Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tamura, Koji
Japanese Journal of Applied Physics, Part 1, 41(9), p.5845 - 5848, 2002/09
Times Cited Count:3 Percentile:15.34(Physics, Applied)The collimation properties of a Neodymium ion beam extracted from a laser photoionized plasma were measured by scanning a multichannel Faraday cup. The ion beam was collimated at the position of the Faraday cup by the electric field produced with a pair of semispherical electrodes set at the extraction electrode. The central ion beam intensity was increased about 36 times. The results were useful for the applications of laser photoionized ion source.
Horita, M.*; ; Okutsu, Kazuo*; Yamamoto, Takuya*; Amemiya, Kiyoshi*
JNC TJ8400 2000-022, 303 Pages, 2000/02
This report gives supplementary information and discussions on issues of the high-level waste geological disposal study. The following subjects are discussed ; (1)Evaluation of the effects of coefficient of lateral pressure to the specifications of disposal facilities (2)Functional development of remote operational machinery (3)Arrangement of basic data on cost estimation for disposal (4)Understanding of engineering countermeasures to potential phenomena deep under the ground (5)Selection of construction technologies (6)Establishment of the disposal concept under the coasts. For the coefficient of lateral pressure equal to 2, the cross section of disposal drift, the disposal drift spacing, the waste package pitch in the disposal drift and the specification of supporting system are designed. They are compared with those for the case of coefficient of lateral pressure equal to 1. In the case of coefficient of lateral pressure equal to 2, total length of drifts is 1.5 times, and total excavation volume is 1.8 times larger than later case. For the sealing, transportation and emplacement equipment for waste, technology of the fundamental function, remote operation, accidental events and countermeasures are discussed. The plan for developments on those items is proposed. The item of the cost for the construction, operation, and backfilling are discussed. The surface facilities, and, worker arrangement plan are proposed. For the potential phenomena encountered deep under the ground, the countermeasures are investigated form the construction experience, and the future research subjects are discussed for the underground research laboratory. For the construction technologies, the experience of construction management for the tunnel is investigated, and, the research subjects are proposed. For the disposal concept under coasts, rock condition, design condition, construction management, and quality control are compared with the disposal concept under the ground. The ...
Ashida, Takashi; ; Sato, Haruo; ; Kitamura, Akira; Kawamura, Kazuhiro
JNC TN8400 99-083, 63 Pages, 1999/11
Studies on the chemical and migration behaviour of radionuclides were carried out in the Quantitative Assessment Radionuclide Migration Experimental Facility (QUALITY)for assuring the relaiability and for improving the propriety of data concerning nuclide migration used in the Second Progress Report for the geoloical disposal of high-level radioactive waste. Five studies for solubility, sorption and diffusion concerning nuclide migration were carried out. The overview of each study and the result is as follows: (1)Study on Effect of Carbonate on Np Solubility. Solubilities of Np(IV) were measured as functions of pH and carbonate concentration under reducing conditions. The obtained data could be well described by considering two hydroxo-carbonate complexes, and those stability constants were estimated and compared with the literature data. Consequently, the data obtained in this study were similar to the literature data. (2)Study on Effect of Carbonate on Np Sorption on Bentonite. Distribution coefficients (Kd) of Np(IV) on smectite were measured as a function of carbonate concentration. The obtained Kd values were approximately constant over the carbonate concentration (total carbon concentration 0.04-0.15M). The results of desorption tests by 1M KCl and HCl at the end of sorption experiments showed two different desorption behaviour; Np(IV) was well removed by HCl for the experiments in low carbonate concentration and by KCl for those in high carbonate concentration. (3)Distribution Coefficient Measurements for Cs, Pb and Cm on Rocks. Distribution Coefficients for Cs, Pb and Cm on Japanese major rocks (basalt, mudstone, sandstone, granodiorite and tuff) were measured as a function of ionic strength. The obtained Kd values were either the same orders or higher compared with data used to both fresh and saline groundwater systems in the Second Progress Report. This indicates that the Kd data used in the Second Progress Report are either proper or conservative. ...
; ; Sato, Haruo; Shibata, Masahiro
JNC TN8400 99-088, 58 Pages, 1999/06
Sorption and diffusion behavior of palladium, which has been identified as one of the hazardous radionuclides in performance assessment of HLW disposal, in bentonite, granodiorite and tuff was studied in order to make reliable data set for the performance assessment. Sorption experiments of Pd on bentonite, granodiorite and tuff were conducted as functions of pH, ionic strength and liquid to solid ratio by batch method under aerobic conditions at room temperature. The distribution coefficients (K ) of Pd on these solids were almost in the range of 10
) of Pd on these solids were almost in the range of 10 to 10
to 10 m
m /kg and were in the order of bentonite
/kg and were in the order of bentonite  granodiorite
granodiorite 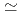 tuff. The sorption trends with change in PH, ionic strength and liquid to solid ratio are very similar between three solids. The K
tuff. The sorption trends with change in PH, ionic strength and liquid to solid ratio are very similar between three solids. The K values were the highest at pH5 and decreased with increasing pH between 5 and 11. The effect of ionic strength on K
values were the highest at pH5 and decreased with increasing pH between 5 and 11. The effect of ionic strength on K was not found in a range of 10
was not found in a range of 10 to 10
to 10 , but K
, but K values increased with increasing liquid to solid ratio. The width of variation in K
values increased with increasing liquid to solid ratio. The width of variation in K was one order of magnitude in a liquid to solid ratio of 0.1 to 1 m
was one order of magnitude in a liquid to solid ratio of 0.1 to 1 m /kg. Sorption behavior of Pd is different from that of divalent metal ions such as Ni and Co etc. and chemical analogy may be inappropriate. The dominant aqueous species of Pd in the expermental conditions studied is estimated to be neutral species, Pd(OH)
/kg. Sorption behavior of Pd is different from that of divalent metal ions such as Ni and Co etc. and chemical analogy may be inappropriate. The dominant aqueous species of Pd in the expermental conditions studied is estimated to be neutral species, Pd(OH) (aq) by the thermodynamic calculations. The K
(aq) by the thermodynamic calculations. The K values of Pd on three solids were relatively high and uncharged complexes may be more strongly sorbed. The pH dependency of K
values of Pd on three solids were relatively high and uncharged complexes may be more strongly sorbed. The pH dependency of K values suggests that Pd sorption is most likely to be occurring onto positively charged S-OH
values suggests that Pd sorption is most likely to be occurring onto positively charged S-OH type site which are progressively removed (to from SOH and SO
type site which are progressively removed (to from SOH and SO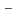 sites) at higher pH values. Diffusion behavior of Pd in bentonite was also studied by in-diffusion method as a function of dry density. The D
sites) at higher pH values. Diffusion behavior of Pd in bentonite was also studied by in-diffusion method as a function of dry density. The D values obtained based on the instantaneous planar source model were in the orders of ...
values obtained based on the instantaneous planar source model were in the orders of ...
PATRICIA F SALTE*; Sasamoto, Hiroshi; Apted, M. J.*; Yui, Mikazu
JNC TN8400 99-023, 231 Pages, 1999/05
The groundwater chemistry is one of important geological environment for performance assessment of high level radioactive disposal system. This report describes the results of geostatistical analysis of groundwater chemistry in Japan. Over 15,000 separate groundwater analyses have been collected of deep Japanese groundwaters for the purpose of evaluating the range of geochemical conditions for geological radioactive waste repositories in Japan. The significance to issues such as radioelement solubility limits, sorption, corrosion of overpack, behavior of compacted clay buffers, and many other factors involved in safety assessment. It is important therefore, that a small, but representative set of groundwater types be identified so that defensible models and data for generic repository performance assessment can be established. Principal component analysis (PCA) is used to categorize representative deep groundwater types from this extensive data set. PCA is a multi-variate statistical analysis technique, similar to factor analysis or eigenvector analysis, designed to provide the best possible resolution of the variability within multi-variate data sets. PCA allows the graphical inspection of the most important similarities (clustering) and differences among samples, based on simultaneous consideration of all variables in the dataset, in a low dimensionality plot. It also allows the analyst to determine the reasons behind any pattern that is observed. In this study, PCA has been aided by hierarchical cluster analysis (HCA), in which statistical indices of similarity among multiple samples are used to distinguish distinct clusters of samples. HCA allows the natural, a priori, grouping of data into clusters showing similar attributes and is graphically represented in a dendrogram Pirouette is the multivariate statistical software package used to conduct the PCA and HCA for the Japanese groundwater dataset. An audit of the initial 15,000 sample dataset on the ...
Lothenbach, B.*; Ochs, M.*; Wanner, H.*; Yui, Mikazu
JNC TN8400 99-011, 340 Pages, 1999/01
This report provides thermodynamic data for predicting concentrations of palladium Pd, lead Pb, tin Sn, antimony Sb, niobium Nb and bismuth Bi in geologic environments, and contributes to an integration of the JNC chemical thermodynamic database, JNC-TDB (previously PNC-TDB), for the performance analysis of geological isolation system of high-level radioactive wastes. Besides treating hydrolysis in detail, this report focuses on the formation of complexes or compounds with chloride, fluoride, carbonate, nitrate, sulfate and phosphate. Other important inorganic ligands (sulfide for lead and antimony, ammonia for palladium) are also included. In this study, the specific ion interaction theory (SIT) approach is used to extrapolate thermodynamic constants to zero ionic strength at 25 C.
C.
Rai, D.*; Rao, L.*; Weger, H. T.*; GREGORY R.CHOPPI*; Yui, Mikazu
JNC TN8400 99-010, 95 Pages, 1999/01
This report provides thermodynamic data for predicting concentrations of Pu(III), Am(III), and Cm(III) in geologic environments, and contributes to an integration of the JNC chemical thermodynamic database, JNC-TDB (previously PNC-TDB), for the performance analysis of geological isolation system for high-level radioactive wastes. Thermodynamic data for the formation of complexes or compounds with hydroxide, chloride, fluoride, carbonate, nitrate, sulfate and phosphate are discussed in this report. Where data for specific actinide(III) species are lacking, the data were selected based on chemical analogy to other trivalent actinides. In this study, the Pitzer ion-interaction model is mainly used to extrapolate thermodynamic constants to zero ionic strength at 25 C.
C.
Chinju, H.*; Nagasaki, Shinya*; Tanaka, Satoru*; Tanaka, Tadao; Ogawa, Hiromichi
Materials Research Society Symposium Proceedings, Vol.556, p.743 - 750, 1999/00
no abstracts in English
Ikeda, Takao*; Amaya, Takayuki*; Chiba, Tamotsu*
PNC TJ1281 97-004, 42 Pages, 1997/03
It is very important to explain the sorption mechanisms of relevant radionuclides, for the performance assessment of geological disposal and for the technical development of engineering barrier system. In the previous studies 1993-1996, these tests were carried out to understand sorption mechanism of SN; -solubility tests -sorption tests onto bentonite, pure montmorillonite, A-FEO(OH) -extraction tests from these minerals -sorption tests onto tuff as a next step, these tests were carried out ragarding SN in this study; -diffusion tests (in-diffusion) -tests for effect of SN concentration and ionic strength on sorption -sorption tests onto granite -tests for effect of coexistent ions on solubility
Sakamoto, Yoshiaki; Nagao, Seiya; Tanaka, Tadao
JAERI-Research 96-055, 14 Pages, 1996/10
no abstracts in English
Yamaguchi, Tetsuji;
Hoshasei Haikibutsu Kenkyu, 3(1), p.49 - 61, 1996/08
no abstracts in English
; ; Yoshida, Masaru; ; Asano, Masaharu; Kumakura, Minoru*
Makromol. Chem., 193, p.715 - 722, 1992/00
no abstracts in English