Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Numakura, Masahiko*; Sato, Nobuaki*; Bessada, C.*; Okamoto, Yoshihiro; Akatsuka, Hiroshi*; Nezu, Atsushi*; Shimohara, Yasuaki*; Tajima, Keisuke*; Kawano, Hirokazu*; Nakahagi, Takeshi*; et al.
Progress in Nuclear Energy, 53(7), p.994 - 998, 2011/11
Times Cited Count:13 Percentile:67.37(Nuclear Science & Technology)X-ray absorption fine structure (XAFS) measurements on thorium fluoride in molten lithium-calcium fluoride mixtures and molecular dynamics (MD) simulation of zirconium and yttrium fluoride in molten lithium-calcium fluoride mixtures have been carried out. In the molten state, coordination number of thorium and inter ionic distances between thorium and fluorine in the first neighbor are nearly constant in all mixtures. However the fluctuation factors (Debye-Waller factor and C cumulant) increase until
cumulant) increase until 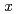 CaF
CaF = 0.17 and decrease by addition of excess CaF
= 0.17 and decrease by addition of excess CaF . It means that the local structure around Th
. It means that the local structure around Th is disordered until
is disordered until 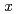 CaF
CaF =0.17 and stabilized over
=0.17 and stabilized over 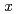 CaF
CaF = 0.17. The variation of fluctuation factors is related to the number density of F
= 0.17. The variation of fluctuation factors is related to the number density of F in ThF
in ThF mixtures and the stability of local structure around Th
mixtures and the stability of local structure around Th increases with decreasing the number density of F
increases with decreasing the number density of F in ThF
in ThF mixtures. This tendency is common to those in the ZrF
mixtures. This tendency is common to those in the ZrF and YF
and YF mixtures.
mixtures.
Numakura, Masahiko; Yaita, Tsuyoshi; Shiwaku, Hideaki; Suzuki, Shinichi; Kobayashi, Toru*; Akutsu, Kazuhiro; Madden, P. A.*; Okamoto, Yoshihiro
JAEA-Research 2009-003, 26 Pages, 2009/04
We investigated the mixing behavior of molten TbCl in LiCl-KCl eutectuic melt by MD simulation. Also, simulation of molten YCl
in LiCl-KCl eutectuic melt by MD simulation. Also, simulation of molten YCl and LaCl
and LaCl systems were performed to elucidate the difference of the structural change by the difference in cation size (Y
systems were performed to elucidate the difference of the structural change by the difference in cation size (Y
 Tb
Tb
 La
La ). The coordination number of the molten pure YCl
). The coordination number of the molten pure YCl , TbCl
, TbCl and LaCl
and LaCl are mainly 6, 7 and 8 respectively. We consider that, in the molten pure state, the coordination number is influenced by the cation size. However, independent of cation size, the molten MCl
are mainly 6, 7 and 8 respectively. We consider that, in the molten pure state, the coordination number is influenced by the cation size. However, independent of cation size, the molten MCl tend to be formed the stable 6-fold structure by the mixing with LiCl-KCl eutectic. And, MD simulations on MCl
tend to be formed the stable 6-fold structure by the mixing with LiCl-KCl eutectic. And, MD simulations on MCl -LiCl and MCl
-LiCl and MCl -KCl systems revealed that the mixing effect was different between LiCl and KCl.
-KCl systems revealed that the mixing effect was different between LiCl and KCl.
Shiwaku, Hideaki; Yaita, Tsuyoshi; Kobayashi, Toru; Numakura, Masahiko; Suzuki, Shinichi; Okamoto, Yoshihiro
Proceedings of 4th Workshop on Speciation, Techniques, and Facilities for Radioactive Materials at Synchrotron Light Sources (Actinide-XAS-2006), p.301 - 306, 2007/05
no abstracts in English
Akutsu, Kazuhiro; Shiwaku, Hideaki; Okamoto, Yoshihiro; Suzuki, Shinichi; Ikeda, Atsushi; Kobayashi, Toru*; Numakura, Masahiko; Mahara, Saori*; Nakano, Yoshiharu*; Fujii, Yuki*; et al.
no journal, ,
no abstracts in English
Numakura, Masahiko; Okamoto, Yoshihiro; Yaita, Tsuyoshi; Shiwaku, Hideaki; Suzuki, Shinichi; Kobayashi, Toru; Akutsu, Kazuhiro; Matsuura, Haruaki*
no journal, ,
The high energy XAFS using K-absorption edge is very effective to obtain information on behavior of small amount of rare earth elements in molten chlorides. In the present study, we performed the XAFS measurement to obtain the local structure change for molten TbCl and the mixture of 40% TbCl
and the mixture of 40% TbCl with LiCl-KCl eutectic, 15% TbCl
with LiCl-KCl eutectic, 15% TbCl with LiCl-KCl eutectic and 1.5% TbCl
with LiCl-KCl eutectic and 1.5% TbCl with LiCl-KCl eutectic. We found that as the concentration of TbCl
with LiCl-KCl eutectic. We found that as the concentration of TbCl is lower, the coordination number decrease a little and become near 6 but the nearest Tb-Cl distance is almost same. In addition, we found that Debye-Waller factor become very small. We also performed molecular dynamics simulation to obtain the information of structure change. This simulation showed the intense structure change occurs at vicinity of 25% TbCl
is lower, the coordination number decrease a little and become near 6 but the nearest Tb-Cl distance is almost same. In addition, we found that Debye-Waller factor become very small. We also performed molecular dynamics simulation to obtain the information of structure change. This simulation showed the intense structure change occurs at vicinity of 25% TbCl with LiCl-KCl eutectic. Its simulation result accorded with this XAFS experiment result.
with LiCl-KCl eutectic. Its simulation result accorded with this XAFS experiment result.
Yaita, Tsuyoshi; Ikeda, Takashi; Kobayashi, Toru*; Suzuki, Shinichi; Shiwaku, Hideaki; Okamoto, Yoshihiro; Ikeda, Atsushi; Akutsu, Kazuhiro; Numakura, Masahiko
no journal, ,
no abstracts in English
Shiwaku, Hideaki; Kobayashi, Toru; Numakura, Masahiko; Yaita, Tsuyoshi; Okamoto, Yoshihiro; Suzuki, Shinichi; Suda, Hiroki*; Fujii, Yuki*
no journal, ,
no abstracts in English
Okamoto, Yoshihiro; Shiwaku, Hideaki; Numakura, Masahiko; Kobayashi, Toru; Akutsu, Kazuhiro; Yaita, Tsuyoshi; Suzuki, Shinichi
no journal, ,
We have studied peculiar behaviors of lanthanide ion in molten chlorides by using high-energy XAFS measurement techniques in the SPring-8. The sample is usually used under static condition in the measurement, since it is sealed off in a quartz cell under reduced pressure. However, there are many reactions in the pyrochemistry of spent nuclear fuels. In the present study, we tried to obtain a XAFS pattern in a few minutes using the Quick scan XAFS (QXAFS) technique developed in the BL11XU, SPring-8. In the QXAFS measurement, a XAFS spectrum of the La K-absorption edge of molten salts were obtained in 5 minutes. It is far shorter than 30-60 minutes in the normal step-scan XAFS measurement. According to the data analysis, it was clarified that the QXAFS data can be used enough to observe molten salt reactions.
Shiwaku, Hideaki; Akutsu, Kazuhiro; Yaita, Tsuyoshi; Okamoto, Yoshihiro; Kobayashi, Toru; Numakura, Masahiko; Nakano, Yoshiharu*; Mahara, Saori*; Fujii, Yuki*
no journal, ,
no abstracts in English
Numakura, Masahiko; Ikeda, Atsushi; Kobayashi, Toru; Shiwaku, Hideaki; Suzuki, Shinichi; Yaita, Tsuyoshi
no journal, ,
no abstracts in English
 -LiF
-LiFNumakura, Masahiko*; Bessada, C.*; Ory, S.*; Rakhamatullin, A.*; Akatsuka, Hiroshi*; Nezu, Atsushi*; Yaita, Tsuyoshi; Okamoto, Yoshihiro; Shiwaku, Hideaki; Matsuura, Haruaki*
no journal, ,
To investigate the local structure of molten TbF - LiF (x[TbF
- LiF (x[TbF = 0.2), X-ray absorption fine structure technique (XAFS) was applied using by BL11XU in SPring-8. By several analyses, e.g. X-ray diffraction and differential scanning calorimetry, it had been confirmed that TbF
= 0.2), X-ray absorption fine structure technique (XAFS) was applied using by BL11XU in SPring-8. By several analyses, e.g. X-ray diffraction and differential scanning calorimetry, it had been confirmed that TbF and boron nitride reacted at less than 1073 K even under helium gas flow environment, thus, we focused on the spectrum obtained below 1073 K. According to the curve fitting, the interionic distance obtained from the data of heating between 933 K and 1013 K was shorter than solid state. It would be surely molten state of TbF
and boron nitride reacted at less than 1073 K even under helium gas flow environment, thus, we focused on the spectrum obtained below 1073 K. According to the curve fitting, the interionic distance obtained from the data of heating between 933 K and 1013 K was shorter than solid state. It would be surely molten state of TbF - LiF (x[TbF
- LiF (x[TbF ] = 0.2), since the value of distance obtained, i.e. 2.26
] = 0.2), since the value of distance obtained, i.e. 2.26 corresponds to the sum of ionic radii of Tb
corresponds to the sum of ionic radii of Tb and F
and F .
.
Shiwaku, Hideaki; Yaita, Tsuyoshi; Kobayashi, Toru; Numakura, Masahiko; Suzuki, Shinichi; Okamoto, Yoshihiro
no journal, ,
no abstracts in English
Numakura, Masahiko; Yaita, Tsuyoshi; Shiwaku, Hideaki; Kobayashi, Toru; Okamoto, Yoshihiro; Suzuki, Shinichi; Ikeda, Atsushi*
no journal, ,
Influences of thee different phenanthorolines (Phens) with which substituent differs, i.e., Phen, Methyl-Phen:MPhen, and Nitro-Phen:NPhen, on their structure and electronic structure formation were studied by EXAFS and XPS methods. The radial structural functions of EXAFS suggested that Lu ion forms 1:3, 1:2, and 1:1 complexes in metal to Phen ratios with Phen, MPhen, and NPhen, respectively. Moreover, the process of which Phen nitrogen with a small backscattering factor exchanges with chloride ion with a large one was clearly observed in the radial structural function of EXAFS. XPS spectra did not show any difference in both the valence level and inner-shell structures among the spectra for three Phens, while clear differences on the spectra of valence level between light lanthanide and heavy lanthanide complexes were observed.