Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yamamoto, Kazami; Kinsho, Michikazu; Hayashi, Naoki; Saha, P. K.; Tamura, Fumihiko; Yamamoto, Masanobu; Tani, Norio; Takayanagi, Tomohiro; Kamiya, Junichiro; Shobuda, Yoshihiro; et al.
Journal of Nuclear Science and Technology, 59(9), p.1174 - 1205, 2022/09
Times Cited Count:7 Percentile:74.40(Nuclear Science & Technology)In the Japan Proton Accelerator Research Complex, the purpose of the 3 GeV rapid cycling synchrotron (RCS) is to accelerate a 1 MW, high-intensity proton beam. To achieve beam operation at a repetition rate of 25 Hz at high intensities, the RCS was elaborately designed. After starting the RCS operation, we carefully verified the validity of its design and made certain improvements to establish a reliable operation at higher power as possible. Consequently, we demonstrated beam operation at a high power, namely, 1 MW. We then summarized the design, actual performance, and improvements of the RCS to achieve a 1 MW beam.
Kobayashi, Daisuke; Yamamoto, Masahiko; Nishida, Naoki; Miyoshi, Ryuta; Nemoto, Ryo*; Hayashi, Hiroyuki*; Kato, Keisuke; Nishino, Saki; Kuno, Takehiko; Kitao, Takahiko; et al.
Nihon Hozen Gakkai Dai-18-Kai Gakujutsu Koenkai Yoshishu, p.237 - 240, 2022/07
All gloves attached to glove-box in Tokai Reprocessing Plant have a fixed expiration date and have to be replaced every 4-year. However, degrees of glove deterioration are different depending on its usage environment (frequency, chemicals, radiation, etc.), because of rubber products. Therefore, physical properties such as tensile strength, elongation, hardness of gloves are measured, and the life-time of gloves are estimated. As a result, gloves without any defects in its appearance have enough physical property for acceptance criteria of new glove. The extrapolated physical property of glove is sufficiently larger than the reported values of damaged glove. No deterioration in physical properties of gloves, that are periodically replaced without any defects in its appearance, is observed and the usable life-time of the glove is estimated to be 8 years.
Kitazato, Kohei*; Milliken, R. E.*; Iwata, Takahiro*; Abe, Masanao*; Otake, Makiko*; Matsuura, Shuji*; Takagi, Yasuhiko*; Nakamura, Tomoki*; Hiroi, Takahiro*; Matsuoka, Moe*; et al.
Nature Astronomy (Internet), 5(3), p.246 - 250, 2021/03
Times Cited Count:58 Percentile:96.23(Astronomy & Astrophysics)Here we report observations of Ryugu's subsurface material by the Near-Infrared Spectrometer (NIRS3) on the Hayabusa2 spacecraft. Reflectance spectra of excavated material exhibit a hydroxyl (OH) absorption feature that is slightly stronger and peak-shifted compared with that observed for the surface, indicating that space weathering and/or radiative heating have caused subtle spectral changes in the uppermost surface. However, the strength and shape of the OH feature still suggests that the subsurface material experienced heating above 300  C, similar to the surface. In contrast, thermophysical modeling indicates that radiative heating does not increase the temperature above 200
C, similar to the surface. In contrast, thermophysical modeling indicates that radiative heating does not increase the temperature above 200  C at the estimated excavation depth of 1 m, even if the semimajor axis is reduced to 0.344 au. This supports the hypothesis that primary thermal alteration occurred due to radiogenic and/or impact heating on Ryugu's parent body.
C at the estimated excavation depth of 1 m, even if the semimajor axis is reduced to 0.344 au. This supports the hypothesis that primary thermal alteration occurred due to radiogenic and/or impact heating on Ryugu's parent body.
Osamura, Kozo*; Machiya, Shutaro*; Kawasaki, Takuro; Harjo, S.; Kato, Takeshi*; Kobayashi, Shinichi*; Osabe, Goro*
Materials Research Express (Internet), 6(2), p.026001_1 - 026001_13, 2019/02
Times Cited Count:7 Percentile:25.67(Materials Science, Multidisciplinary)Sato, Junya; Kikuchi, Hiroshi*; Kato, Jun; Sakakibara, Tetsuro; Matsushima, Ryotatsu; Sato, Fuminori; Kojima, Junji; Nakazawa, Osamu
QST-M-8; QST Takasaki Annual Report 2016, P. 62, 2018/03
no abstracts in English
Okabe, Kota; Yamamoto, Kazami; Kamiya, Junichiro; Takayanagi, Tomohiro; Yamamoto, Masanobu; Yoshimoto, Masahiro; Takeda, Osamu*; Horino, Koki*; Ueno, Tomoaki*; Yanagibashi, Toru*; et al.
Proceedings of 14th Annual Meeting of Particle Accelerator Society of Japan (Internet), p.853 - 857, 2017/12
The most important issue is to reduce the uncontrolled beam loss in the high intensity hadron accelerator such as J-PARC proton accelerators. The J-PARC 3 GeV Synchrotron (RCS) has a collimator system which narrows a high intensity beam in the RCS. After startup of RCS in 2007, the collimator system of the RCS worked well. However, in April 2016, vacuum leakage at the collimator system occurred during the maintenance operation. To investigate a cause of the failure, we took apart iron shields of the collimator reducing exposed dose of operators. As a result of inspection, we succeeded to identify the cause of the vacuum leakage failure. In this presentation, we report the failure investigation of the beam collimator system in the RCS.
Kato, Jun; Nakagawa, Akinori; Taniguchi, Takumi; Sakakibara, Tetsuro; Nakazawa, Osamu; Meguro, Yoshihiro
JAEA-Review 2017-015, 173 Pages, 2017/07
Various radioactive wastes have been generated at the Fukushima Daiichi Nuclear Power Station (1F). To dispose of the wastes underground, it is necessary to make a suitable waste package by the volume reduction and solidification of the wastes. To plan the future decommissioning of 1F, it is also necessary to estimate feasibility of existing treatment technology for those wastes. Therefore the document survey has been performed about volume reduction and solidification technologies that have domestic or foreign experiences of practical treatment for radioactive wastes to assist selection of suitable treatment of the wastes. This report shows the arranged results. The 1F wastes are classified into two groups, homogeneous particulate and liquid wastes and heterogeneous solid wastes. The needful items for the feasibility study such as a technology name, a fundamental principle, treatment efficiency, and characteristic of solidified waste are summarized in each group.
Sato, Junya; Suzuki, Shinji*; Kato, Jun; Sakakibara, Tetsuro; Meguro, Yoshihiro; Nakazawa, Osamu
QST-M-2; QST Takasaki Annual Report 2015, P. 87, 2017/03
no abstracts in English
Sato, Junya; Suzuki, Shinji*; Kato, Jun; Sakakibara, Tetsuro; Meguro, Yoshihiro; Nakazawa, Osamu
QST-M-2; QST Takasaki Annual Report 2015, P. 88, 2017/03
no abstracts in English
Meguro, Yoshihiro; Nakagawa, Akinori; Kato, Jun; Sato, Junya; Nakazawa, Osamu; Ashida, Takashi
Proceedings of International Conference on the Safety of Radioactive Waste Management (Internet), p.139_1 - 139_4, 2016/11
A variety of radioactive wastes have been generated in decommissioning of Fukushima Daiichi Nuclear Power Station. It is necessary to evaluate feasibility of conditioning methods to these wastes, because the majority of such wastes have not been solidified in Japan. The authors investigated an approach for screening of conditioning methods for the Fukushima wastes on the basis of the findings of the existing methods and results of fundamental solidification tests using synthetic Fukushima wastes. Here five solidification methods were selected, and also 13 wastes with different chemical composition are solidified, and characteristics of the solidified form are studied. A screening flow was proposed, and evaluation criteria on each step in the flow was set up. In this presentation a trial result was opened for a waste and improvements of the screening flow found in the trial evaluation was described.
Kada, Wataru*; Miura, Kenta*; Kato, Hijiri*; Saruya, Ryota*; Kubota, Atsushi*; Sato, Takahiro; Koka, Masashi; Ishii, Yasuyuki; Kamiya, Tomihiro; Nishikawa, Hiroyuki*; et al.
Nuclear Instruments and Methods in Physics Research B, 348, p.218 - 222, 2015/04
Times Cited Count:8 Percentile:52.35(Instruments & Instrumentation)Nakayama, Takuya; Suzuki, Shinji; Hanada, Keiji; Tomioka, Osamu; Sato, Junya; Irisawa, Keita; Kato, Jun; Kawato, Yoshimi; Meguro, Yoshihiro
Proceedings of 2nd International Symposium on Cement-based Materials for Nuclear Wastes (NUWCEM 2014) (CD-ROM), 12 Pages, 2014/06
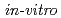 experimental results to model
experimental results to model 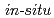 experiments; Bio-denitrification under geological disposal conditions
experiments; Bio-denitrification under geological disposal conditionsMasuda, Kaoru*; Murakami, Hiroshi*; Kurimoto, Noritaka*; Kato, Osamu*; Kato, Ko*; Honda, Akira
SpringerPlus (Internet), 2, p.339_1 - 339_13, 2013/07
Times Cited Count:2 Percentile:5.55(Multidisciplinary Sciences)Some of the low level radioactive wastes from reprocessing of spent nuclear fuels contain nitrates. Nitrates can be present in the form of soluble salts and be reduced by microorganisms. In this study, 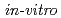 experiments of the nitrate reduction reaction were conducted using model organic materials purported to exist in underground conditions relevant to geological disposal. A reaction model was developed and verified by running simulations against data obtained from
experiments of the nitrate reduction reaction were conducted using model organic materials purported to exist in underground conditions relevant to geological disposal. A reaction model was developed and verified by running simulations against data obtained from 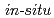 experiments using actual groundwaters and microorganisms. The simulation showed a good correlation with the experimental data and contributes to the understanding of microbially mediated denitrification in geological disposal systems.
experiments using actual groundwaters and microorganisms. The simulation showed a good correlation with the experimental data and contributes to the understanding of microbially mediated denitrification in geological disposal systems.
Otsuka, Ichiro*; Iida, Yoshihisa; Yamaguchi, Tetsuji; Kato, Osamu*; Tateishi, Tsuyoshi*; Tanaka, Tadao
Materials Research Society Symposium Proceedings, Vol.1475, p.507 - 512, 2012/06
We conducted corrosion experiments of carbon wires to identify the cathode reaction with overpack corrosion. Carbon steel wires were immersed in selected aqueous solutions of distilled water, sodium hydrogen carbonate solution and sodium sulfate solution sealed in glass ampoules. The ampoules were kept at 60  C for 150 days. Corrosion products, liquid phase and gas phases were analyzed. The results showed that hydrogen gas generation by reduction of hydrogen ions was dominant cathode reaction. Reduction of sulfate and carbonate anions was not observed. Amorphous ferrous hydroxide and/or iron oxides were identified in all the experiments as the corrosion products. Iron carbonate was also identified in the sodium hydrogen carbonate solution. This result indicates that the hydrogen generation reaction might be the dominant cathode reaction under geological disposal environment and that the Eh of the pore water would reach the equilibrium potential of the hydrogen generation reaction.
C for 150 days. Corrosion products, liquid phase and gas phases were analyzed. The results showed that hydrogen gas generation by reduction of hydrogen ions was dominant cathode reaction. Reduction of sulfate and carbonate anions was not observed. Amorphous ferrous hydroxide and/or iron oxides were identified in all the experiments as the corrosion products. Iron carbonate was also identified in the sodium hydrogen carbonate solution. This result indicates that the hydrogen generation reaction might be the dominant cathode reaction under geological disposal environment and that the Eh of the pore water would reach the equilibrium potential of the hydrogen generation reaction.
Honda, Akira; Masuda, Kaoru*; Tateishi, Tsuyoshi*; Kato, Osamu*; Inoue, Hiroyuki*
Zairyo To Kankyo, 60(12), p.541 - 552, 2011/12
Immersion tests and rest potential measurements under hyper-alkaline and high sodium nitrate concentrations were conducted to elucidate and improve model predictions of chemical interactions between carbon steel and nitrate ion in high concentrations of nitrate salt. The modified model can estimate the tendency of time dependent variation of chemical species and of rest potentials.
Honda, Akira; Masuda, Kaoru*; Imakita, Tsuyoshi*; Kato, Osamu*; Nishimura, Tsutomu*
Zairyo To Kankyo, 58(5), p.182 - 189, 2009/05
Interaction between carbon steel and nitrate was modeled using the mixed potential concept. Carbon steel was selected as an example of metal components in the repository of radioactive waste. The nitrate reduction accompanied by the corrosion of carbon steel was modeled as a reaction series such as nitrate - nitrite - ammonia. The sum of the current of the reaction series of nitrate - nitrite - ammonia and that of water reduction was assumed to balance with the corrosion current of carbon steel. The input parameters for this kinetic model were determined by electrochemical measurement and immersion test. The results of the immersion test can be interpreted by the analyses of the model.
Ishida, Takekazu*; Matsushima, Yoshiaki*; Shimizu, Makoto*; Hayashi, Masahiko*; Ebisawa, Hiromichi*; Sato, Osamu*; Kato, Masaru*; Koyama, Tomio*; Machida, Masahiko; Sato, Kazuo*; et al.
Physica C, 468(7-10), p.576 - 580, 2008/04
Times Cited Count:3 Percentile:16.13(Physics, Applied)The extended Little-Parks effect of superconducting network is known as a periodic 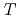
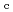 variation as a function of magnetic field. Superconducting Pb honeycomb networks of matching field 0.106 G and triangular microhole lattice of Pb of matching field 0.425 G have been fabricated by the combination of electron-beam lithography and a lift-off process of evaporated Pb films. The application of magnetic field corresponds to the vortex filling into superconducting networks. We measured the magnetization of the networks systematically by using a SQUID magnetometer. We found that flux jump appears rather periodically as a function of magnetic field. Flux jumps may be induced by a periodic decrease of the critical current density of the network. To the authors' knowledge, this is for the first time to observe the regular flux jumps due to the critical current modification coming from the extended Little-Parks effect of the superconducting networks.
variation as a function of magnetic field. Superconducting Pb honeycomb networks of matching field 0.106 G and triangular microhole lattice of Pb of matching field 0.425 G have been fabricated by the combination of electron-beam lithography and a lift-off process of evaporated Pb films. The application of magnetic field corresponds to the vortex filling into superconducting networks. We measured the magnetization of the networks systematically by using a SQUID magnetometer. We found that flux jump appears rather periodically as a function of magnetic field. Flux jumps may be induced by a periodic decrease of the critical current density of the network. To the authors' knowledge, this is for the first time to observe the regular flux jumps due to the critical current modification coming from the extended Little-Parks effect of the superconducting networks.
Ioka, Seiichiro; Iwatsuki, Teruki; Kato, Osamu*; Imakita, Tsuyoshi*
Bunseki Kagaku, 55(10), p.793 - 797, 2006/10
Times Cited Count:2 Percentile:7.94(Chemistry, Analytical)Performance test for redox potential measurement of solution was conducted using continuous polished and untreated Platinum working electrodes in under inert condition. The redox potential measured by polished electrode immediately shows steady value, and approximately coincides with the equilibrium value of redox reaction between HS and S
and S
 . Then the redox potentials would be controlled by the equilibrium of the redox reaction. On the other hand, redox potential measured by untreated electrode was +0.2V higher than that measured by continuous polished electrode. The redox potential measured implies that the SO
. Then the redox potentials would be controlled by the equilibrium of the redox reaction. On the other hand, redox potential measured by untreated electrode was +0.2V higher than that measured by continuous polished electrode. The redox potential measured implies that the SO
 is dominant species in solution. Disagreement of redox potentials measured by polished and untreated electrodes may be caused by deactivation of platinum electrode of adsorption of sulfuric acid anions.
is dominant species in solution. Disagreement of redox potentials measured by polished and untreated electrodes may be caused by deactivation of platinum electrode of adsorption of sulfuric acid anions.
Honda, Akira; Kato, Takashi; Tateishi, Tsuyoshi*; Imakita, Tsuyoshi*; Masuda, Kaoru*; Kato, Osamu*; Nishimura, Tsutomu*
Zairyo To Kankyo, 55(10), p.458 - 465, 2006/10
Migration of radioactive material can be affected by the redox condition and the concentration of ligands in the repository of radioactive waste. It is possible that radioactive waste contains nitrate which can affect the migration behavior of radioactive nuclides by both changing the redox condition of the environment and acting as a ligand. On the other hand, several researchers observed the reduction of nitrate ions in ammonia due to the iron. Ammonia has a potential to ligand for radioactive nuclides. Nitrate can also affect the rate of hydrogen gas evolution accompanied by metal corrosion through changing the rest potential of metal by its oxidizing nature. Carbon steel was, therefore, immersed in an aqueous solution of sodium nitrate in a closed system for observing both the chemical interaction between metal and nitrate, and the effect of nitrate on the hydrogen gas evolution rate. The experimental pH range of the solution was 10.0-13.5 which corresponds to the pH range of pore fluid of cementitious material. The cathodic current density shows a Tafel equation type potential dependency in the aqueous solution containing nitrate or nitrite. In spite of the acceleration of cathodic reaction due to the existence of nitrate, the corrosion rates of carbon steel were not accelerated in the nitrate solutions. This fact suggests that the system is controlled by the anodic reaction. The nitrate reduction accompanied by the corrosion of carbon steel is considered to be a series reaction such as nitrate nitrite ammonia. The nitrate reduction reaction compete with the water reduction reaction within the anodic controlled condition, therefore nitrate strongly reduced the hydrogen evolution rate. The generation rates of ammonia were independent of the concentration of nitrate.
Sugai, Hiroyuki; Matsunami, Noriaki*; Fukuoka, Osamu*; Sataka, Masao; Kato, Teruo; Okayasu, Satoru; Shimura, Tetsuo*; Tazawa, Masato*
Nuclear Instruments and Methods in Physics Research B, 250(1-2), p.291 - 294, 2006/09
Times Cited Count:15 Percentile:69.20(Instruments & Instrumentation)We have investigated the effects on electrical properties of Al-doped ZnO (AZO) semiconductor films induced by high-energy heavy ion. The AZO films with c-axis on SiO glass substrate were prepared by a RF-sputter-deposition method at 400
glass substrate were prepared by a RF-sputter-deposition method at 400  C. Rutherford backscattering spectroscopy shows that the Al/Zn composition and the film thickness are 4 % and 0.3
C. Rutherford backscattering spectroscopy shows that the Al/Zn composition and the film thickness are 4 % and 0.3  m. We find that the conductivity monotonically increases from 1.5
m. We find that the conductivity monotonically increases from 1.5 10
10 to 8
to 8 10
10 S/cm with increasing the fluence up to 4
S/cm with increasing the fluence up to 4 10
10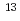 /cm
/cm , as already been observed for 100 keV Ne irradiation. The fluence of 100 keV Ne at which the conductivity takes its maximum is 3
, as already been observed for 100 keV Ne irradiation. The fluence of 100 keV Ne at which the conductivity takes its maximum is 3 10
10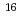 /cm
/cm (7 dpa). The dpa of 100 MeV Xe at 4
(7 dpa). The dpa of 100 MeV Xe at 4 10
10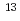 /cm
/cm is estimated as 0.008. Hence, the conductivity increase by 100 MeV Xe ion is ascribed to the electronic excitation effects.
is estimated as 0.008. Hence, the conductivity increase by 100 MeV Xe ion is ascribed to the electronic excitation effects.