Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yuguchi, Takashi*; Kato, Takenori*; Ogita, Yasuhiro; Watanabe, Minori*; Yamazaki, Hayato*; Kato, Asuka*; Ito, Daichi*; Yokoyama, Tatsunori; Sakata, Shuhei*; Ono, Takeshi*
Journal of Asian Earth Sciences, 265, p.106091_1 - 106091_13, 2024/04
Times Cited Count:2 Percentile:36.99(Geosciences, Multidisciplinary)Kijima, Jun; Koyama, Hayato; Owada, Mitsuhiro; Hagiwara, Masayoshi; Aoyagi, Yoshitaka
JAEA-Technology 2022-012, 14 Pages, 2022/07
Steam reforming system has been developed for the treatment of organic wastes which are not suitable materials (halogenated oil) for the incineration due to generation of corrosive compounds and plugging materials. The refractory material is cast inside the main reactor, which is a part of the steam reforming system. Since the surface of this refractory material has deteriorated over time, the main reactor was replaced. If the refractory material surface of the used main reactor can be repaired, the used main reactor can be reused as a spare. The refractory material surface was repaired using two types of repair materials ("S" and "P"). Combustion tests were conducted on samples simulating organic wastes to evaluate each repair material. As a result of the combustion test, it was concluded that the repair of the main reactor was possible to use the repair material "P" because no cracks or flakes were observed.
Yuguchi, Takashi*; Yamazaki, Hayato*; Ishibashi, Kozue*; Sakata, Shuhei*; Yokoyama, Tatsunori; Suzuki, Satoshi*; Ogita, Yasuhiro; Sando, Kazusa*; Imura, Takumi*; Ono, Takeshi*
Journal of Asian Earth Sciences, 226, p.105075_1 - 105075_9, 2022/04
Times Cited Count:8 Percentile:57.12(Geosciences, Multidisciplinary)Simultaneous determination of the U-Pb age of zircon and concentration of titanium in a single analysis spot, using inductively coupled plasma mass spectrometry with laser ablation sample introduction, produces paired age and temperature data of zircon crystallisation, potentially revealing time-temperature (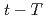 ) histories for evolved magma. The Kurobegawa granite, central Japan, contains abundant mafic magmatic enclaves (MMEs). We applied this method to evaluate MMEs and their host (enclosing) granites. Cooling behaviour common to both MMEs and host rocks was found between 1.5 and 0.5 Ma. Rapid cooling from the zircon crystallisation temperature to the closure temperature of biotite K-Ar system was within
) histories for evolved magma. The Kurobegawa granite, central Japan, contains abundant mafic magmatic enclaves (MMEs). We applied this method to evaluate MMEs and their host (enclosing) granites. Cooling behaviour common to both MMEs and host rocks was found between 1.5 and 0.5 Ma. Rapid cooling from the zircon crystallisation temperature to the closure temperature of biotite K-Ar system was within  1 million year. Combining the obtained
1 million year. Combining the obtained 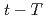 paths of MMEs and host rocks with petrological information can provide insights into magma chamber processes. This suggests that MME flotation, migration, and spread through the magma chamber ceased at 1.5-0.5 Ma, indicating the emplacement age of the Kurobegawa granitic pluton, as no large-scale reheating episodes have occurred since then.
paths of MMEs and host rocks with petrological information can provide insights into magma chamber processes. This suggests that MME flotation, migration, and spread through the magma chamber ceased at 1.5-0.5 Ma, indicating the emplacement age of the Kurobegawa granitic pluton, as no large-scale reheating episodes have occurred since then.
Nishimura, Hayato*; Hojo, Tomohiko*; Ajita, Saya*; Shibayama, Yuki*; Koyama, Motomichi*; Saito, Hiroyuki*; Shiro, Ayumi*; Yasuda, Ryo*; Shobu, Takahisa; Akiyama, Eiji*
Tetsu To Hagane, 107(9), p.760 - 768, 2021/09
Times Cited Count:0 Percentile:0.00(Metallurgy & Metallurgical Engineering)no abstracts in English
Nishimura, Hayato*; Hojo, Tomohiko*; Ajita, Saya*; Shibayama, Yuki*; Koyama, Motomichi*; Saito, Hiroyuki*; Shiro, Ayumi*; Yasuda, Ryo*; Shobu, Takahisa; Akiyama, Eiji*
ISIJ International, 61(4), p.1170 - 1178, 2021/04
Times Cited Count:8 Percentile:37.21(Metallurgy & Metallurgical Engineering)Kasamatsu, Yoshitaka*; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Li, Z.; Ishii, Yasuo; Tome, Hayato*; Sato, Tetsuya; Kikuchi, Takahiro; Nishinaka, Ichiro; et al.
Chemistry Letters, 38(11), p.1084 - 1085, 2009/10
Times Cited Count:17 Percentile:50.80(Chemistry, Multidisciplinary)We report on the characteristic anion-exchange behavior of the superheavy element dubnium (Db) with atomic number Z = 105 in HF/HNO solution at the fluoride ion concentration [F
solution at the fluoride ion concentration [F ] = 0.003 M. The result clearly demonstrates that the fluoro complex formation of Db is significantly different from that of the group-5 homologue Ta in the 6th period of the periodic table while the behavior of Db is similar to that of the lighter homologue Nb in the 5th period.
] = 0.003 M. The result clearly demonstrates that the fluoro complex formation of Db is significantly different from that of the group-5 homologue Ta in the 6th period of the periodic table while the behavior of Db is similar to that of the lighter homologue Nb in the 5th period.
Sone, Tomoyuki; Nakagawa, Akinori; Koyama, Hayato; Gunji, Kiyoshi; Nonaka, Kazuharu; Sasaki, Toshiki; Tashiro, Kiyoshi; Yamashita, Toshiyuki
JAEA-Technology 2009-023, 33 Pages, 2009/06
Steam reforming (SR) method consists of the gasification process in which organics are vaporized and decomposed with superheated steam and the oxidation process in which vaporized organics are decomposed by oxidizing reaction with heated air. 2,500L of waste TBP/n-dodecane contaminated with uranium was treated using the demonstration scale steam reforming system to examine the performance of the system. Results obtained in this study show that the temperature in the SR system was controlled under the self-regulation temperature, the concentration of CO and NOx in the off-gas were controlled less than 100ppm and 250ppm respectively, the distribution ratio of uranium to off-gas treatment system was under 0.12% and the gasification ratio of waste TBP was more than 99%. This long-term waste treatment test has demonstrated that the SR system can safely and effectively reduce the volume of the waste.
Sasaki, Toshiki; Sone, Tomoyuki; Koyama, Hayato; Yamaguchi, Hiromi
Journal of Nuclear Science and Technology, 46(3), p.232 - 238, 2009/03
Times Cited Count:15 Percentile:67.57(Nuclear Science & Technology)A demonstration scale steam reforming treatment system was tested for the treatment of waste tri-n-butyl phosphate/n-dodecane contaminated uranium and simulated halogenated organic wastes stored in the Japan Atomic Energy Agency. The steam reforming treatment system was designed for the organic waste which produced a lot of acidic compounds by incineration and gave severe damage to incinerators. The system comprises a gasification process for decontamination by pyrolysis and steam reforming in a gasification chamber and a destruction process by oxidation with heated air in a submerged combustion type reactor for corrosion prevention. Wastes were reduced by 98 to 99.4% in weight during the treatment without any trouble by the acidic compounds. Almost all uranium was retained in the gasification chamber and uranium concentration of secondary waste water was very low that can permit the discharge of the neutralized waste water in the river.
Haba, Hiromitsu*; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Ishii, Yasuo; Tome, Hayato; Sato, Tetsuya; Nishinaka, Ichiro; Ichikawa, Takatoshi; Ichikawa, Shinichi; et al.
Radiochimica Acta, 95(1), p.1 - 6, 2007/01
Times Cited Count:16 Percentile:70.88(Chemistry, Inorganic & Nuclear)no abstracts in English
Owada, Mitsuhiro; Koyama, Hayato; Kijima, Jun; Shibata, Atsuhiro
no journal, ,
Japan Atomic Energy Agency (JAEA) has been developing a steam reforming system and equipment for the treatment of difficult-to-treat organic liquid wastes. In the continuous treatment of fluorine oil, soot generated during fluorine oil decomposition clogs the exhaust filter and causes unstable combustion. The influence of soot was evaluated by conducting treatment tests using the mixing ratio of fluorine oil and auxiliary fuel as parameters and checking the effect of decomposition temperature. As a result, the filter was not blocked at fluorine oil concentrations up to 20%, and increasing the decomposition temperature burns off the soot and reduces the blockage of the filter. The effects of equipment corrosion due to long-term continuous processing were also identified as an issue.
Toyoshima, Atsushi; Haba, Hiromitsu*; Tsukada, Kazuaki; Asai, Masato; Akiyama, Kazuhiko*; Ishii, Yasuo; Tome, Hayato*; Nishinaka, Ichiro; Sato, Tetsuya; Ichikawa, Takatoshi; et al.
no journal, ,
no abstracts in English
Nagame, Yuichiro; Toyoshima, Atsushi; Ishii, Yasuo; Tsukada, Kazuaki; Asai, Masato; Tome, Hayato; Kasamatsu, Yoshitaka; Nishinaka, Ichiro; Sato, Tetsuya; Haba, Hiromitsu*; et al.
no journal, ,
We present a study of fluoride complex formation of Rf at Japan Atomic Energy Agency (JAEA). Anion- and cation-exchange behavior of Rf was systematically investigated in HF/HNO mixed solution together with the lighter homologues Zr and Hf using the automated ion-exchange separation apparatus coupled with the detection system for alpha spectroscopy. We unequivocally determined the species of Rf on the binding sites of the resin as the hexafluoro complex [RfF
mixed solution together with the lighter homologues Zr and Hf using the automated ion-exchange separation apparatus coupled with the detection system for alpha spectroscopy. We unequivocally determined the species of Rf on the binding sites of the resin as the hexafluoro complex [RfF ]
] . It is found that there is about two-orders of magnitude difference in the fluoride ion concentration between Rf and the homologues for the formation of the hexafluoro complexes. The result clearly demonstrates that the formation of the fluoride complexes of Rf is much weaker than those of the homologues Zr and Hf.
. It is found that there is about two-orders of magnitude difference in the fluoride ion concentration between Rf and the homologues for the formation of the hexafluoro complexes. The result clearly demonstrates that the formation of the fluoride complexes of Rf is much weaker than those of the homologues Zr and Hf.
 solutions
solutionsKasamatsu, Yoshitaka; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Haba, Hiromitsu*; Ishii, Yasuo; Tome, Hayato; Nishinaka, Ichiro; Akiyama, Kazuhiko*; Kikunaga, Hidetoshi*; et al.
no journal, ,
Dubnium-262 was produced in the nuclear reaction of  Cm(
Cm( F,5n)
F,5n) Db using the JAEA Tandem accelerator. The reaction products were rapidly transported to the chemistry laboratory and the anion-exchange behavior of Db in HF/HNO
Db using the JAEA Tandem accelerator. The reaction products were rapidly transported to the chemistry laboratory and the anion-exchange behavior of Db in HF/HNO media was investigated with an on-line rapid ion-exchange apparatus. Based on the comparison of the behavior of Db with that of its homologues (Nb, Ta, and Pa), the fluoride complex formation of Db was discussed.
media was investigated with an on-line rapid ion-exchange apparatus. Based on the comparison of the behavior of Db with that of its homologues (Nb, Ta, and Pa), the fluoride complex formation of Db was discussed.
Ajita, Saya*; Nishimura, Hayato*; Hojo, Tomohiko*; Koyama, Motomichi*; Fujita, Kenichi*; Kakinuma, Hiroshi*; Akiyama, Eiji*; Shibayama, Yuki
no journal, ,
no abstracts in English
Tsukada, Kazuaki; Toyoshima, Atsushi; Haba, Hiromitsu*; Asai, Masato; Akiyama, Kazuhiko*; Ishii, Yasuo; Tome, Hayato*; Nishinaka, Ichiro; Sato, Tetsuya; Ichikawa, Takatoshi; et al.
no journal, ,
no abstracts in English
 media
mediaKasamatsu, Yoshitaka; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Haba, Hiromitsu*; Ishii, Yasuo; Tome, Hayato; Nishinaka, Ichiro; Akiyama, Kazuhiko*; Kikunaga, Hidetoshi*; et al.
no journal, ,
Dubnium-262 was produced in the  Cm(
Cm( F, 5
F, 5 ) reaction at the JAEA tandem accelerator. Adsorption of Db on the anion-exchange resin was investigated in 0.89 M HF/0.3 M HNO
) reaction at the JAEA tandem accelerator. Adsorption of Db on the anion-exchange resin was investigated in 0.89 M HF/0.3 M HNO solution. The anion-exchange behavior of Nb, Ta, and Pa as homologues of Db was also examined in details in HF/HNO
solution. The anion-exchange behavior of Nb, Ta, and Pa as homologues of Db was also examined in details in HF/HNO solutions. From the comparison of those results, we found that the adsorption of Db on the anion-exchange resin is considerably weaker than that of Ta and is relatively similar to those of Nb and Pa in the studied conditions.
solutions. From the comparison of those results, we found that the adsorption of Db on the anion-exchange resin is considerably weaker than that of Ta and is relatively similar to those of Nb and Pa in the studied conditions.
Koyama, Hayato; Aoyama, Yoshio; Sone, Tomoyuki
no journal, ,
no abstracts in English
Shimizu, Koki; Koyama, Hayato; Aoyama, Yoshio
no journal, ,
no abstracts in English
Koyama, Hayato; Kijima, Jun; Shimojo, Hiroto; Nakagawa, Takuya; Owada, Mitsuhiro; Sone, Tomoyuki
no journal, ,
Spent tri-n-butyl phosphate/n-dodecane contaminated with uranium and spent fluorine oil are stored in the Japan Atomic Energy Agency (JAEA). The purpose of this study is to reduce the volume of these wastes, convert it into a stable chemical form, and reduce the amount of storage using the steam assisted pyrolysis system we are developing. We report on the results of treatment of several organic liquid wastes and explain the corrosion prevention methods of this system.
 OH mixed solution
OH mixed solutionTsukada, Kazuaki; Toyoshima, Atsushi; Haba, Hiromitsu*; Asai, Masato; Akiyama, Kazuhiko*; Ishii, Yasuo; Tome, Hayato; Nishinaka, Ichiro; Sato, Tetsuya; Ichikawa, Shinichi; et al.
no journal, ,
no abstracts in English