Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Sato, Tetsuya
Kagaku To Kogyo, 72(10), P. 867, 2019/10
We conducted measurements of the first ionization potential (IP ) of the heavy actinide elements, lawrencium (Lr,
) of the heavy actinide elements, lawrencium (Lr, 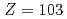 ), nobelium (No,
), nobelium (No, 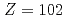 ), mendelevium (Md,
), mendelevium (Md, 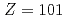 ) and fermium (Fm,
) and fermium (Fm, 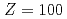 ) by using a novel method based on a surface ionization process. The IP
) by using a novel method based on a surface ionization process. The IP measurements have been performed using the ISOL (Isotope Separator On-Line) system equipped with a surface ion-source with short-lived heavy actinide isotopes,
measurements have been performed using the ISOL (Isotope Separator On-Line) system equipped with a surface ion-source with short-lived heavy actinide isotopes,  Lr (
Lr (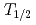 = 27s),
= 27s),  No (
No (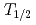 = 24.5s),
= 24.5s),  Md (
Md (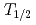 = 4.27 min), and
= 4.27 min), and  Fm (
Fm (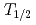 = 2.6 min). Our experimental results clearly showed that the IP
= 2.6 min). Our experimental results clearly showed that the IP of Lr is distinctly low among actinide elements. Moreover, No has the highest IP
of Lr is distinctly low among actinide elements. Moreover, No has the highest IP among them due to its full-filled 5f and 7s orbitals; the IP
among them due to its full-filled 5f and 7s orbitals; the IP value increased with an atomic number up to No and decreased dramatically at Lr, indicating the similar trend with that of heavy lanthanide elements. Therefore, we concluded Lr would be the last member of the actinide series.
value increased with an atomic number up to No and decreased dramatically at Lr, indicating the similar trend with that of heavy lanthanide elements. Therefore, we concluded Lr would be the last member of the actinide series.
Taguchi, Shigeo; Miyauchi, Hironari*; Horigome, Kazushi; Yamamoto, Masahiko; Kuno, Takehiko
Bunseki Kagaku, 67(11), p.681 - 686, 2018/11
In thermal ionization mass spectrometry, de-gassing is one of the important treatments to release impurities of filaments and to minimize the influence of background. In this work, the effect of the surface change in the tungsten filament induced by the conductively heating treatment on uranium isotopic ( U/
U/ U) measurement has been investigated. It was found that the conductively heating treatment of the filament has the effect of smoothing the surface of the filament and also has the effect of improving the deposition of the sample on the filament surface. As a result of either these effects, the precision of uranium isotopic (
U) measurement has been investigated. It was found that the conductively heating treatment of the filament has the effect of smoothing the surface of the filament and also has the effect of improving the deposition of the sample on the filament surface. As a result of either these effects, the precision of uranium isotopic ( U/
U/ U) measurement was improved.
U) measurement was improved.
Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Beerwerth, R.*; Kaneya, Yusuke*; Makii, Hiroyuki; Mitsukai, Akina*; Nagame, Yuichiro; Osa, Akihiko; Toyoshima, Atsushi; et al.
Journal of the American Chemical Society, 140(44), p.14609 - 14613, 2018/11
Times Cited Count:37 Percentile:71.50(Chemistry, Multidisciplinary)The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
Baba, Yuji; Shimoyama, Iwao
Photon Factory Activity Report 2017, 3 Pages, 2018/00
When a solid is heated in vacuum, a part of the surface layer desorbs as ions, which is known as "surface ionization". In this report, we present the results for surface ionization of bulk cesium chloride (CsCl). When the positive potential was applied to the sample, we found that the Cs ions were desorbed at around 410
ions were desorbed at around 410  C, which is lower than the melting point of CsCl (645
C, which is lower than the melting point of CsCl (645  C). The low desorption temperature was explained by the changes in the work function of the CsCl surface. As an application, we also investigated the desorption of Cs
C). The low desorption temperature was explained by the changes in the work function of the CsCl surface. As an application, we also investigated the desorption of Cs ions from Cs-adsorbed soil. When Cs-adsorbed soil was heated at 460
ions from Cs-adsorbed soil. When Cs-adsorbed soil was heated at 460  C for 2 hours, about 13% of Cs was desorbed as Cs
C for 2 hours, about 13% of Cs was desorbed as Cs ions. The results suggest that the surface ionization would possibly be applied to the desorption of Cs from contaminated soil.
ions. The results suggest that the surface ionization would possibly be applied to the desorption of Cs from contaminated soil.
Okubo, Ayako; Shinohara, Nobuo; Magara, Masaaki
Journal of Radioanalytical and Nuclear Chemistry, 314(1), p.231 - 234, 2017/10
Times Cited Count:3 Percentile:23.67(Chemistry, Analytical)The model date of two enriched uranium materials were determined using a new method for nuclear forensics investigation. In this method, the 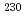 Th/
Th/ U ratio was calculated without spike addition from measured ratios of
U ratio was calculated without spike addition from measured ratios of 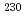 Th/
Th/ Th and
Th and  U/
U/ U, and calculated
U, and calculated  Th/
Th/ U ratio in secular equilibrium. The obtained model date for the low-enriched uranium material was agreed with the known production date within uncertainty. For the highly enriched uranium material, slightly younger model date than the known production date was obtained. The
U ratio in secular equilibrium. The obtained model date for the low-enriched uranium material was agreed with the known production date within uncertainty. For the highly enriched uranium material, slightly younger model date than the known production date was obtained. The  U interference on
U interference on  Th counting in thermal ionization mass spectrometry measurement was suspected as a potential cause.
Th counting in thermal ionization mass spectrometry measurement was suspected as a potential cause.
Sato, Tetsuya
Genshikaku Kenkyu, 61(1), p.96 - 106, 2016/09
We successfully determined the first ionization potential of lawrencium (Lr, Z=103). The result experimentally substantiated for the first time that Lr is the last member of the actinide series. Measured ionization potential suggested that Lr atom would have the electronic configuration which is different from the configuration expected based on the Periodic table. For the measurement, we have developed a novel method applied the surface ionization process. Public responses after the publication are also introduced.
Sato, Tetsuya
Isotope News, (740), p.16 - 19, 2015/12
We successfully determined the first ionization potential of lawrencium (Lr, Z=103). The result experimentally substantiated for the first time that Lr is the last member of the actinide series. Measured ionization potential suggested that Lr atom would have the electronic configuration which is different from the configuration expected based on the Periodic table.
Sato, Tetsuya
Nihon Genshiryoku Gakkai-Shi ATOMO , 57(11), p.741 - 744, 2015/11
, 57(11), p.741 - 744, 2015/11
We have experimentally confirmed that Lr would be the last member of actinides series for the first time by a measurement of the first ionization potential of lawrencium (Lr, element 103). The electronic orbital of Lr atom which is estimated by the result suggests that Lr could have the outermost electronic orbital similar with group-13 elements. This work triggered a discussion concerning positions of Lr and lutetium, lanthanide homologue of Lr.
Sato, Tetsuya
Hosha Kagaku, (32), p.34 - 41, 2015/09
In the surface ionization process, an ionization efficiency depends on the first ionization potential of the atom of the element. The ionization potential can be estimated by using the relationship. This method has been developed in order to determine the first ionization potential of lawrencium (Lr, element 103). The value of the ionization potential of Lr have not been measured experimentally due to its low production rate and short half-life. The surface-ionization method is described in detail in this paper.
Sato, Tetsuya; Nagame, Yuichiro; Tsukada, Kazuaki
Kagaku To Kogyo, 68(9), p.824 - 826, 2015/09
We successfully confirmed that lawrencium (Lr, element 103) is the last member of actinide series by a measurement of its first ionization potential. Obtained experimental result suggested that the outermost electronic orbital of Lr atom would have p-orbital similar to elements of group-13. Our result triggered again the discussion of the position of Lr and lutetium, the lanthanide homologue of Lr, on the Periodic Table.
Sato, Tetsuya
Saiensu Potaru (Internet), 3 Pages, 2015/07
On April 9th, a press release titled "Measurement of the first ionization potential of lawrencium (element 103) - Unravelling Relativistic Effects in the Heaviest Actinide Element -" was issued. This research result published from Nature was not only introduced in its "News & Views" but also appeared on the cover. I made a commentary on the result and introduced its response for public.
Esaka, Fumitaka; Suzuki, Daisuke; Magara, Masaaki
Analytical Chemistry, 87(5), p.3107 - 3113, 2015/03
Times Cited Count:15 Percentile:44.97(Chemistry, Analytical)The analysis of uranium particles in environmental samples taken from nuclear facilities is a useful tool to unveil undeclared nuclear activities related to the production of nuclear weapons. An efficient method to analyze isotope ratios of individual uranium particles is thermal ionization mass spectrometry (TIMS) combined with a fission track technique. A drawback in the fission track-TIMS technique is so-called particle-mixing. Here, some uranium particles are measured as a single particle and an averaged isotope ratio is obtained, which may lead to misunderstanding conclusions for source identification. In the present study, micro-sampling under a scanning electron microscope has been added to the procedure of the fission track-TIMS technique. The analysis of a mixed sample containing uranium particles in SRM 950a and CRM U100 materials indicated that the problem of particle mixing was almost avoidable with the proposed technique.
Fujii, Toshiyuki*; Suzuki, Daisuke; Watanabe, Kazuo; Yamana, Hajimu*
Talanta, 69(1), p.32 - 36, 2006/03
Times Cited Count:12 Percentile:34.52(Chemistry, Analytical)The total evaporation technique of thermal ionization mass spectrometry was applied to the isotopic analysis of chromium. High measurement reproducibility of the chromium isotope ratios was verified (2 S.E.  0.05% (
0.05% (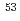 Cr/
Cr/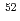 Cr)), while a clear mass fractionation effect was observed by using conventional measurement technique. The chromium isotope ratios analyzed by the total evaporation method were not affected by the sample amount on the rhenium filament (50-500 ng Cr). The isotopic analysis under the coexistence of zinc was also performed, and its effect to the chromium isotope ratios was confirmed.
Cr)), while a clear mass fractionation effect was observed by using conventional measurement technique. The chromium isotope ratios analyzed by the total evaporation method were not affected by the sample amount on the rhenium filament (50-500 ng Cr). The isotopic analysis under the coexistence of zinc was also performed, and its effect to the chromium isotope ratios was confirmed.
Esaka, Fumitaka; Watanabe, Kazuo; Fukuyama, Hiroyasu; Onodera, Takashi; Esaka, Konomi; Inagawa, Jun; Iguchi, Kazunari; Suzuki, Daisuke; Lee, C. G.; Magara, Masaaki; et al.
Dai-25-Kai Kaku Busshitsu Kanri Gakkai Nihon Shibu Nenji Taikai Rombunshu, p.128 - 135, 2004/00
Japan Atomic Energy Research Institute (JAERI) was qualified as a member of the IAEA network analytical laboratories (NWALs) for particle and bulk analyses of safeguards environmental samples in January 2003. The particle analysis gives more detailed information on nuclear facility operation than the bulk analysis because the isotope ratios of nuclear materials in the samples collected inside nuclear facilities (swipe samples) can be determined for individual particles. We applied, as a method for uranium isotope ratio measurement, secondary ion mass spectrometry (SIMS) to particle analysis. Prior to the SIMS analysis, the particles in a swipe sample are recovered onto a carrier by impaction. The carriers with the recovered particles are then screened by total reflection X-ray fluorescence spectrometry. We integrated these techniques into a standard procedure, which is applied to domestic and IAEA swipe samples routinely.
Osa, Akihiko; Koizumi, Mitsuo; Sekine, Toshiaki; Katsuragawa, H.*; Jin, W.*; Wakui, Takashi*
JAERI-Review 99-025, TIARA Annual Report 1998, p.203 - 205, 1999/10
no abstracts in English
Tamura, Koji; ; Adachi, Hajime; Shibata, Takemasa; Oba, Hironori
Japanese Journal of Applied Physics, Part 1, 37(12A), p.6651 - 6654, 1998/12
Times Cited Count:0 Percentile:0.00(Physics, Applied)no abstracts in English
Tamura, Koji; Adachi, Hajime; Shibata, Takemasa
JAERI-Research 98-020, 13 Pages, 1998/03
no abstracts in English
Tamura, Koji; Adachi, Hajime; Shibata, Takemasa
Shinku, 41(3), p.332 - 334, 1998/00
no abstracts in English
; Shibata, Takemasa
JAERI-Tech 97-011, 14 Pages, 1997/03
no abstracts in English
KURRI-KR-3, 0, p.28 - 32, 1996/00
no abstracts in English