Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 and Fe-Zr melt
and Fe-Zr meltNakajima, Kunihisa; Takano, Masahide
Journal of Nuclear Science and Technology, 62(1), p.78 - 85, 2025/01
Times Cited Count:1 Percentile:57.00(Nuclear Science & Technology)At TEPCO's Fukushima Daiichi Nuclear Power Station, it is estimated that considerable amounts of cesium still remain in the reactors from the analysis results using the severe accident analysis codes and the reverse analysis from contaminated water. Since cesium is known to form stable compounds with uranium and zirconium, chemisorption experiments with uranium dioxide pellets and iron-zirconium melts for cesium hydroxide vapor were carried out. As the results, formations of cesium uranate, Cs UO
UO , and cesium zirconate, Cs
, and cesium zirconate, Cs ZrO
ZrO , were confirmed, indicating that cesium was chemisorbed on both of the uranium dioxide pellets and the iron-zirconium melts in an Ar-H
, were confirmed, indicating that cesium was chemisorbed on both of the uranium dioxide pellets and the iron-zirconium melts in an Ar-H -H
-H O flow and an Ar-H
O flow and an Ar-H flow, respectively. Therefore, it was considered that cesium released from fuel might be trapped by chemisorption with fuels and/or iron-zirconium melts during light water reactor severe accidents.
flow, respectively. Therefore, it was considered that cesium released from fuel might be trapped by chemisorption with fuels and/or iron-zirconium melts during light water reactor severe accidents.
 Zr +
Zr +  at 27 MeV/nucleon
at 27 MeV/nucleonHwang, J.*; Chillery, T.*; Dozono, Masanori*; Imai, Nobuaki*; Michimasa, Shinichiro*; Sumikama, Toshiyuki*; Chiga, Nobuyuki*; Ota, Shinsuke*; Nakayama, Shinsuke; 49 of others*
Progress of Theoretical and Experimental Physics (Internet), 2024(9), p.093D03_1 - 093D03_12, 2024/09
Times Cited Count:0 Percentile:0.00(Physics, Multidisciplinary)Nuclear transmutation emerges as a promising approach for reprocessing high-level waste, specifically treating long-lived nuclides like  Zr from spent fuel. It is essential to accumulate reaction data for these nuclei to advance this prominent treatment and to build a comprehensive understanding of reaction mechanisms. In this study, the residual production cross-sections resulting from proton-induced reactions on
Zr from spent fuel. It is essential to accumulate reaction data for these nuclei to advance this prominent treatment and to build a comprehensive understanding of reaction mechanisms. In this study, the residual production cross-sections resulting from proton-induced reactions on  Zr were measured at 27 MeV/nucleon in inverse kinematics. At the RIKEN-RIBF facility the OEDO beamline was used to deduce production cross-sections for isotopes,
Zr were measured at 27 MeV/nucleon in inverse kinematics. At the RIKEN-RIBF facility the OEDO beamline was used to deduce production cross-sections for isotopes, 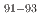 Nb,
Nb, 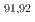 Zr, and
Zr, and 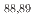 Y. Comparing the results from this study and prior research with calculated excitation functions, a moderate agreement is found with theoretical predictions derived from TALYS and CCONE. Despite the potential limitations of low-energy proton-induced reactions for
Y. Comparing the results from this study and prior research with calculated excitation functions, a moderate agreement is found with theoretical predictions derived from TALYS and CCONE. Despite the potential limitations of low-energy proton-induced reactions for  Zr transmutation, the measured cross-sections offer valuable insights for future considerations in nuclear-waste treatment facilities. This is particularly relevant for facilities exploring innovative methods, such as accelerator-driven systems.
Zr transmutation, the measured cross-sections offer valuable insights for future considerations in nuclear-waste treatment facilities. This is particularly relevant for facilities exploring innovative methods, such as accelerator-driven systems.
 Zr + d reaction cross sections measured at 28 MeV/nucleon
Zr + d reaction cross sections measured at 28 MeV/nucleonChillery, T.*; Hwang, J.*; Dozono, Masanori*; Imai, Nobuaki*; Michimasa, Shinichiro*; Sumikama, Toshiyuki*; Chiga, Nobuyuki*; Ota, Shinsuke*; Nakayama, Shinsuke; 49 of others*
Progress of Theoretical and Experimental Physics (Internet), 2023(12), p.121D01_1 - 121D01_11, 2023/12
Times Cited Count:4 Percentile:40.37(Physics, Multidisciplinary)The deuteron is a loosely bound system which can easily break up into its constituent proton and neutron whilst in the presence of Coulomb and nuclear fields. Previous experimental studies have shown that this breakup process has a significant impact on residual nucleus production from deuteron bombardment in the high energy range of 50 - 210 MeV/nucleon. However, there remains a lack of cross-section data at energies below 50 MeV/nucleon. The current study determined  Zr + d reaction cross sections under inverse kinematics at approximately 28 MeV/nucleon using the BigRIPS separator, OEDO beamline, and SHARAQ spectrometer. Cross sections from this research were compared with previous measurements and theoretical calculations. The experimental results show a large enhancement of the production cross sections of residual nuclei, especially those produced from a small number of particle emissions, compared to the proton-induced reaction data at similar bombarding energy. The DEURACS calculation, which quantitatively takes deuteron-breakup effects into account, reproduces the data well. As a long-lived fission product,
Zr + d reaction cross sections under inverse kinematics at approximately 28 MeV/nucleon using the BigRIPS separator, OEDO beamline, and SHARAQ spectrometer. Cross sections from this research were compared with previous measurements and theoretical calculations. The experimental results show a large enhancement of the production cross sections of residual nuclei, especially those produced from a small number of particle emissions, compared to the proton-induced reaction data at similar bombarding energy. The DEURACS calculation, which quantitatively takes deuteron-breakup effects into account, reproduces the data well. As a long-lived fission product,  Zr remains a challenge for nuclear waste disposal and treatment. This study's low-energy data may assist future consideration of nuclear-waste treatment facilities, where
Zr remains a challenge for nuclear waste disposal and treatment. This study's low-energy data may assist future consideration of nuclear-waste treatment facilities, where  Zr + d may feasibly transmute the waste into short-lived/stable nuclei.
Zr + d may feasibly transmute the waste into short-lived/stable nuclei.
Uehara, Akihiro*; Shuhui, X.*; Sato, Ryotaro*; Matsumura, Daiju; Tsuji, Takuya; Yakumaru, Haruko*; Shiro, Ayumi*; Saito, Hiroyuki*; Tanaka, Izumi*; Ishihara, Hiroshi*; et al.
Advances in X-Ray Chemical Analysis, Japan, 53, p.223 - 229, 2022/03
no abstracts in English
Uehara, Akihiro*; Akiyama, Daisuke*; Ikeda, Atsushi; Numako, Chiya*; Terada, Yasuko*; Nitta, Kiyofumi*; Ina, Toshiaki*; Takeda-Homma, Shino*; Kirishima, Akira*; Sato, Nobuaki*
Journal of Nuclear Materials, 559, p.153422_1 - 153422_11, 2022/02
Times Cited Count:4 Percentile:53.26(Materials Science, Multidisciplinary)Murakami, Tsuyoshi*; Hayashi, Hirokazu
Journal of Nuclear Materials, 558, p.153330_1 - 153330_7, 2022/01
Times Cited Count:2 Percentile:20.79(Materials Science, Multidisciplinary)Excess amounts of dissolution agents, CdCl and ZrCl
and ZrCl , are required to dissolve transuranium (TRU: Pu and minor actinides) nitrides into LiCl-KCl melts at the chemical dissolution step, which is the first step in the reprocessing of used nitride fuels. We propose an electrochemical process where the remaining Zr and Cd are recovered from the melts to be recycled as dissolution agents for the chemical dissolution step, leaving TRU in the melts. Since the initial concentration ratio of CdCl
, are required to dissolve transuranium (TRU: Pu and minor actinides) nitrides into LiCl-KCl melts at the chemical dissolution step, which is the first step in the reprocessing of used nitride fuels. We propose an electrochemical process where the remaining Zr and Cd are recovered from the melts to be recycled as dissolution agents for the chemical dissolution step, leaving TRU in the melts. Since the initial concentration ratio of CdCl /ZrCl
/ZrCl remaining in the melts would depend on the condition of the chemical dissolution step and would vary during the proposed electrochemical recovery process, electrochemical behaviors of Zr and Cd were investigated in LiCl-KCl melts with various concentration ratios of CdCl
remaining in the melts would depend on the condition of the chemical dissolution step and would vary during the proposed electrochemical recovery process, electrochemical behaviors of Zr and Cd were investigated in LiCl-KCl melts with various concentration ratios of CdCl /ZrCl
/ZrCl at 723 K to confirm the basic feasibility of the proposed process. Potentiostatic electrolysis was performed using a liquid Cd cathode at -1.05 V (vs. Ag/AgCl), which was a more positive potential than the redox potentials of TRU on the liquid Cd electrode. The obtained results showed that the current efficiency for recovering Zr and Cd from the melts was as high as 100% regardless of the CdCl
at 723 K to confirm the basic feasibility of the proposed process. Potentiostatic electrolysis was performed using a liquid Cd cathode at -1.05 V (vs. Ag/AgCl), which was a more positive potential than the redox potentials of TRU on the liquid Cd electrode. The obtained results showed that the current efficiency for recovering Zr and Cd from the melts was as high as 100% regardless of the CdCl /ZrCl
/ZrCl concentration ratio in the melts.
concentration ratio in the melts.
Hashikura, Yasuaki*; Ishijima, Yasuhiro; Nakahara, Masaumi; Sano, Yuichi; Ueno, Fumiyoshi; Abe, Hitoshi
Hozengaku, 19(3), p.95 - 102, 2020/10
A plutonium concentrator was selected, and constant load tensile tests with controlled applied potentials and electrochemical tests were conducted in nitric acid and sodium nitrate solutions. From the results, a map which shows the effect of nitric acid concentration to crack initiation potential was drawn. And, it was pointed out that not only the nitric acid but also the nitrate ion coordinated to the nitrate must be considered in evaluating the possibility of stress corrosion cracking.
Morita, Keisuke; Suzuki, Hideya; Matsumura, Tatsuro; Takahashi, Yuya*; Omori, Takashi*; Kaneko, Masaaki*; Asano, Kazuhito*
Proceedings of International Nuclear Fuel Cycle Conference / Light Water Reactor Fuel Performance Conference (Global/Top Fuel 2019) (USB Flash Drive), p.464 - 468, 2019/09
High level liquid waste (HLLW) contains several radionuclides with half-lives longer than 10 year. For reduce environmental burden of waste disposal, minor actinoids and long-lived fission products will to be partitioned and transmuted. JAEA and Toshiba developed process for recovering Se, Zr, Pd and Cs from HLLW. Solvent extraction for Zr with novel extractant,
year. For reduce environmental burden of waste disposal, minor actinoids and long-lived fission products will to be partitioned and transmuted. JAEA and Toshiba developed process for recovering Se, Zr, Pd and Cs from HLLW. Solvent extraction for Zr with novel extractant,  -didodecyl-2-hydroxyacetoamide (HAA) was detailed. The HAA system showed high selectivity for Zr, as indicated by the extraction order of Zr
-didodecyl-2-hydroxyacetoamide (HAA) was detailed. The HAA system showed high selectivity for Zr, as indicated by the extraction order of Zr  Mo
Mo  Pd
Pd  Ag
Ag  Sb
Sb  Sn
Sn  Lns
Lns  Fe. The extracted species was determined as Zr(HAA)
Fe. The extracted species was determined as Zr(HAA) (NO
(NO )
) (HNO
(HNO )
) . A continuous countercurrent extraction with HAA was applied to a simulated, concentrated HLLW after Pd, Se, and Cs removal, where the quantitative extraction of Zr and Mo was effectively demonstrated.
. A continuous countercurrent extraction with HAA was applied to a simulated, concentrated HLLW after Pd, Se, and Cs removal, where the quantitative extraction of Zr and Mo was effectively demonstrated.
 solution; Comparison with tri-
solution; Comparison with tri- -butyl phosphate
-butyl phosphateUeda, Yuki; Kikuchi, Kei; Sugita, Tsuyoshi; Motokawa, Ryuhei
Solvent Extraction and Ion Exchange, 37(5), p.347 - 359, 2019/07
Times Cited Count:10 Percentile:34.94(Chemistry, Multidisciplinary)We have newly-designed fluorous phosphate(TFP) for the effective Zr(IV) ion extractant as an alternative extractant against the conventional organic phosphate, tri- -butyl phosphate(TBP). Zr(IV) ion extraction system using the TBP has many problems such as the formation of the third phase during liquid-liquid extraction. Here, we develop the fluorous extraction system based on the TFP-perfluorohexane for the Zr(IV) ion extraction to improve the Zr(IV) ion extraction system with an effective extractability and without the third phase formation. Our main findings were that the significant high extraction performance of the TFP for Zr(IV) ion as compared with TBP, and the origins of the high extraction performance of the TFP are related to the water and HNO
-butyl phosphate(TBP). Zr(IV) ion extraction system using the TBP has many problems such as the formation of the third phase during liquid-liquid extraction. Here, we develop the fluorous extraction system based on the TFP-perfluorohexane for the Zr(IV) ion extraction to improve the Zr(IV) ion extraction system with an effective extractability and without the third phase formation. Our main findings were that the significant high extraction performance of the TFP for Zr(IV) ion as compared with TBP, and the origins of the high extraction performance of the TFP are related to the water and HNO contents in the fluorous phase and the stability of the complex, Zr(No
contents in the fluorous phase and the stability of the complex, Zr(No )
) (TFP)
(TFP) .
.
Yamauchi, Akihiro*; Amaya, Masaki
Proceedings of Annual Topical Meeting on Reactor Fuel Performance (TopFuel 2018) (Internet), 7 Pages, 2018/10
Differential scanning calorimetry (DSC) measurements on pre-hydrided cold worked, stress relieved and recrystallized Zry-4 cladding were performed in a temperature range between 50 and 600 C in order to elucidate the effect of final heat treatment at fabrication of Zircaloy-4 (Zry-4) cladding on the terminal solid solubility during the dissolution of zirconium hydrides at heating up (TSSD). Obtained DSC curves and Metallography indicate that the initial state of hydrides affects the dissolution behavior of hydride. The Arrhenius plots of the TSSD temperatures and hydrogen contents obtained from this study revealed that cold worked samples exhibited the largest TSSD and followed by stress relieved and recrystallized samples. The results of this study indicated that the difference in microstructure due to final heat treatment at fabrication of Zry-4 cladding affects the dissolution behavior of hydrides.
C in order to elucidate the effect of final heat treatment at fabrication of Zircaloy-4 (Zry-4) cladding on the terminal solid solubility during the dissolution of zirconium hydrides at heating up (TSSD). Obtained DSC curves and Metallography indicate that the initial state of hydrides affects the dissolution behavior of hydride. The Arrhenius plots of the TSSD temperatures and hydrogen contents obtained from this study revealed that cold worked samples exhibited the largest TSSD and followed by stress relieved and recrystallized samples. The results of this study indicated that the difference in microstructure due to final heat treatment at fabrication of Zry-4 cladding affects the dissolution behavior of hydrides.
Arai, Takahiro*; Ito, Daiyu*; Hirasawa, Izumi*; Miyazaki, Yasunori; Takeuchi, Masayuki
Chemical Engineering & Technology, 41(6), p.1199 - 1204, 2018/06
Times Cited Count:4 Percentile:14.75(Engineering, Chemical)In reprocessing process, spent nuclear fuel is dissolved by HNO . Mo and Zr in the acidic solution react to form zirconium molybdate hydrate (ZMH). ZMH adheres to the inner-surface of reprocessing equipment, giving a great influence on stable operation. Conventional anti-encrustation is achieved by cleaning the surface of the reprocessing equipment by high pressure water and dissolving wall deposits by acid or alkaline solution. However, the former increases radioactive waste volume, and the latter contributes to corrosion of the metal surface. In this study, encrustation mechanism of ZMH crystal was investigated based on the deposited mass on the solid surface. Observing the deposition process, it was clarified that ZMH fine crystal growth is accelerated and agglomerated, followed by the rapid deposition. Total deposited mass decreased not only by lowering initial ratio of Mo and Zr concentration under the range of 1.0
. Mo and Zr in the acidic solution react to form zirconium molybdate hydrate (ZMH). ZMH adheres to the inner-surface of reprocessing equipment, giving a great influence on stable operation. Conventional anti-encrustation is achieved by cleaning the surface of the reprocessing equipment by high pressure water and dissolving wall deposits by acid or alkaline solution. However, the former increases radioactive waste volume, and the latter contributes to corrosion of the metal surface. In this study, encrustation mechanism of ZMH crystal was investigated based on the deposited mass on the solid surface. Observing the deposition process, it was clarified that ZMH fine crystal growth is accelerated and agglomerated, followed by the rapid deposition. Total deposited mass decreased not only by lowering initial ratio of Mo and Zr concentration under the range of 1.0  Mo/Zr
Mo/Zr  2.0, but also by lowering HNO
2.0, but also by lowering HNO concentration.
concentration.
Kumagai, Yuta; Takano, Masahide; Watanabe, Masayuki
Journal of Nuclear Materials, 497, p.54 - 59, 2017/12
Times Cited Count:15 Percentile:77.68(Materials Science, Multidisciplinary)We studied oxidative dissolution of uranium and zirconium oxide [(U,Zr)O ] in aqueous H
] in aqueous H O
O solution. The interfacial reaction is essential for anticipating how a (U,Zr)O
solution. The interfacial reaction is essential for anticipating how a (U,Zr)O -based molten fuel may chemically degrade after a severe accident under influence of ionizing radiation. We conducted our experiments with (U,Zr)O
-based molten fuel may chemically degrade after a severe accident under influence of ionizing radiation. We conducted our experiments with (U,Zr)O powder and quantitated the H
powder and quantitated the H O
O reaction via dissolved U and H
reaction via dissolved U and H O
O concentrations. The dissolution yield relative to H
concentrations. The dissolution yield relative to H O
O consumption was far less for (U,Zr)O
consumption was far less for (U,Zr)O compared to that of UO
compared to that of UO . The reaction kinetics indicates that most of the H
. The reaction kinetics indicates that most of the H O
O catalytically decomposed to O
catalytically decomposed to O at the surface of (U,Zr)O
at the surface of (U,Zr)O . We confirmed the H
. We confirmed the H O
O catalytic decomposition via O
catalytic decomposition via O production (quantitative stoichiometric agreement). In addition, post-reaction Raman scattering spectra of the undissolved (U,Zr)O
production (quantitative stoichiometric agreement). In addition, post-reaction Raman scattering spectra of the undissolved (U,Zr)O showed no additional peaks (indicating a lack of secondary phase formation). The (U,Zr)O
showed no additional peaks (indicating a lack of secondary phase formation). The (U,Zr)O matrix is much more stable than UO
matrix is much more stable than UO against H
against H O
O -induced oxidative dissolution.
-induced oxidative dissolution.
Morita, Yasuji; Yamagishi, Isao
JAEA-Research 2017-006, 27 Pages, 2017/06
Separation of Pd by extraction with 5,8-diethyl-7-hydroxy-6-dodecanone oxime (DEHDO) was examined by batch and continuous tests for the purpose of developing Pd separation process. Batch extraction tests using n-dodecane solution of DEHDO revealed that Pd, Zr and Mo were extracted from simulated high-level radioactive liquid wastes (HLLW) and other elements were not, and also showed that the extraction rate was a little slow and a white precipitate appeared in the aqueous phase but its formation could be avoided by raising temperature. The extracted Pd was found to be back-extracted with sodium nitrite. In the continuous extraction tests with simulated HLLW without Zr and Mo, about 98% of Pd were extracted with DEHDO-n-dodecane and 95% of the extracted Pd were back-extracted with sodium nitrite and nitric acid. Continuous extraction test with simulated HLLW with Zr and Mo showed the possibility of the simultaneous separation of Pd and Mo by DEHDO extraction.
Takeuchi, Masayuki; Aihara, Haruka; Nakahara, Masaumi; Tanaka, Kotaro*
Procedia Chemistry, 21, p.182 - 189, 2016/12
Times Cited Count:2 Percentile:79.62(Chemistry, Inorganic & Nuclear)A simulation technology with electrolyte thermodynamic model has been developed to evaluate the precipitation behavior in reprocessing solution based on nitric acid solution. The simulation results were compared with the experiment data from non-radioactive simulated HLLW with ten elements and Pu-Zr-Mo solution, and the reliability of the thermodynamic model was verified. Most of the precipitation species was zirconium molybdate hydrate from the both data. It is demonstrated that the chemical species and amount of the precipitation calculated by thermodynamic model reflected well that of experiments. This study has shown the thermodynamic simulation model is one of the useful tools to estimate the behavior of precipitation from the reprocessing solution.
Konda, Miki; Asai, Shiho; Hanzawa, Yukiko; Magara, Masaaki
JAEA-Technology 2015-054, 22 Pages, 2016/03
Isotope dilution mass spectrometry (IDMS) with ICP-MS is reliable method for determination of Zr-93, which is one of the long-lived fission products found in spent nuclear fuel and high-level radioactive wastes. In order to use an isotope standard solution of zirconium as the spike for IDMS, dissolving a commercially available solid isotope standard is indispensable. Prior to the dissolution of the Zr-91 isotope standard, solubility of metal zirconium in a mixture of HNO and HF was evaluated using zirconium metal chips. Then, 2 mg of the Zr-91 isotope standard was dissolved with 0.2 mL of 1 M HNO
and HF was evaluated using zirconium metal chips. Then, 2 mg of the Zr-91 isotope standard was dissolved with 0.2 mL of 1 M HNO -3 v/v% HF mixed solution, followed by adjusting the concentration of Zr-91 to approximately 1,000
-3 v/v% HF mixed solution, followed by adjusting the concentration of Zr-91 to approximately 1,000  g/g. IDMS, in which a natural isotopic abundance standard solution of zirconium was used as the spike, was employed for the determination of the concentration of Zr-91 in the prepared Zr-91 isotope standard solution. The concentration of Zr-91 in the prepared Zr-91 isotope standard solution was (9.6
g/g. IDMS, in which a natural isotopic abundance standard solution of zirconium was used as the spike, was employed for the determination of the concentration of Zr-91 in the prepared Zr-91 isotope standard solution. The concentration of Zr-91 in the prepared Zr-91 isotope standard solution was (9.6 1.0)
1.0)  10
10
 g/g, which is in good agreement with the predicted concentration. This indicates that the Zr-91 metal isotope standard was completely dissolved with sufficient chemical stability. Additionally, no impurities were detected in the prepared Zr-91 isotope standard solution. These positive results denote that the Zr-91 isotope standard solution with the preferable quality for IDMS of Zr-93 can be obtained by the proposed dissolution procedures.
g/g, which is in good agreement with the predicted concentration. This indicates that the Zr-91 metal isotope standard was completely dissolved with sufficient chemical stability. Additionally, no impurities were detected in the prepared Zr-91 isotope standard solution. These positive results denote that the Zr-91 isotope standard solution with the preferable quality for IDMS of Zr-93 can be obtained by the proposed dissolution procedures.
Matsumoto, Yoshinobu*; Do, Thi-Mai-Dung*; Inoue, Masao; Nagaishi, Ryuji; Ogawa, Toru
Journal of Nuclear Science and Technology, 52(10), p.1303 - 1307, 2015/10
Times Cited Count:4 Percentile:30.44(Nuclear Science & Technology)Effects of zirconium oxides and oxidation products of zircaloy-4 on water radiolysis were investigated to predict the hydrogen generation from the water-immersed debris after a severe accident of a nuclear power plant. Observed yields of hydrogen in water containing the oxides were measured as a function of their weight fractions. Assuming that energies of Co-60  -ray deposited to water and the oxides brought about the water radiolysis to generate hydrogen independently, the radiolysis showed an additional term of hydrogen generation due to the energy deposition to the oxides. This term seemed to be dependent on the specific surface area or particle size of oxides, but not on the crystal structure of oxides in our experimental results. The oxides in distilled water gave the strong enhancement of term. The enhancement tended to saturate with increasing the weight fraction of oxides and was not apparent in the seawater.
-ray deposited to water and the oxides brought about the water radiolysis to generate hydrogen independently, the radiolysis showed an additional term of hydrogen generation due to the energy deposition to the oxides. This term seemed to be dependent on the specific surface area or particle size of oxides, but not on the crystal structure of oxides in our experimental results. The oxides in distilled water gave the strong enhancement of term. The enhancement tended to saturate with increasing the weight fraction of oxides and was not apparent in the seawater.
Machida, Masahiko; Nakamura, Hiroki; Srinivasan, S. G.*; Van Duin, A. C. T.*
Proceedings of 23rd International Conference on Nuclear Engineering (ICONE-23) (DVD-ROM), 4 Pages, 2015/05
Zircalloy has been widely employed as an excellent material covering the fuel rod. The mechanical and thermal properties have been explored by various experiments. In terms of its use as the fuel cladding, its response to oxidation reactions is an important topic when it is exposed to high temperature and high pressure steam during severe accidents. Especially, the hydrogen production accompanied by the oxidation is critical because it can lead to the crisis of the hydrogen explosion, as observed in the Fukushima Nuclear Power Plant accidents. Silicon carbide (SiC) has been considered as an alternative cladding material owing to an advantage that hydrogen production is much suppressed in the equivalent condition compared to Zircalloy. Therefore, we simulate the oxidation reaction for both materials, i.e. Zirconium metal and SiC in atomistic level by using the ReaxFF reactive force field method to simulate the chemical reaction molecular dynamics. Through such comparative studies between Zirconium and SiC in the same condition, we clarify how the temperature and the steam pressure accelerates the oxidation reaction and the resultant hydrogen production in both materials at typical severe accident conditions. The advantage using ReaxFF is that it allows us to directly trace the oxygen diffusion inside the Zirconium metal and SiC depending on the temperature and vapor pressure together with the oxidation reaction. We can compare the reaction processes in both materials. Especially, we paid attention to the rate of hydrogen production in both materials.
Kuramoto, Kenichi; Shirasu, Noriko; Yamashita, Toshiyuki
Journal of Nuclear Materials, 319(1-3), p.180 - 187, 2003/06
Times Cited Count:9 Percentile:52.47(Materials Science, Multidisciplinary)no abstracts in English
Kato, Chiaki; Yano, Masaya*; Kiuchi, Kiyoshi; Sugimoto, Katsuhisa*
Corrosion Engineering, 52(1), p.53 - 67, 2003/01
The effects of heat-transfer on the corrosion of zirconium was examined in boiling nitric acid solutions with various concentrations. Corrosion mass losses and electrochemical polarization curves were measured on the heat-transfer and isothermal surfaces in the solutions. It was found that the corrosion rate of zirconium was higher on the heat-transfer surface than that on the isothermal surface. The rate increased with increasing nitric acid concentration and solution temperature. The increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition on the surface and the removal of the decomposition product from solution by boiling bubbles. The redox potential of 12 mol/dm nitric acid on a boiling heat-transfer surface was very close to the breakdown potential of primary passivity of zirconium. This suggests the initiation of SCC on a boiling heat-transfer surface in a nuclear fuel reprocessing.
nitric acid on a boiling heat-transfer surface was very close to the breakdown potential of primary passivity of zirconium. This suggests the initiation of SCC on a boiling heat-transfer surface in a nuclear fuel reprocessing.
Kato, Chiaki; Yano, Masaya*; Kiuchi, Kiyoshi; Sugimoto, Katsuhisa*
Zairyo To Kankyo, 52(1), p.35 - 43, 2003/01
The effects of heat-transfer on the corrosion of zirconium was examined in boiling nitric acid solutions with various concentrations. Corrosion mass losses and electrochemical polarization curves were measured on the heat-transfer and isothermal surfaces in the solutions. It was found that the corrosion rate of zirconium was higher on the heat-transfer surface than that on the isothermal surface. The rate increased with increasing nitric acid concentration and solution temperature. The increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition on the surface and the removal of the decomposition product from solution by boiling bubbles. The redox potential of 12 mol/dm3 nitric acid on a boiling heat-transfer surface was very close to the breakdown potential of primary passivity of zirconium. This suggests the initiation of SCC on a boiling heat-transfer surface in a nuclear fuel reprocessing.