Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tobita, Minoru*; Haraga, Tomoko; Endo, Tsubasa*; Omori, Hiroyuki*; Mitsukai, Akina; Aono, Ryuji; Ueno, Takashi; Ishimori, Kenichiro; Kameo, Yutaka
JAEA-Data/Code 2021-013, 30 Pages, 2021/12
Radioactive wastes generated from nuclear research facilities in Japan Atomic Energy Agency are planning to be buried in the near surface disposal field. Therefore, it is required to establish the method to evaluate the radioactivity concentrations of radioactive wastes until the beginning of disposal. In order to contribute to this work, we collected and analyzed concrete samples generated from JPDR facility. In this report, we summarized the radioactivity concentrations of 21 radionuclides ( H,
H,  C,
C, 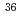 Cl,
Cl, 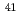 Ca,
Ca,  Co,
Co,  Ni,
Ni,  Sr,
Sr, 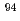 Nb,
Nb, 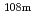 Ag,
Ag,  Cs,
Cs, 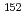 Eu,
Eu, 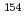 Eu,
Eu, 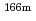 Ho,
Ho,  U,
U,  U,
U,  Pu,
Pu,  Pu,
Pu, 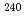 Pu,
Pu,  Am,
Am,  Am,
Am,  Cm) which were obtained from radiochemical analysis of the samples in fiscal year 2018-2019.
Cm) which were obtained from radiochemical analysis of the samples in fiscal year 2018-2019.
Tsuchida, Daiki; Haraga, Tomoko; Tobita, Minoru*; Omori, Hiroyuki*; Omori, Takeshi*; Murakami, Hideaki*; Mitsukai, Akina; Aono, Ryuji; Ishimori, Kenichiro; Kameo, Yutaka
JAEA-Data/Code 2020-022, 34 Pages, 2021/03
Radioactive wastes generated from nuclear research facilities in Japan Atomic Energy Agency are planning to be buried in the near surface disposal field. Therefore, it is required to establish the method to evaluate the radioactivity concentrations of radioactive wastes until the beginning of disposal. In order to contribute to this work, we collected and analyzed concrete samples generated from JRR-3 and JPDR. In this report, we summarized the radioactivity concentrations of 22 radionuclides( H,
H,  C,
C, 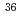 Cl,
Cl, 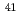 Ca,
Ca,  Co,
Co,  Ni,
Ni,  Sr,
Sr, 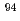 Nb,
Nb, 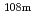 Ag,
Ag,  Ba,
Ba,  Cs,
Cs, 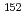 Eu,
Eu, 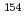 Eu,
Eu, 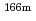 Ho,
Ho,  U,
U,  U,
U,  Pu,
Pu, 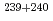 Pu,
Pu,  Am,
Am,  Am,
Am,  Cm) which were obtained from radiochemical analysis of the samples.
Cm) which were obtained from radiochemical analysis of the samples.
Tobita, Minoru*; Haraga, Tomoko; Sasaki, Takayuki*; Seki, Kotaro*; Omori, Hiroyuki*; Kochiyama, Mami; Shimomura, Yusuke; Ishimori, Kenichiro; Kameo, Yutaka
JAEA-Data/Code 2019-016, 72 Pages, 2020/02
In the future, radioactive wastes which generated from research and testing reactors in Japan Atomic Energy Agency are planning to be buried for the near surface disposal. Therefore, it is required to establish the method to evaluate the radioactivity concentrations of radioactive wastes by the time it starts disposal. In order to contribute to this work, we collected and analyzed the samples generated from JRR-2, JRR-3 and Hot laboratory facilities. In this report, we summarized the radioactivity concentrations of 25 radionuclides ( H,
H,  C,
C, 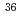 Cl,
Cl,  Co,
Co,  Ni,
Ni,  Sr,
Sr, 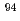 Nb,
Nb,  Mo,
Mo,  Tc,
Tc, 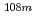 Ag,
Ag, 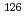 Sn,
Sn,  I,
I,  Cs,
Cs, 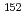 Eu,
Eu, 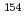 Eu,
Eu, 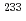 U,
U,  U,
U,  U,
U,  Pu,
Pu,  Pu,
Pu, 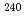 Pu,
Pu,  Pu,
Pu,  Am,
Am,  Am,
Am,  Cm) which were obtained from radiochemical analysis of those samples.
Cm) which were obtained from radiochemical analysis of those samples.
 Mo content in metal waste generated at the Japan Power Demonstration Reactor
Mo content in metal waste generated at the Japan Power Demonstration ReactorShimada, Asako; Omori, Hiroyuki*; Kameo, Yutaka
Journal of Radioanalytical and Nuclear Chemistry, 314(2), p.1361 - 1365, 2017/11
Times Cited Count:3 Percentile:28.82(Chemistry, Analytical)A separation method of Mo from Nb, Zr, and the matrix elements of rubble waste was modified to determine the content of  Mo in metal waste. A separation scheme to treat 1 g of metal waste was established by optimizing the amount of ascorbic acid, the rinsing solution, and repeating of the procedure. A thin-layer source was prepared using direct drop deposition and evaporation to measure
Mo in metal waste. A separation scheme to treat 1 g of metal waste was established by optimizing the amount of ascorbic acid, the rinsing solution, and repeating of the procedure. A thin-layer source was prepared using direct drop deposition and evaporation to measure  Mo content. Finally,
Mo content. Finally,  Mo content in the metal waste generated at the Japan Power Demonstration Reactor was analyzed using the developed method.
Mo content in the metal waste generated at the Japan Power Demonstration Reactor was analyzed using the developed method.
Omori, Hiroyuki; Nebashi, Koji; Shimada, Asako; Tanaka, Kiwamu; Yasuda, Mari; Hoshi, Akiko; Tsuji, Tomoyuki; Ishimori, Kenichiro; Kameo, Yutaka
JAEA-Data/Code 2014-029, 31 Pages, 2015/03
Simple and rapid methods to evaluate the radioactivity concentrations are required for the radioactive waste generated from research facilities in the Japan Atomic Energy Agency to dispose of in a near-surface repository. In order to establish the methods to evaluate the radioactivity concentrations of miscellaneous solid waste generated from research and testing reactors, we collected and analyzed samples from miscellaneous solid waste generated by the decommissioning of JPDR (Japan Power Demonstration Reactor). In this report, we reported the analytical data determined in fiscal 2014 ( Cs and
Cs and  Mo) and summarized them with the radioactivity concentrations obtained in the past as basic data to consider the evaluation method of radioactivity concentrations in the stored waste taken from JPDR.
Mo) and summarized them with the radioactivity concentrations obtained in the past as basic data to consider the evaluation method of radioactivity concentrations in the stored waste taken from JPDR.
Maeda, Toshikatsu; Watanabe, Koichi; Omori, Hiroyuki*; Sakamaki, Keiko; Inagaki, Yaohiro*; Idemitsu, Kazuya*
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 21(2), p.63 - 74, 2014/12
Static leach tests were conducted for simulated HLW glass in CaCl /Ca(OH)
/Ca(OH) solutions to investigate the corrosion behavior of HLW glass under calcium-rich environments induced by cement based materials in geological repositories. Another series of leach tests were conducted in deionized water in the presence of iron to investigate the effects of iron over-pack on the glass corrosion. In Ca solutions, corrosion of the glass was inhibited during the test period compared to that in deionized water, while the corrosion was enhanced at the presence of iron. The enhancement of the glass corrosion was assumed to be accompanied with transformation of silica, a glass network former, into iron silicates.
solutions to investigate the corrosion behavior of HLW glass under calcium-rich environments induced by cement based materials in geological repositories. Another series of leach tests were conducted in deionized water in the presence of iron to investigate the effects of iron over-pack on the glass corrosion. In Ca solutions, corrosion of the glass was inhibited during the test period compared to that in deionized water, while the corrosion was enhanced at the presence of iron. The enhancement of the glass corrosion was assumed to be accompanied with transformation of silica, a glass network former, into iron silicates.
Maeda, Toshikatsu; Omori, Hiroyuki*
no journal, ,
no abstracts in English
Omori, Hiroyuki; Maeda, Toshikatsu; Mitsui, Seiichiro; Bamba, Tsunetaka
no journal, ,
no abstracts in English
Maeda, Toshikatsu; Omori, Hiroyuki; Mitsui, Seiichiro
no journal, ,
Static leach tests were conducted for P0798 type simulated HLW glass in magnesium chloride solution to investigate the effects of Mg ion on the glass corrosion. The glass corroded at higher rate under the condition that Mg ion exists in the leachate, while the rate dropped after the depletion of Mg ion in the leachate. Surface analyses showed that altered layer including Mg and Si was observed at the surface of the leached glass. The present results implied that corrosion of the glass with the higher rate occurred as dissolution of Si from the glass network accompanied with formation of magnesium silicates. Corrosion rate of the HLW glass would be low for long-term if cations forming silicate compounds are scarce in groundwater.
Maeda, Toshikatsu; Bamba, Tsunetaka*; Omori, Hiroyuki; Yamaguchi, Tetsuji
no journal, ,
no abstracts in English
Maeda, Toshikatsu; Omori, Hiroyuki; Yamaguchi, Tetsuji; Kataoka, Masaharu; Bamba, Tsunetaka*
no journal, ,
no abstracts in English
Maeda, Toshikatsu; Omori, Hiroyuki; Sakamaki, Keiko
no journal, ,
no abstracts in English