. K.*; Akimune, Hidetoshi*; Asano, Yoshihiro; Chang, W. C.*; Date, S.*; Ejiri, Hiroyasu*; Fujimura, Hisako*; Fujiwara, Mamoru; et al.
Physical Review Letters, 91(1), p.012002_1 - 012002_4, 2003/07
Times Cited Count:1023 Percentile:99.84(Physics, Multidisciplinary)no abstracts in English
Nakatsuka, Noboru; Nohara, Tsuyoshi; Umeda, Koji; Fujiwara, Osamu
JNC TN7440 2003-002, 19 Pages, 2003/04
None