Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Terasaka, Yuta; Iimoto, Takeshi*; Saso, Michitaka*; Fujita, Reiko*
Nihon Genshiryoku Gakkai-Shi ATOMO , 66(4), p.203 - 207, 2024/04
, 66(4), p.203 - 207, 2024/04
no abstracts in English
Collaborative Laboratories for Advanced Decommissioning Science; National Institutes of Natural Sciences*
JAEA-Review 2022-059, 34 Pages, 2023/01
The Collaborative Laboratories for Advanced Decommissioning Science (CLADS), Japan Atomic Energy Agency (JAEA), had been conducting the Nuclear Energy Science & Technology and Human Resource Development Project (hereafter referred to "the Project") in FY2021. The Project aims to contribute to solving problems in the nuclear energy field represented by the decommissioning of the Fukushima Daiichi Nuclear Power Station, Tokyo Electric Power Company Holdings, Inc. (TEPCO). For this purpose, intelligence was collected from all over the world, and basic research and human resource development were promoted by closely integrating/collaborating knowledge and experiences in various fields beyond the barrier of conventional organizations and research fields. The sponsor of the Project was moved from the Ministry of Education, Culture, Sports, Science and Technology to JAEA since the newly adopted proposals in FY2018. On this occasion, JAEA constructed a new research system where JAEA-academia collaboration is reinforced and medium-to-long term research/development and human resource development contributing to the decommissioning are stably and consecutively implemented. Among the adopted proposals in FY2021, this report summarizes the research results of the "Development of the continuous monitoring of tritium water by mid-infrared laser spectroscopy" conducted in FY2021. The present study aims to demonstrate the principle of short-time measurement of tritiated water at the "60 Bq/cc level" using a cavity ring-down measurement system with a mid-infrared laser. In order to achieve the above goal, (1) research on the cavity ring-down system and (2) evaluation of hydrogen isotope composition under environmental conditions and preparation of standard samples (subcontractor: Hirosaki University) were conducted this fiscal year. In (1), a mid-infrared cavity ring-down test was conducted. An optical bench (3 m  1.2 m) was set up in the laboratory, …
1.2 m) was set up in the laboratory, …
Hamamoto, Shimpei; Ishitsuka, Etsuo; Nakagawa, Shigeaki; Goto, Minoru; Matsuura, Hideaki*; Katayama, Kazunari*; Otsuka, Teppei*; Tobita, Kenji*
Proceedings of 2021 International Congress on Advances in Nuclear Power Plants (ICAPP 2021) (USB Flash Drive), 5 Pages, 2021/10
Impurity concentrations of hydrogen and hydride in the coolant were investigated in detail for the HTTR, a block type high-temperature gas reactor owned by Japan. As a result, it was found that CH was 1/10 of H
was 1/10 of H concentration, which was under the conventional detection limit. If the ratio of H
concentration, which was under the conventional detection limit. If the ratio of H to CH
to CH in the coolant is the same as the ratio of HT to CH
in the coolant is the same as the ratio of HT to CH T, the CH
T, the CH T has a larger dose conversion factor, and this compositional ratio is an important finding for the optimal dose evaluation. Further investigation of the origin of CH
T has a larger dose conversion factor, and this compositional ratio is an important finding for the optimal dose evaluation. Further investigation of the origin of CH suggested that CH
suggested that CH was produced as a result of a thermal equilibrium reaction rather than being released as an impurity from the core.
was produced as a result of a thermal equilibrium reaction rather than being released as an impurity from the core.
Kenzhina, I.*; Ishitsuka, Etsuo; Ho, H. Q.; Sakamoto, Naoki*; Okumura, Keisuke; Takemoto, Noriyuki; Chikhray, Y.*
Fusion Engineering and Design, 164, p.112181_1 - 112181_5, 2021/03
Tritium release into the primary coolant during operation of the JMTR (Japan Materials Testing Reactor) and the JRR-3M (Japan Research Reactor-3M) had been studied. It is found that the recoil release by  Li(n
Li(n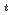 ,
, )
) H reaction, which comes from a chain reaction of beryllium neutron reflectors, is dominant. To prevent tritium recoil release, the surface area of beryllium neutron reflectors needs to be minimum in the core design and/or be shielded with other material. In this paper, as the feasibility study of the tritium recoil barrier for the beryllium neutron reflectors, various materials such as Al, Ti, V, Ni, and Zr were evaluated from the viewpoint of the thickness of barriers, activities after long-term operations, and effects on the reactivities. From the results of evaluations, Al would be a suitable candidate as the tritium recoil barrier for the beryllium neutron reflectors.
H reaction, which comes from a chain reaction of beryllium neutron reflectors, is dominant. To prevent tritium recoil release, the surface area of beryllium neutron reflectors needs to be minimum in the core design and/or be shielded with other material. In this paper, as the feasibility study of the tritium recoil barrier for the beryllium neutron reflectors, various materials such as Al, Ti, V, Ni, and Zr were evaluated from the viewpoint of the thickness of barriers, activities after long-term operations, and effects on the reactivities. From the results of evaluations, Al would be a suitable candidate as the tritium recoil barrier for the beryllium neutron reflectors.
Kasugai, Yoshimi; Sato, Koichi; Takahashi, Kazutoshi*; Miyamoto, Yukihiro; Kai, Tetsuya; Harada, Masahide; Haga, Katsuhiro; Takada, Hiroshi
JPS Conference Proceedings (Internet), 33, p.011144_1 - 011144_6, 2021/03
A spallation neutron source with a mercury target has been in operation at the Materials and Life Science Experimental Facility of J-PARC since 2008. The target vessel made of stainless steel is required to be exchanged periodically due to radiation damage etc. In this presentation, tritium gas release observed in the first series of exchange work in 2011 and the analytical results will be shown.
Kenzhina, I.*; Ishitsuka, Etsuo; Okumura, Keisuke; Ho, H. Q.; Takemoto, Noriyuki; Chikhray, Y.*
Journal of Nuclear Science and Technology, 58(1), p.1 - 8, 2021/01
Times Cited Count:1 Percentile:8.72(Nuclear Science & Technology)The sources and mechanisms for the tritium release into the primary coolant in the JMTR and the JRR-3M containing beryllium reflectors are evaluated. It is found that the recoil release from chain reaction of  Be is dominant and its calculation results agree well with trends derived from the measured variation of tritium concentration in the primary coolant. It also indicates that the simple calculation method used in this study for the tritium recoil release from the beryllium reflectors can be utilized for an estimation of the tritium release into the primary coolant for a research and testing reactors containing beryllium reflectors.
Be is dominant and its calculation results agree well with trends derived from the measured variation of tritium concentration in the primary coolant. It also indicates that the simple calculation method used in this study for the tritium recoil release from the beryllium reflectors can be utilized for an estimation of the tritium release into the primary coolant for a research and testing reactors containing beryllium reflectors.
Takiya, Hiroaki; Kadowaki, Haruhiko; Matsushima, Akira; Matsuo, Hidehiko; Ishiyama, Masahiro; Aratani, Kenta; Tezuka, Masashi
JAEA-Technology 2020-001, 76 Pages, 2020/05
Advanced Thermal Reactor (ATR) FUGEN was operated for about 25 years, and now has been proceeding decommissioning after the approval of the decommissioning plan in Feb. 2008. The reactor, heavy water system and helium system are contaminated by tritium because of neutron absorption of heavy water, which is a moderator. Before dismantling these facilities, it is necessary to remove tritium from them for not only reducing the amount of tritium released to surrounding environment and the risk of internal exposure by tritium but also ensuring the workability. In first phase of decommissioning (Heavy Water and Other system Decontamination Period), tritium decontamination of the reactor, heavy water system and helium system started in 2008 and completed in 2018. This report shows the results of tritium decontamination of the reactor, heavy water system and helium system.
Chikhray, Y.*; Askerbekov, S.*; Kenzhin, Y.*; Gordienko, Y.*; Ishitsuka, Etsuo
Fusion Science and Technology, 76(4), p.494 - 502, 2020/05
Times Cited Count:1 Percentile:8.72(Nuclear Science & Technology)Ishitsuka, Etsuo; Sakamoto, Naoki*
Physical Sciences and Technology, 6(2), p.60 - 63, 2019/12
Tritium release into the primary coolant of the research and test reactors during operation had been studied, and it is found that the recoil release from chain reaction of  Be is dominant. To reduce tritium concentration of the primary coolant, feasibility study of the tritium recoil barrier for the beryllium neutron reflectors was carried out, and the tritium recoils of various materials were calculated by PHITS. From these calculation results, it is clear that the thickness of tritium recoil barrier depends on the material and 20
Be is dominant. To reduce tritium concentration of the primary coolant, feasibility study of the tritium recoil barrier for the beryllium neutron reflectors was carried out, and the tritium recoils of various materials were calculated by PHITS. From these calculation results, it is clear that the thickness of tritium recoil barrier depends on the material and 20 40
40  m is required for three orders reduction.
m is required for three orders reduction.
Uchida, Shunsuke
Genshiryoku No Ima To Ashita, p.63 - 68, 2019/03
The latest situation of contaminated water treatment in Fukushima Daiichi NPP for 8 years after its accident is reviewed. Major subjects, especially tritium treatment, to be solved related to the contaminated water and some proposal for the subjects are introduced.
Ho, H. Q.; Ishitsuka, Etsuo
Physical Sciences and Technology, 5(2), p.53 - 56, 2019/00
Increasing of tritium concentration in the primary coolant of the research and test reactors during operation had been reported. To check the source for tritium release into the primary coolant during operation of the JMTR and the JRR-3M, the tritium release from the driver fuels was calculated by MCNP6 and PHITS. It is clear that the calculated values of tritium release from fuels are as about 10 and 10
and 10 Bq for the JMTR and JRR-3M, respectively, and that calculated values are about 4 order of magnitude smaller than that of the measured values. These results show that the tritium release from fuels is negligible for both the reactors.
Bq for the JMTR and JRR-3M, respectively, and that calculated values are about 4 order of magnitude smaller than that of the measured values. These results show that the tritium release from fuels is negligible for both the reactors.
Ishitsuka, Etsuo; Kenzhina, I.*; Okumura, Keisuke; Ho, H. Q.; Takemoto, Noriyuki; Chikhray, Y.*
JAEA-Technology 2018-010, 33 Pages, 2018/11
As a part of study on the mechanism of tritium release to the primary coolant in research and testing reactors, tritium recoil release rate from Li and U impurities in the neutron reflector made by beryllium, aluminum and graphite were calculated by PHITS code. On the other hand, the tritium production from Li and U impurities in beryllium neutron reflectors for JMTR and JRR-3M were calculated by MCNP6 and ORIGEN2 code. By using both results, the amount of recoiled tritium from beryllium neutron reflectors were estimated. It is clear that the amount of recoiled tritium from Li and U impurities in beryllium neutron reflectors are negligible, and 2 and 5 orders smaller than that from beryllium itself, respectively.
Goto, Minoru; Okumura, Keisuke; Nakagawa, Shigeaki; Inaba, Yoshitomo; Matsuura, Hideaki*; Nakaya, Hiroyuki*; Katayama, Kazunari*
Fusion Engineering and Design, 136(Part A), p.357 - 361, 2018/11
Times Cited Count:8 Percentile:56.25(Nuclear Science & Technology)A High Temperature Gas-cooled Reactor (HTGR) is proposed as a tritium production device, which has the potential to produce a large amount of tritium using  Li(n,
Li(n, )T reaction. In the HTGR design, generally, boron is loaded into the core as a burnable poison to suppress excess reactivity. In this study, lithium is loaded into the HTGR core instead of boron and is used as a burnable poison aiming to produce thermal energy and tritium simultaneously. The nuclear characteristics and the fuel temperature were calculated to confirm the feasibility of the lithium-loaded HTGR. It was shown that the calculation results satisfied the design requirements and hence the feasibility was confirmed for the lithium-loaded HTGR, which produce thermal energy and tritium.
)T reaction. In the HTGR design, generally, boron is loaded into the core as a burnable poison to suppress excess reactivity. In this study, lithium is loaded into the HTGR core instead of boron and is used as a burnable poison aiming to produce thermal energy and tritium simultaneously. The nuclear characteristics and the fuel temperature were calculated to confirm the feasibility of the lithium-loaded HTGR. It was shown that the calculation results satisfied the design requirements and hence the feasibility was confirmed for the lithium-loaded HTGR, which produce thermal energy and tritium.
Kai, Tetsuya; Uchida, Toshitsugu; Kinoshita, Hidetaka; Seki, Masakazu; Oi, Motoki; Wakui, Takashi; Haga, Katsuhiro; Kasugai, Yoshimi; Takada, Hiroshi
Journal of Physics; Conference Series, 1021(1), p.012042_1 - 012042_4, 2018/06
Times Cited Count:0 Percentile:0.00(Nuclear Science & Technology)Ishitsuka, Etsuo; Kenzhina, I. E.*
Physical Sciences and Technology, 4(1), p.27 - 33, 2018/06
Increase of tritium concentration in the primary coolant for the research and testing reactors during reactor operation had been reported. To clarify the tritium sources, a curve of the tritium release rate into the primary coolant for the JMTR and the JRR-3M are evaluated. It is also observed that the amount of released tritium is lower in the case of new beryllium components installation, and increases with the reactor operating cycle. These results show the beryllium components in core strongly affect to the tritium release into the primary coolant. As a result, the tritium release rate is related with produced  Li by (n,
Li by (n, ) reaction from
) reaction from  Be, and evaluation results of tritium release curve are shown as the dominant source of tritium release into the primary coolant for the JMTR and the JRR-3M are beryllium components. Scattering of the tritium release rate with irradiation time were observed, and this phenomena in the JMTR occurred in earlier time than that of the JRR-3M.
Be, and evaluation results of tritium release curve are shown as the dominant source of tritium release into the primary coolant for the JMTR and the JRR-3M are beryllium components. Scattering of the tritium release rate with irradiation time were observed, and this phenomena in the JMTR occurred in earlier time than that of the JRR-3M.
Ota, Masakazu; Kwamena, N.-O. A.*; Mihok, S.*; Korolevych, V.*
Journal of Environmental Radioactivity, 178-179, p.212 - 231, 2017/11
Times Cited Count:18 Percentile:46.91(Environmental Sciences)Environmental transfer models assume that organically-bound tritium (OBT) is formed directly from tissue-free water tritium (TFWT) in environmental compartments. Nevertheless, studies in the literature have shown that measured OBT/TFWT ratios are variable. The importance of soil-to-leaf HTO transfer pathway in controlling the leaf tritium dynamics is not well understood. A model inter-comparison of two tritium transfer models (CTEM-CLASS-TT and SOLVEG-II) was carried out with measured environmental samples from an experimental garden plot set up next to a tritium-processing facility. The garden plot received one of three different irrigation treatments - no external irrigation, irrigation with low tritium water and irrigation with high tritium water. The contrast between the results obtained with the different irrigation treatments provided insights into the impact of soil-to-leaf HTO transfer on the leaf tritium dynamics. Concentrations of TFWT and OBT in the garden plots that were not irrigated or irrigated with low tritium water were variable, responding to the arrival of the HTO-plume from the tritium-processing facility. In contrast, for the plants irrigated with high tritium water, the TFWT concentration remained elevated due to a continuous source of high HTO in the soil. Calculated concentrations of OBT in the leaves showed an initial increase followed by quasi-equilibration with the TFWT concentration. In this quasi-equilibrium state, concentrations of OBT remained elevated and unchanged despite the arrivals of the plume. These results from the model inter-comparison demonstrate that soil-to-leaf HTO transfer significantly affects OBT/TFWT ratio in the leaf regardless of the atmospheric HTO concentration, only if there is elevated HTO concentrations in the soil. The results of this work indicate that assessment models should be refined to consider the importance of soil-to-leaf HTO transfer to ensure that dose estimates are accurate and conservative.
Edao, Yuki; Sato, Katsumi; Iwai, Yasunori; Hayashi, Takumi
Journal of Nuclear Science and Technology, 53(11), p.1831 - 1838, 2016/11
Times Cited Count:9 Percentile:59.71(Nuclear Science & Technology)Ishitsuka, Etsuo; Kenzhina, I. E.*; Okumura, Keisuke; Takemoto, Noriyuki; Chikhray, Y.*
JAEA-Technology 2016-022, 35 Pages, 2016/10
As a part of study on the mechanism of tritium release to the primary coolant in research and testing reactors, the calculation methods by PHITS code is studied to evaluate the recoil tritium release rate from beryllium core components. Calculations using neutron and triton sources were compared, and it is clear that the tritium release rates in both cases show similar values. However, the calculation speed for the triton source cases is two orders faster than that for the neutron source case. It is also clear that the calculation up to history number per unit volume of 2 10
10 (cm
(cm ) is necessary to determine the recoil tritium release rate of two effective digits precision. Furthermore, the relationship between the beryllium shape and recoil tritium release rate using the triton sources was studied. Recoil tritium release rate showed linear relation to the surface area per volume of beryllium, and the recoil tritium release rate showed about half of the conventional equation value.
) is necessary to determine the recoil tritium release rate of two effective digits precision. Furthermore, the relationship between the beryllium shape and recoil tritium release rate using the triton sources was studied. Recoil tritium release rate showed linear relation to the surface area per volume of beryllium, and the recoil tritium release rate showed about half of the conventional equation value.
Dipu, A. L.; Ohashi, Hirofumi; Hamamoto, Shimpei; Sato, Hiroyuki; Nishihara, Tetsuo
Annals of Nuclear Energy, 88, p.126 - 134, 2016/02
Times Cited Count:6 Percentile:45.07(Nuclear Science & Technology)The tritium concentration in the high temperature engineering test reactor (HTTR) was measured during the high temperature continuous operation for 50 days. The tritium concentration in the primary helium gas increased after startup and reached a maximum value. It then decreased slightly over the course during the normal operation phase. Decrease of concentration of tritium in primary helium gas during the normal operation phase could be attributed to the effect of tritium chemisorption on graphite. The tritium concentration in the secondary helium gas showed a peak value during the power ramp up phase. Afterwards, it decreased gradually at the end of normal power operation. It was assessed that the concentration and total quantity of tritium in the secondary helium cooling system for the HTTR-Iodine Sulfur (IS) system can be maintained below the regulatory limits, which means the hydrogen production plant can be exempt from the safety function of the nuclear facility.
Yamanishi, Toshihiko
Purazuma, Kaku Yugo Gakkai-Shi, 92(1), p.21 - 25, 2016/01
In a fusion reactor, the hydrogen isotope separation system is required in the fuel cycle system to supply deuterium (D) and tritium (T) as its fuel. In ITER, 90% of T must be recycled through the isotope separation system. On the other hand; since the hydrogen (H) gas is finally exhausted to the environment, the T concentration in the H gas from the isotope separation system should be as low as reasonable achievable. Hence, the isotope separation system of a fusion reactor must have a large separation factor. The flow rate of the isotope separation system of a fusion reactor reaches to 300 mol/h. Only the cryogenic distillation method can meet the above conditions (large flow rate and separation factor) and is most likely used as a hydrogen separation system in a fusion reactor. In this chapter, several simulation methods and a set of experimental data of the cryogenic distillation columns are described in detail.